Introduction
Aquaculture has risen as a rapidly expanding industry globally, responding to the growing demand for seafood products (Wang et al., 2024). The ongoing proliferation of global fish farming is concomitant with a rising prominence of parasites as pathogenic factors. Notably, Trichodina sp., a prevalent parasite residing in the gills and skin of fish, represents the largest cluster among fish parasites and plays a pivotal role in fish pathology. These parasites possess the capacity to compromise the physiological well-being of fish, resulting in mortality via infestations on gills and the hindrance of oxygen (O2) exchange across the gill lamellae (Yao et al., 2011). The white-spotted walking catfish (Clarias fuscus) stands out as a highly favored freshwater catfish species in Vietnam, albeit prone to trichodinosis—a protozoan ailment attributable to Trichodinanigra. This parasitic organism is frequently localized on the fish’s skin and gills, giving rise to abrasions, profound ulceration, tissue impairment, and immunosuppression, thereby fostering secondary infections. The augmented incidence of trichodinosis triggers oxidative stress within the fish (Tran & Tran, 2023). Oxidative stress arises when a significant quantity of free radicals, encompassing free O2 ions, hydrogen peroxide (H2O2), and reactive oxygen species (ROS), is generated. This exerts detrimental effects on cellular structures, and other biomolecules, causing DNA, protein, and lipid damage, and influencing internal organs within the fish’s body. Oxidative stress disrupts the metabolic equilibrium in the fish’s body, reducing the capacity for nutrient absorption and metabolism, leading to an energy imbalance. In response, the fish’s organism upregulates the production of antioxidant enzymes, such as superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx), aiming to mitigate the impact of oxidative stress. This mechanism serves as a natural defense against the adverse effects of diseases and stress. However, unchecked persistence of this condition can result in damage and contribute to the degradation of the fish’s health (Menon et al., 2023). Formaldehyde is a widely employed agent for the management of Trichodina sp. infections; however, its effectiveness has shown a decline in recent years. Alternative chemicals, such as malachite green, potassium permanganate, acriflavine, and bithionol, have been applied to combat Trichodina sp. Nevertheless, the frequent utilization of these chemical agents has brought about significant drawbacks, including the emergence of drug-resistant parasites, environmental contamination, residue accumulation, and imposition of stress on the host organism (Yao et al., 2011). Furthermore, synthetic antioxidants like ethoxyquin and butylated hydroxytoluene, extensively utilized in aquaculture, have raised safety concerns for both human consumers and cultured animals (Wang et al., 2024). Consequently, the introduction of antioxidants devoid of adverse effects is imperative for the viability and sustainability of aquaculture practices. Ongoing research indicates that integrating plant-derived antioxidants into aquaculture operations has the potential to augment the stress resilience of cultured organisms (Tadese et al., 2021).
The Bead tree (Melia azedarach Linn.) represents a versatile botanical species originating in West Asia. Currently, it is cultivated in multiple countries, including India, Pakistan, Nepal, Sri Lanka, East Timor, Indonesia, Lebanon, Palestine, Syria, Tunisia, Algeria, Cyprus, Greece, Argentina, China, Uganda, Kenya, Brazil, Australia, Southern France, Northern Italy, Croatia, and Portugal, where the climate ranges from mild to warm (Dias et al., 2022). Extracts obtained from various components of M. azedarach have exhibited both pharmacological and toxicological properties. The leaves are particularly abundant in bioactive compounds such as flavonoids, terpenoids, limonoids, fatty acids, carbohydrates, steroids, alkaloids, saponins, and tannins. Both leaves and seeds of M. azedarach showcase remarkable antibacterial, antifungal, antiparasitic, antiviral, and insecticidal properties (Khoshraftar et al., 2020). The leaves, roots, bark, and flowers of M. azedarach demonstrate a wide spectrum of antibacterial activities, with the dichloromethane fraction from the bark being particularly effective (Peng et al., 2021). Various parts of M. azedarach, including stem bark, leaves, fruit, and seeds, have been integral in traditional medicine for addressing diverse gastrointestinal issues, vomiting, bloody diarrhea, mitigating oxidative stress, and fostering angiogenesis. Additionally, M. azedarach has been acknowledged for its roles in antioxidant activity, cytotoxicity, wound healing, anthelmintic effects, anti-protozoal properties, insect repellency, and antibacterial actions (M’rabet et al., 2017). The influence of the methanol leaf extract of M. azedarach on bacterial membrane integrity, diverse O2 reactions, lipid peroxidation, and the modulation of oxidative stress enzymes (SOD and CAT) has been experimentally validated (Touzout et al., 2023). The antioxidative and stress-alleviating properties of M. azedarach leaf extract have been underscored. The incorporation of M. azedarach leaves in aquaculture is gaining traction. However, the stress-mitigating and immunosuppression effects of M. azedarach in aquatic organisms remain unexplored. This study seeks to elucidate the influence of M. azedarach leaf extract on bolstering oxidative stress resistance and immunosuppression induced by T. nigra in White-spotted freshwater catfish (C. fuscus). The outcomes of this investigation contribute to the continuous exploration and application of herbal extracts in aquaculture.
Materials and Methods
The M. azedarach leaves were procured in May 2023 from Hung Dinh commune, Thuan An District, Binh Duong province, Vietnam. A voucher specimen (code MA140523VST) has been archived at the Plant Biotechnology Laboratory, Institute of Biotechnology and Food Technology, Industry University of Ho Chi Minh City, for future identification and reference. The freshly collected leaves underwent a thorough cleansing process to eliminate dust and impurities. Subsequently, they were subjected to air-drying at room temperature, followed by additional drying in a Memmert drying cabinet (Memmert GmbH + Co. KG, Germany) at 50°C for 3 days. The desiccated leaves were crudely pulverized using an herbal grinder TMND-A01 (Tan Minh, Ha Noi, Vietnam). The resulting leaf powder was carefully stored in desiccant bags at room temperature, shielded from light, and earmarked for subsequent laboratory investigations.
Roughly 1,000 g of M. azedarach crude powder was immersed in absolute ethanol and allowed to stand undisturbed for 48 h. The resultant ethanol extract underwent filtration through muslin cloth on a glass wool plug within a glass column, followed by additional filtration using Whatman No. 4 filter paper (Cytiva, Wilmington, DE, USA). The filtrate was concentrated and evaporated at 45°C utilizing a rotary evaporator RV 10 Digital V-C (IKA, Staufen, Germany) to preserve the integrity of active components. Following this, the resulting concentrated extract (200 g, yield of ethanol extract of 20%), denoted as M. azedarach leaf ethanol extract (MAEE), was diluted to 1,000 mL using distilled water and stored in a refrigerator at 4°C for subsequent research purposes.
Phytochemical analysis was qualitatively performed to identify compounds in the ethanol extract obtained from M. azedarach leaves, following established methodologies outlined by Tran et al. (2023) (Table 1).
Polyphenols, flavonoids, and tannins are crucial actors in plant physiology, serving as robust antioxidants to shield against oxidative stress and showcasing protozoan-killing attributes. These compounds bolster the plant’s defense mechanisms against Trichodina sp. invasion. Precise quantification of these components in plant extracts is imperative and was executed employing well-established methods as detailed by Nhung (2024). The quantification of total polyphenol content was carried out using the Folin-Ciocalteu method, expressed as milligrams of gallic acid equivalent per gram of dried plant material (mg GAE/g) according to the gallic acid standard. Total flavonoid content was measured using the aluminum chloride method, with results presented in milligrams of quercetin equivalent per gram of dried plant material (mg QE/g) based on the quercetin standard. Tannin content was determined reliably through the vanillin spectrophotometric method, and the total condensed tannin content was computed as milligrams of catechin equivalent (mg CE/g) using the standard curve. These established methodologies yield robust data for quantifying the levels of polyphenols, flavonoids, and tannins in plant extracts, offering valuable insights into their potential roles in enhancing of C. fuscus health and fortifying defense mechanisms against damage induced by Trichodina sp.
The fish selected for this investigation were White-spotted freshwater catfish (C. fuscus L.) weighing between 45 and 50 g, sourced from the Tư Hải fish hatchery located in Tân An Hội commune, Củ Chi district, Ho Chi Minh City, Vietnam. After transportation to the Laboratory of Animal Biotechnology at the Institute of Biotechnology and Food Technology, Industry of Ho Chi Minh City University, the fish underwent a 3-day acclimation period. A total of 120 disease-free and healthy fish were randomly assigned to various experimental tanks (10 fish per tank). In each tank, the fish were fed a commercial diet for freshwater catfish and supplemented with frozen bloodworms at a rate of 1% of their body weight per day. Excess feed and waste were systematically removed before water changes. The water level was consistently maintained at 20 L per tank. Adherence to suitable water quality for fish cultivation was achieved by maintaining parameters within the following ranges: temperature at 28°C–30°C, pH at 7.55 ± 0.11, dissolved O2 at 4.35 ± 0.21 mg/L, and total alkalinity at 75.52 ± 0.16 mg/L, per the natural light-dark cycle (Tran & Tran, 2023). The care and use procedure of experimental fishes were strictly followed the Guidelines of Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA) for Experimentation on Fishes (CPCSEA, 2021). Every effort was made to minimize potential distress experienced by the animals.
Preparation of T. nigra cell suspension: T. nigra trophonts were provided by the Department of biotechnology, faculty of natural sciences, Hanoi National University, Vietnam. The parasites were induced and multiplied on the skin of freshwater catfish, and the mucous secretion from infected fish was used in subsequent experiments.
In an in vivo experiment designed according to the methodology outlined by Tran & Tran (2023), the effectiveness of M. azedarach leaf ethanol extract (MAEE) in mitigating oxidative stress induced by T. nigra in C. fuscus was assessed. A total of 60 healthy male walking catfish were randomly assigned to six experimental treatments (10 fish/treatment). Control treatments included catfish immersed in plain water without treatment with the extract or T. nigra infection. The remaining fish were infected with T. nigra cell suspension and observed for 24 h, following which they were categorized into five treatments: untreated, formalin (100 ppm), and three extract doses MAEE (MAEE800, MAEE900, and MAEE1000). All fish were exposed to tanks containing either plain water (control and untreated) or formalin (100 ppm) and extract solution (800, 900, and 1,000 ppm MAEE) for 15 min each day for 30 days. Various experimental parameters were continuously monitored and recorded throughout the study.
After the experiment, sample collection and preparation were performed using the methodology outlined by Enis Yonar et al. (2011). Blood was collected from the tail vein of each fish after anesthesia with benzocaine (25 mg/L in water). Tissue samples (liver, kidney, gill, and heart) were obtained from each fish after blood sampling. The concentrations of malondialdehyde (MDA), H2O2, glutathione (GSH), ascorbic acid (AsA), CAT, GPx, SOD, and glutathione reductase (GR) were determined in liver, kidney, gill, and heart tissues to assess the oxidative-antioxidative status. White blood cell count (WBC), oxidative burst (ROS activity), total serum protein (TP), total immunoglobulin (TI), and PA were measured in blood and serum to evaluate the immune response. A portion of whole blood from heparinized syringes was used for assessing WBC activity, PA, and ROS. The residual blood fraction underwent serum separation through centrifugation at 1,500 g for 5 min at 4°C. Subsequently, the serum samples were employed for TP and TI determination. The washed red blood cells were lysed using cold distilled water, and this resulting hemolysate was utilized for evaluating biochemical activities. Upon blood collection, liver, kidney, gill, and heart samples were immediately and meticulously excised, rinsed with physiological saline solution (0.9% NaCl), and stored at –80°C until biochemical analysis, conducted within one month of extraction. The tissues were homogenized using Teflon-glass homogenizers in a 1:10 (w/v) uniform buffer solution containing 1.15% KCl. The resulting homogenates underwent centrifugation at 18,000 g (4°C) for 30 min to determine MDA levels, H2O2 levels, GSH concentration, AsA concentration, and the activities of CAT, GPx, GR, and SOD.
The MDA concentration was determined using the thiobarbituric acid reaction. The quantification of substances reacting with thiobarbituric acid was assessed by comparing the absorbance with the standard curve of MDA equivalents, generated through the hydrolysis of 1,1,3,3-tetramethoxypropane with acid catalysis (Enis Yonar et al., 2011).
The assessment of H2O2 content adhered to the protocol detailed by Sinha et al. (2015). To quantify H2O2, 50 mg of liver tissues were homogenized in a 50 mM phosphate buffer (pH 6.5) and subjected to analysis using the FOX1 (ferrous oxidation-xylenol orange) method.
The GSH concentration was quantified using a recycling assay with dithionitrobenzoic acid, following the methodology outlined by Enis Yonar et al. (2011). In this procedure, the absorbance at 412 nm of the colored product resulting from the reaction between the DTNB reagent and non-erythrocyte sulfhydryl groups was measured. One milliliter of the sample underwent deproteinization by adding a solution containing 1.67 g metaphosphoric acid, 0.2 g Na2EDTA, and 30 g NaCl in distilled water. Na2HPO4 (2.4 mL) and 0.3 mL DTNB were added to the upper layer and subjected to centrifugation (10 min, 3,000 g/min). The formation of 5-thio-2-nitrobenzoic acid, directly proportional to the GSH concentration, was monitored at 412 nm, at 25°C, with a blank control of the reagent.
The methodology described by Sinha et al. (2015) was utilized to quantify the content of AsA. Homogenization of 0.5 g of tissues was achieved using a 50 mM tris buffer solution with a pH of 10.0, followed by centrifugation for 20 min at 13,000 rpm. Homogenized tissues of 0.5 mL, 100 mg of wood charcoal, 4 mL of distilled deionized water, and 0.5 mL of 50% TCA, underwent an addition and subsequent filtration using Whatman No. 4 filter paper (Cytiva). Incubation took place at 37°C, initiated 3 h after introducing 0.4 mL of 2,4-dinitrophenylhydrazine solution and 1.6 mL of cold (65%) H2SO4 to the mixture. The mixture was then kept at room temperature for 30 min. Results were recorded at an absorbance of 520 nm using a spectrophotometer, with AsA (1 mg/100 mL) serving as the standard solution.
CAT activity was assessed following the methodology outlined by Enis Yonar et al. (2011). The test is based on determining the rate constant of H2O2 decomposition by the CAT enzyme. In the experiment, 2 mL of the sample was introduced to 1 mL of 40 mM H2O2 in a phosphate buffer solution (50 mM, pH 7.0, adjusted by combining 0.681 g KH2PO4 in 100 mL and 1.335 g Na2HPO4·2H2O in 150 mL). The reduction of H2O2 was quantified through spectrophotometric analysis at a wavelength of 240 nm over a 3-minute duration.
The GPx activity was assessed using the method outlined by Enis Yonar et al. (2011), measuring the decline in NADPH absorbance at 340 nm. The reaction mix comprised a 50 mM potassium phosphate buffer (pH 7.0), 1 mM EDTA, 1 mM sodium azide (NaN3), 0.2 mM NADPH, 1 EU/mL GPx, 1 mM GSH, and 0.25 mM H2O2. An enzyme source (0.1 mL) was introduced to 0.8 mL of this mixture and incubated at 25°C for 5 min before initiating the reaction with 0.1 mL of peroxide solution. The absorbance at 340 nm was recorded over 5 min, and the enzyme activity was subsequently computed based on the slope of the curves in micromoles of NADPH oxidized per minute.
The SOD activity was assessed using the methodology outlined by Enis Yonar et al. (2011). The underlying principle involves the reaction of xanthine with xanthine oxidase, leading to the generation of superoxide radicals (O2•−). These radicals subsequently react with nitrobluetetrazolium, resulting in the formation of the dye formazan. To execute this, a 600 mL reaction mixture, consisting of 0.1 mM xanthine, 0.1 mM EDTA, 50 mg bovine serum albumin, and 25 mmol nitrobluetetrazolium per liter, was added to either 125 mL of the upper layer or 125 mL of the SOD standard solution. Subsequently, 25 mL of a 9.9 nM xanthine oxidase solution was introduced into each tube at 30-second intervals. The incubation period for each tube was 20 min at 25°C, and the reaction was halted by adding 0.5 mL of 0.8 mM CuCl2 solution to each tube every 30 seconds. The production of formazan was quantified at a wavelength of 560 nm.
GR activity was assayed following the protocol outlined by Tkachenko et al. (2014). Enzyme activity was assessed using a spectrophotometric method by measuring the consumption of NADPH. In the presence of GSSG and NADPH, GR reduces GSSG while oxidizing NADPH, resulting in a decrease in absorbance at 340 nm. Quantification was based on the molar extinction coefficient of NADPH, which is 6.22 mM/cm. One unit of GR was defined as the amount of enzyme required to reduce 1 μmol of NADPH with GSSG per minute. GR activities were expressed as units per milligram of protein.
The TAC was assessed using spectrophotometric measurements at a wavelength of 532 nm following the Tween 80 oxidation method (Tkachenko et al., 2014). In brief, 0.2 mL of homogenized tissue was combined with 2 mL of 1% Tween 80. For the blank test, 0.1 mL of distilled water replaced the sample. This mixture was incubated for 48 h at 37°C. After cooling, 1 mL of 40% TCA was introduced. The resulting mixture underwent centrifugation at 3,000 g for 10 min. Post-centrifugation, 2 mL of the upper layer was mixed with 2 mL of 0.25% TBA reagent. The blend was then heated in a boiling water bath at 100°C for 15 min. The absorbance of the resultant solution was measured at 532 nm and compared to the blank sample. TAC levels were expressed as a percentage.
In the WBC quantification process, a hemocytometer and Natt and Herrick solution were employed (Enis Yonar et al., 2011). WBC were fixed and stained with the Natt-Herrick solution. The enumeration of WBC was calculated from hemocytometer counts.
NBT serves as the substrate utilized for ROS assays. To assess the generation of neutral pH-dependent ROS by neutrophils, quantified through the NBT assay, 0.1 mL of blood was introduced into a microtiter plate well, followed by the addition of an equivalent volume of 0.2% NBT solution. Following a 30-minute incubation at room temperature, 0.05 mL of the NBT-stained blood cell suspension was extracted and introduced into a glass tube containing 1.0 mL N,N-dimethylformamide. After centrifugation, the absorbance of the sample was measured at a wavelength of 620 nm using a 1.0 mL cuvette. Typically, the higher the absorbance, the greater the production of ROS. This is because NBT is reduced by ROS to form a colored formazan product, and the intensity of the color correlates with the amount of ROS generated. Therefore, by measuring the absorbance of the NBT-stained sample at a specific wavelength, usually at 620 nm, the concentration or level of ROS produced can be quantified (Enis Yonar et al., 2011).
The concentration of TP was determined by employing the Biuret method. In a spectrophotometer tube, 0.1 mL of plasma was mixed with 0.9 mL of distilled water, ensuring thorough homogenization by inversion. Following this, 4.0 mL of Biuret reagent was introduced into the tube, and the resultant mixture was allowed to incubate for 30 min at room temperature in the absence of light. Subsequently, the absorbance of the samples was measured at a wavelength of 540 nm using a spectrophotometer. In the Biuret method, the standard curve is typically represented by the equation: Absorbance = m × [Protein] + b. In which: 1/ Absorbance - represents the sample’s absorbance measured at a wavelength of 540 nm. 2/ [Protein] - denotes the protein concentration (mg/mL) of the sample. 3/ m - signifies the slope of the standard curve, illustrating the linear relationship between absorbance and protein concentration. 4/ b - representing the absorbance value when the protein concentration is 0 (Enis Yonar et al., 2011).
The determination of TI levels was conducted following the protocol outlined by Enis Yonar et al. (2011). This method involves quantifying the overall protein concentration in the serum using the protein determination technique, both before and after precipitating immune globulin molecules with a 12% polyethylene glycol solution. The disparity in protein content represents the TI content in the serum.
Blood immediately treated with heparin was employed for PA assessment. A suspension containing 1 × 108 cells/mL of Staphylococcus sp. in 0.1 mL of phosphate buffer solution was introduced to 0.1 mL of the blood sample on a Petri dish and incubated for 30 min, following thorough mixing in the well. Post-incubation, gently agitate the dish and apply 0.05 mL of this suspension onto a glass slide. After air-drying, fixate the smear with ethanol, perform cell counting, and prepare the phagocytosis for microscopic analysis (Enis Yonar et al., 2011).
The data is expressed as the mean ± SEM. One-way analysis of variance (ANOVA) was utilized to evaluate variations among the treatments. The significance of differences in the levels of oxidative and antioxidative stress parameters, as well as immunological parameters, was examined using the Mann–Whitney U test following Zar’s guidelines (1999). Significance was defined at p < 0.05. All statistical analyses were performed using Statgraphics Centurion XIX software (Statgraphics Technologies, The Plains, VA, USA).
Results and Discussion
In this investigation, the botanical screening of M. azedarachleaf ethanol extract (MAEE) was conducted to ascertain its chemical composition. The findings revealed the presence of various compounds such as alkaloids, flavonoids, tannins, saponins, polyphenols, steroids, and terpenoids in MAEE, while the absence of cardiac glycosides was noted (Table 2). To precisely quantify the concentrations of the primary constituents, an analysis was carried out to measure the levels of flavonoids, alkaloids, and tannins in MAEE. The results indicated that the content of flavonoids was 44.49 ± 2.16 mg QE/g, polyphenols amounted to 66.75 ± 2.14 mg GAE/g, and tannins were measured at 71.67 ± 2.07 mg CE/g (Table 3). These parameters provide crucial insights into the chemical composition of MAEE, establishing a foundation for a more comprehensive understanding of the biological and medicinal potential of the Bead tree.
| Phytochemicals | Present in MAEE | Phytochemicals | Present in MAEE |
|---|---|---|---|
| Alkaloids | + | Cardiac glycosides | – |
| Tannins | + | Steroids | + |
| Saponins | + | Terpenoids | + |
| Polyphenols | + | Flavonoids | + |
| Sample | Total flavonoids content (mg QE/g) | Total polyphenol content (mg GAE/g) | Total tannins content (mg CE/g) |
|---|---|---|---|
| MAEE | 44.49 ± 2.16 | 66.75 ± 2.14 | 71.67 ± 2.07 |
Plant extracts have garnered significant attention in medical research, particularly for their prowess in combating oxidative stress and bolstering immune response. These extracts commonly harbor an array of antioxidant compounds such as flavonoids, polyphenols, and other compounds. These constituents play a pivotal role in thwarting the damage inflicted by free radicals, mitigating oxidative stress, and stimulating the body’s innate production of antioxidant enzymes. Furthermore, plant extracts also exert positive effects on the immune system, fortifying its functionality, countering pathogenic agents, and enhancing immune responsiveness. This dual action of plant extracts, targeting both oxidative stress and immune modulation, positions them as promising candidates for preventive and adjunctive therapeutic interventions across various pathological conditions. Therefore, investigating and applying the M. azedarach leaf ethanol extract (MAEE) in the realm of oxidative stress mitigation and immune response enhancement in catfish present potential benefits for oxidative stress alleviation and immune response enhancement (Kowalczewski & Zembrzuska, 2023). The constituents present in M. azedarach leaf ethanol extract (MAEE), including alkaloids, flavonoids, tannins, saponins, polyphenols, steroids, and terpenoids, play pivotal roles in oxidative stress mitigation and immune response enhancement. Alkaloids exhibit antioxidative effects and contribute to the stabilization of biochemical reactions in the body. Flavonoids, by safeguarding cells against free radical-induced damage, reduce oxidative stress and stimulate the activity of antioxidant enzymes. Tannins, on the other hand, impede the absorption of harmful substances and demonstrate antioxidative properties. Saponins stimulate the immune system, potentially enhancing the body’s immune responsiveness. Polyphenols act as antioxidants, protecting cells from injury and reducing oxidative stress. Certain steroids influence immune function, participating in the regulation of the body’s immune response. Terpenoids exhibit antibacterial and antiviral properties, contributing to the antioxidative process (Forni et al., 2019). In particular, the quantified values of flavonoids (44.49 ± 2.16 mg QE/g), polyphenols (66.75 ± 2.14 mg GAE/g), and tannins (71.67 ± 2.07 mg CE/g) provide profound insights into the potential antioxidative and immune-enhancing effects of MAEE. The substantial presence of flavonoids in MAEE (44.49 ± 2.16 mg QE/g) enhances its antioxidative capacity, shielding cells from damage caused by oxidative stress. The significant quantity of polyphenols (66.75 ± 2.14 mg GAE/g) represents a robust group of antioxidative compounds, assisting in the prevention of free radical-induced harm and reducing oxidative stress. Tannins (71.67 ± 2.07 mg CE/g) play a crucial role in impeding the absorption of harmful substances, exhibiting antioxidative properties, and promoting overall health.
MDA and H2O2 as pivotal indicators of oxidative stress, providing valuable insights into cellular responses to oxidative challenges. Table 4 illustrates the dynamic changes in the levels of MDA and H2O2 in the gill, liver, kidney, and heart tissues of catfish subjected to oxidative stress induced by T. nigra. Upon exposure to T. nigra, a significant increase in MDA and H2O2 levels was observed in the gill, liver, kidney, and heart tissues of catfish (p < 0.05) (Untreated group), markedly differing from the Control group (p < 0.05). Subsequent treatment with MAEE resulted in a substantial reduction in MDA and H2O2 levels in the gill, liver, kidney, and heart tissues of catfish (p < 0.05). The most significant reduction was achieved with MAEE1000 (p < 0.05), approaching the efficacy observed with formalin treatment. This data underscores the potential of MAEE in mitigating oxidative stress-induced elevations in MDA and H2O2 levels, highlighting its promise as a therapeutic intervention.
In fish, the gills, being directly exposed to the aquatic environment, typically constitute the initial site impacted, primarily associated with respiratory functions. Meanwhile, major internal organs such as the liver, kidney, and heart emerge as primary targets for oxidative stress, actively engaging in antioxidant processes to uphold the body’s internal equilibrium (Kaur & Jindal, 2017). Upon attack by T. nigra, the gill, liver, kidney, and heart tissues of catfish undergo a significant increase in MDA and H2O2 levels (p < 0.05) compared to the control group. This signifies a clear indication of enhanced oxidative stress in catfish, leading to a substantial elevation of MDA and H2O2. This heightened oxidative stress is often a result of the harmful effects induced by T. nigra, potentially involving the generation of ROS or the promotion of lipid peroxidation, resulting in increased production of MDA and H2O2. Oxidative stress initiates with the generation of ROS such as hydroxyl radicals (•OH), O2•−, and H2O2 through cellular metabolism, exposure to environmental stressors, or stimulation by bacteria such as T. nigra. ROS typically impacts lipids in cell membranes, inducing lipid peroxidation and generating MDA as a byproduct. O2•−, in particular, can further contribute to the production of H2O2 through chemical reactions. Additionally, the body employs natural mechanisms to reduce H2O2 levels, including enzymes like CAT and peroxidases. However, rapid increases in ROS production can overwhelm this system, leading to an elevation of H2O2 (Chaudhary et al., 2023). The reduction in MDA and H2O2 levels after treatment with the M. azedarach leaf ethanol extract (MAEE), especially with MAEE1000, highlights the potential of this extract in mitigating the impact of oxidative stress, protecting the aforementioned tissues from the detrimental effects of T. nigra. The polyphenols and flavonoids within MAEE, renowned for their antioxidant properties, play crucial roles in reducing the production of MDA and H2O2. Both polyphenols and flavonoids directly act on ROS like O2•− and •OH, inhibiting their free radical chain reactions and reducing ROS production. The chemical structure of flavonoids enables them to act as potent antioxidants, shielding cells from damage caused by oxidative stress. Polyphenols can inhibit lipid peroxidation, the primary source of increased MDA, preserving lipids in cell membranes from severe damage and preventing excessive MDA production. Simultaneously, flavonoids also stabilize cell membranes and impede lipid peroxidation, aiding in the reduction of MDA production (Rajashekar, 2023).
GSH and AsA are indispensable components in the body’s antioxidant defense system, acting synergistically to maintain cell integrity, support immune function, and counteract the adverse effects of oxidative stress. Figs. 1 and 2 depict the dynamic changes in GSH and AsA levels in various organs of catfish subjected to oxidative stress induced by T. nigra. The concentrations of GSH and AsA significantly decreased (p < 0.05) in the spleen, liver, kidney, and heart of catfish following T. nigra invasion (Untreated group). This outcome stands in stark contrast to the GSH and AsA levels observed in the control group (p < 0.05). Nevertheless, the levels of GSH and AsA gradually increased in the groups treated with MAEE (p < 0.05). This suggests a potential ameliorative effect of MAEE on the oxidative stress-induced reduction in GSH and AsA levels, highlighting its promising role in bolstering the antioxidant defense mechanisms in catfish tissues.

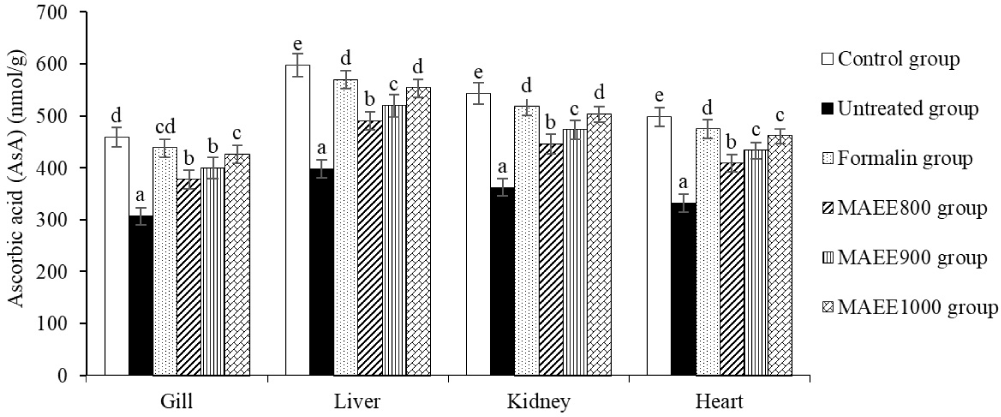
GSH acts as a potent free radical scavenger, neutralizing ROS to protect cells from oxidative damage. Through an “electron hopping” process, GSH forms non-toxic compounds with free radicals and other oxidative molecules. GSH also supports the immune system and DNA repair. AsA functions as a free radical scavenger, neutralizing ROS to prevent oxidative stress-induced cell damage. AsA captures and eliminates free radicals, shielding cells from damage and enhancing cellular repair capabilities (Niu et al., 2023). In the gill, liver, kidney, and heart of fish, GSH and AsA play a crucial role in maintaining the integrity of these organs in an environment prone to oxidative stress. GSH participates in scavenging and detoxifying free radicals and other oxidative molecules, eliminating toxins and oxidative by-products from the body while preserving cell membranes and other cellular components. AsA, with its ability to scavenge free radicals, prevents their damaging effects on cells, protecting cells from harm and maintaining cellular structure (Kaur & Jindal, 2017). Upon invading the catfish body, T. nigra can increase the production of ROS such as superoxide and •OH, inducing oxidative stress by negatively impacting molecules and cell structures. T. nigra enhances water and nutrient permeability across cell membranes, allowing harmful substances and free radicals to infiltrate cells and induce oxidative stress. T. nigra stimulates enzymatic systems that produce ROS, such as NADPH oxidase, intensifying ROS formation and escalating oxidative stress. T. nigra invasion results in reduced production and regeneration of GSH and AsA in fish bodies, attributed to an imbalance between the consumption of antioxidants and their regenerative capacity (Abou-Okada et al., 2021). The M. azedarach leaf ethanol extract (MAEE) acts as a supportive agent to maintain and enhance the antioxidant system in fish bodies, leading to increased levels of GSH and AsA and reducing the risk of oxidative stress-induced damage. Polyphenols safeguard cells from the adverse effects of free radicals and oxidative stress, while flavonoids prevent damage from free radicals and other oxidative molecules. The active compounds in MAEE interact with signaling pathways in the body, potentially modulating cellular processes and genes related to GSH and AsA regeneration (Rajashekar, 2023).
CAT and GPx function to shield cells from the detrimental effects of free radicals and oxidative substances, aiding in the maintenance of cellular stability and integrity in oxidative environments. Upon invasion by T. nigra, vital organs such as the spleen, liver, kidney, and heart of catfish undergo severe damage, manifested by a substantial decrease in CAT and GPx levels in the untreated group (p < 0.05) (Table 5). This outcome significantly differs from the control group (p < 0.05). However, following treatment with MAEE, CAT and GPx levels experienced a notable increase (p < 0.05). Particularly at a concentration of 1,000 ppm of MAEE, the treatment efficacy approached equivalence with the effects of Formalin (p > 0.05; Table 5). This result indicates a pronounced restorative effect of MAEE on the depleted levels of CAT and GPx organs of catfish post-T. nigra invasion. The substantial elevation of CAT and GPx concentrations, especially at the 1,000 ppm MAEE dosage, suggests a potential amelioration of oxidative stress-induced damage. The observed effectiveness, comparable to Formalin treatment, underscores the promising role of MAEE as a therapeutic intervention in mitigating the oxidative stress impact caused by T. nigra invasion.
CAT and GPx are key antioxidant enzymes that play a crucial role in neutralizing oxidative substances and protecting cells from oxidative stress-induced damage. CAT reduces H2O2 to water and O2, preventing the accumulation of H2O2 and minimizing the risk of oxidative stress injuries. GPx utilizes GSH to reduce H2O2 and organic hydroperoxides to non-toxic products, maintaining cellular safety against oxidative stress (Ighodaro & Akinloye, 2018). Oxidative stress induced by T. nigra in catfish has escalated the demand for CAT and GPx to eliminate ROS and oxidative substances. However, due to heightened ROS production and oxidative pressure, the regeneration process is not rapid enough, resulting in a decrease in CAT and GPx levels. In the fish’s gill, oxidative stress has augmented water and nutrient permeability through the cell membrane, providing favorable conditions for the infiltration of free radicals and oxidative substances. The elevated ROS in the gill environment, coupled with reduced elimination by CAT and GPx, diminishes the activity and levels of these enzymes, causing damage to cellular structures. In the liver, T. nigra stimulates enzymatic systems such as NADPH oxidase, escalating ROS production and disrupting the balance between CAT and GPx consumption and regeneration. This leads to a reduced quantity of antioxidant enzymes in the liver. Oxidative stress further inflicts damage on the liver’s structure, compromising its antioxidant capabilities. The kidneys experience increased ROS production and heightened water permeability through the cell membrane, inducing oxidative stress. The detrimental effects of free radicals impair renal cell integrity. Oxidative stress elevates the need for ROS removal and diminishes the regenerative capacity, resulting in reduced levels of CAT and GPx in the kidneys. In the heart, the rapid accumulation of ROS is attributed to increased production and decreased removal by CAT and GPx. Simultaneously, an imbalance between CAT and GPx consumption and regeneration leads to diminished antioxidant enzyme levels in the heart (Kaur & Jindal, 2017). In this study, the M. azedarach leaf ethanol extract (MAEE), enriched with bioactive components such as polyphenols, flavonoids, alkaloids, tannins, etc., demonstrated antioxidative effects and augmentation of CAT and GPx levels. These active compounds interacted with cellular signaling pathways, modulating cellular processes and genes related to the regeneration of CAT and GPx (Rajashekar, 2023).
SOD and GR play pivotal roles in maintaining the structural integrity of cells, preventing damage to DNA, and safeguarding other cellular structures from the detrimental effects of free radicals. Figs. 3 and 4 illustrate the dynamic changes in SOD and GR levels in the gill, liver, kidney, and heart of catfish subjected to oxidative stress induced by T. nigra. Following treatment with the M. azedarach leaf ethanol extract (MAEE), the concentrations of SOD and GR exhibited a substantial increase (p < 0.05), a trend opposed to that observed in the untreated group (p < 0.05). Notably, the elevation of SOD and GR in the MAEE-treated group, specifically at a concentration of 1,000 ppm, approached statistical equivalence with both the Formalin group (p > 0.05) and the control group (p > 0.05). The data strongly imply that MAEE holds promise as a therapeutic agent in mitigating oxidative stress-induced damage in catfish, potentially offering a natural and effective solution to enhance cellular resilience against oxidative challenges.
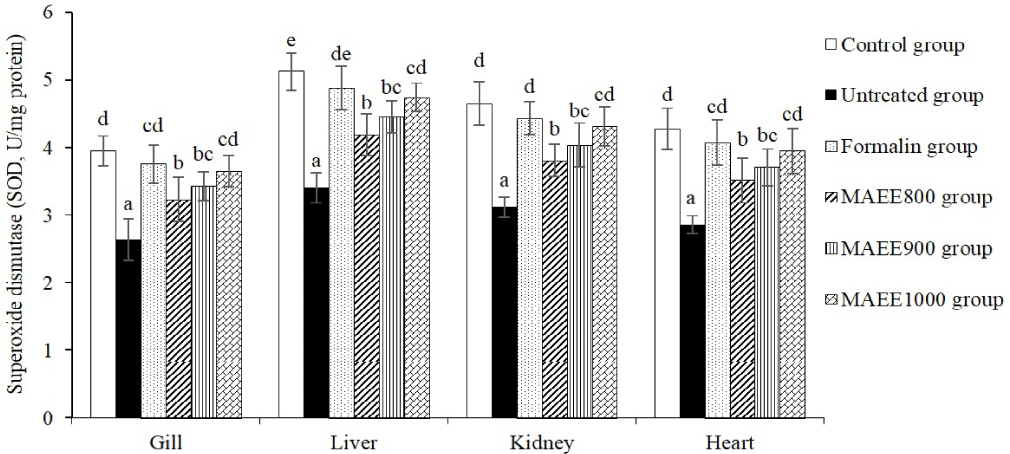
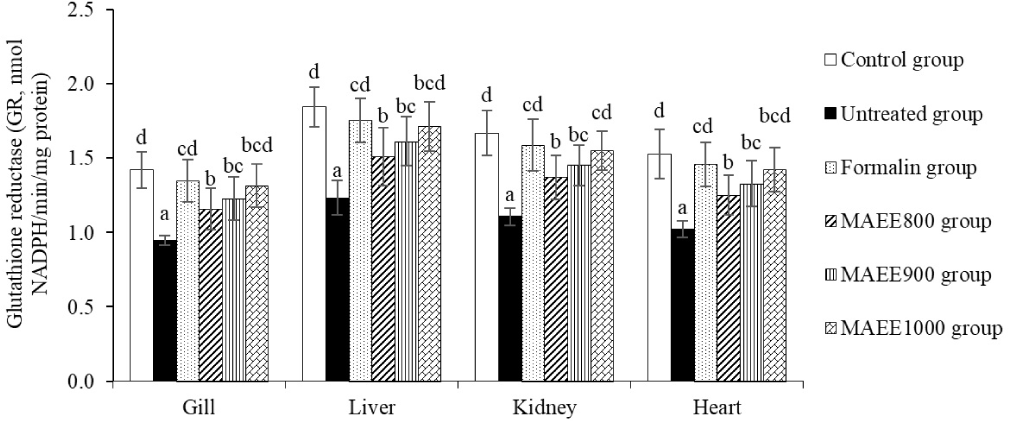
SOD and GR are vital enzymes in the body’s antioxidant defense. SOD transforms (O2−) into H2O2 and O2, averting the creation of highly toxic free radicals. GR contributes to regenerating glutathione (GSH) from its oxidized form (GSSG), with GSH serving as a crucial antioxidant that eliminates free radicals and oxidizing agents from the body (Ighodaro & Akinloye, 2018). The fluctuation of SOD and GR in oxidative stress conditions is an expression of the body’s specific response to the negative impact of free radicals and oxidative agents. These changes not only represent a natural reaction of the body but also serve as a significant indicator of the adaptability of the antioxidant defense system. The reduction of SOD and GR in oxidative stress in catfish caused by T. nigra reflects the body’s limited and diminished antioxidant capacity when facing the adverse effects of free radicals and oxidative agents. This reduction indicates the level of oxidative pressure that the body has to contend with and the ability to regenerate antioxidant substances. The decrease in SOD and GR in the oxidative stress environment induced by T. nigra suggests that the body’s antioxidant system is not efficiently responding to high oxidative pressure. The body may not produce sufficient SOD and GR to eliminate free radicals and oxidizing agents, or the regeneration process may be slow, leading to reduced resilience of the antioxidant system. This implies that the body is undergoing a state of “stress” where oxidative impact is excessive, making it challenging for the body to maintain the necessary antioxidant levels to protect cells from damage. The regeneration or production of SOD and GR may be affected, diminishing the body’s ability to withstand oxidative stress (Zainab et al., 2021). This study demonstrates that the M. azedarach leaf ethanol extract (MAEE) has a pronounced and beneficial effect on the antioxidant defense mechanism in smooth-skinned catfish exposed to oxidative stress induced by T. nigra. The substantial increase in SOD and GR concentrations after MAEE treatment indicates an improvement in the fish’s ability to resist and neutralize free radicals. The significant elevation of SOD and GR, especially at a concentration of 1,000 ppm, closely aligns with the levels observed in the Formalin group and the Control group, suggesting a potent antioxidant effect comparable to conventional treatment and the natural physiological state, respectively. This indicates that MAEE, with bioactive components such as polyphenols, flavonoids, alkaloids, tannins, etc., reinforces the effectiveness of the antioxidant defense system.
TAC quantifies the overall antioxidative capability of a system to assess the collective antioxidant potential of an organism. The percentage of TAC in the gill, liver, kidney, and heart tissues of catfish is depicted in Fig. 5. Observable variations in TAC are evident in the untreated group, where a significant reduction in % TAC is observed in fish subjected to oxidative stress induced by T. nigra (p < 0.05). This outcome is completely reversed after treatment with MAEE, as % TAC shows a substantial increase (p < 0.05), particularly reaching the highest levels in the group treated with MAEE at 1,000 ppm (p < 0.05). The efficacy of MAEE at 1,000 ppm is nearly equivalent to Formalin (p > 0.05). This suggests a pronounced restorative effect of MAEE on the TAC in catfish tissues, particularly in mitigating the oxidative stress induced by T. nigra.
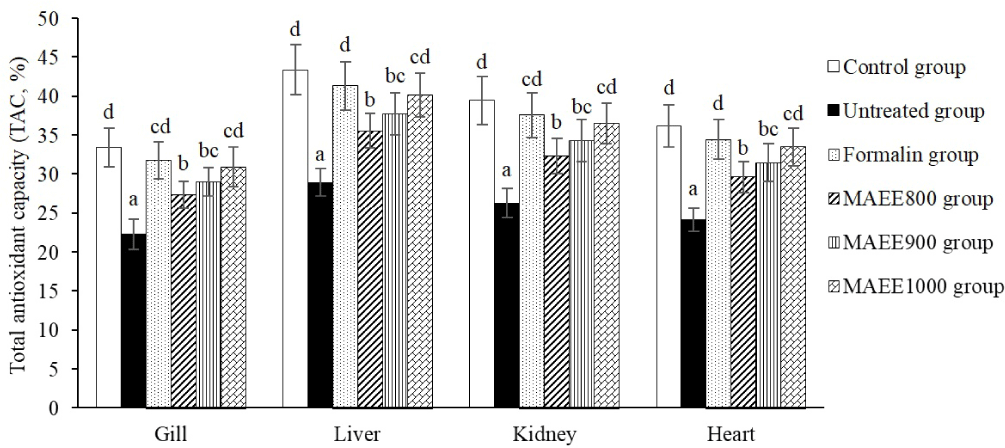
TAC indicates an organism’s or sample’s ability to resist free radicals and oxidative stress. Maintaining TAC is crucial for balancing oxidative levels and protecting cells from damage. A decreased TAC during oxidative stress signals a reduced overall antioxidant capacity. In the untreated group, the significant TAC decrease indicates limited antioxidant capability against free radicals and oxidative stress. This reduction suggests insufficient antioxidant production to counter T. nigra-induced oxidative stress. The TAC decline may stem from an imbalance between antioxidant production and consumption. In oxidative stress, the body depletes antioxidants to combat numerous stressors, revealing a stressed state where oxidative impact surpasses the body’s antioxidant levels needed for cell protection (Gupta et al., 2021). Conversely, after treatment with M. azedarach leaf ethanol extract (MAEE), the sudden increase in TAC, especially in the group treated with MAEE at 1,000 ppm, indicates a robust recovery and significant improvement in antioxidant capacity. This suggests that MAEE has the potential to stimulate and enhance the overall antioxidant system of the body, particularly when facing oxidative stress from T. nigra. Moreover, the comparable effectiveness between MAEE at 1,000 ppm and Formalin provides insights into the potential of MAEE as an intervention that can rival conventional treatment methods.
In general, the M. azedarach leaf ethanol extract (MAEE) exhibits positive effects on the levels of MDA, H2O2, glutathione (GSH), AsA, CAT, GPx, SOD, GR, and TAC in catfish. MAEE may contain various natural antioxidants such as polyphenols, flavonoids, and other compounds, which can help protect cells from oxidative stress and stimulate the production of CAT, SOD, GPx, GR, GSH, AsA, and TAC, while limiting MDA and H2O2. The impact of MAEE on the activities of antioxidant enzymes like CAT, SOD, GPx, etc., is attributed to its direct or indirect influence on enzymes and participants involved in the regeneration of antioxidant compounds. This ensures the maintenance of high levels of these antioxidants. MAEE can contribute to reducing oxidative stress by preventing or decreasing the levels of free radicals and other oxidants in the body, thereby alleviating pressure on the antioxidant system and preserving GSH and AsA. Additionally, MAEE may have the ability to interact with signaling pathways in the fish’s body, potentially influencing the processes involved in the regeneration of CAT, SOD, GPx, GR, GSH, AsA, and TAC. Thus, MAEE can serve as a supportive agent in maintaining and enhancing the antioxidant system in the fish’s body, leading to increased levels of enzymes and antioxidant compounds while reducing the risk of damage from oxidative stress (Ahmed et al., 2008).
WBC, ROS, and PA play pivotal roles in the immune system, safeguarding the body against pathogens and maintaining overall health. Table 6 illustrates the survey results on the fluctuations of WBC, ROS, and PA in the blood of catfish. Following an untreated invasion by T. nigra, the untreated group exhibited a significant increase in WBC levels (p < 0.05), accompanied by a substantial decrease in ROS and PA levels (p < 0.05). These findings markedly differed from those of the control group (p < 0.05). Notably, these parameters improved significantly after treatment with MAEE, as WBC levels decreased (p < 0.05), while ROS and PA levels increased significantly (p < 0.05). This observation suggests that the immune response of catfish to T. nigra invasion is dynamic and influenced by the administration of MAEE. The significant increase in WBC levels post-treatment implies a potential enhancement in the body’s ability to combat the pathogen. Concurrently, the substantial rise in ROS and PA levels signifies an augmented PA, indicative of a reinforced defense mechanism against the invasive threat.
The immune response is a sophisticated system designed to detect, eliminate, and eradicate pathogens like bacteria, viruses, and abnormal cells. WBC play a crucial role in this response. Monocytes phagocytose dead cells, triggering other immune cells via cytokine production. Lymphocytes generate antibodies and eliminate infected cells, while neutrophils use phagocytosis to eradicate pathogens. Neutrophils, actively engaging pathogens, produce a green color using ROS. PA gauges immune cells’ ability to engulf and eliminate pathogens, essentially reflecting the process of phagocytosis (Chaves-Pozo & Cuesta, 2023). In the current study, the significant increase in WBC in the untreated group after exposure to T. nigra suggests an elevated immune response. Elevated WBC levels typically indicate the body’s attempt to counteract infection or microbial invasion. The substantial decrease in ROS in the untreated group indicates a compromised phagocytic capacity, especially of neutrophils. Inefficient phagocytosis by neutrophils diminishes their ability to eliminate pathogens. The pronounced decrease in PA in the untreated group corresponds to a reduced ability to engulf cells. This may result from decreased activity of neutrophils and monocytes, two cell types closely associated with the phagocytosis process. The noteworthy differences between the untreated and control groups, with increased WBC and decreased ROS and PA in the untreated group (p < 0.05), underscore the impact of T. nigra on the fish’s immune system. Following treatment with the M. azedarach leaf ethanol extract (MAEE), improvements were observed, including decreased WBC and increased ROS and PA (p < 0.05), particularly in phagocytic ability. This indicates the potential positive effects of MAEE in treating and reinforcing the fish’s immune system during an invasion. These improvements could result from MAEE’s ability to reduce infection rates and stimulate the immune response (Abidin et al., 2022).
TP and TI furnish in-depth insights into the overall protein levels and the immune system’s capacity to respond to and counteract various pathogens. The results presented in Fig. 6 indicate a substantial reduction in TP and TI levels in the untreated groups (p < 0.05), significantly different from the control groups (p < 0.05). Notably, TP and TI levels exhibited a significant increase in groups treated with MAEE (p < 0.05). Of particular interest, the most favorable outcomes were observed in the MAEE 1,000 ppm group, comparable to the efficacy achieved with the Formalin treatment (p < 0.05). This observation suggests a pronounced positive impact of MAEE treatment on TP and TI levels, indicative of its potential to enhance the overall protein content and immune response within the studied groups. The significant improvement, particularly in the MAEE 1,000 ppm group comparable to Formalin treatment, underscores the promising therapeutic efficacy of MAEE in maintaining robust immune function and protein homeostasis. These findings support the notion that MAEE may play a crucial role in fortifying the immune system.
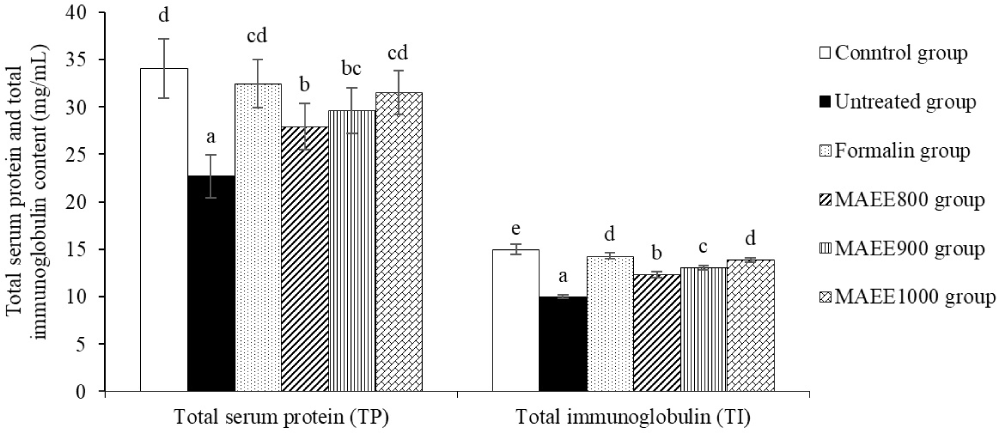
TP represents all serum proteins, including albumin and globulin. TP levels help assess nutritional status, and liver and kidney function and detect protein-related abnormalities. TI, identifies and neutralizes foreign substances, aiding in diagnosing immune disorders or deficiencies (Chaves-Pozo & Cuesta, 2023). In this study, the substantial decrease in TP and TI levels in the untreated groups (p < 0.05), significantly different from the control group (p < 0.05), indicates an imbalance or deterioration in the immune system and overall protein status in these groups. This reduction may be indicative of stress or the influence of the study’s factor, specifically the T. nigra infection. The decline suggests a potential weakening of the immune system’s resistance against pathogens and external agents. Additionally, the decrease in TP may reflect the functional state of the liver or conditions such as dehydration or protein-related disorders (Correa et al., 2022). Conversely, a noteworthy increase in TP and TI was observed in groups treated with MAEE (p < 0.05). Particularly, the most positive outcomes were evident in the MAEE 1,000 ppm group, equivalent to the efficacy of Formalin therapy (p < 0.05). This enhancement may indicate the potential of MAEE in reinforcing immune function and maintaining the stability of TP. It reflects a positive response of the immune system and an improvement in the overall protein regime in groups treated with MAEE. This increase may result from the positive effects of MAEE in stimulating TP production and enhancing immune responsiveness. Specifically, the rise in TP is often associated with improved nutrition and liver function. Meanwhile, the increase in TI may signify MAEE’s ability to stimulate immunoglobulin production, enhancing immune capabilities against pathogens (Abidin et al., 2022).
The freshwater catfish, identifiable by its elongated body, smooth and glossy translucent skin with brown-black hues and white spots, exhibits a lighter hue on its abdomen (Fig. 7A). However, parasitic infestations induce dermal injuries, leading to hyperplasia, thickened skin, desquamation, and ulcer formation. The affected area gradually undergoes melanin alteration. Lesions predominantly manifest along the dorsal region, occasionally extending to the thorax and abdomen (Fig. 7B). Subsequent treatment with MAEE and formalin restores the fish’s morphology to a normal state (Fig. 7C and 7D).

Conclusion
Using ethanol extract from Bead tree leaves (M. azedarach L.) to combat oxidative stress and immunosuppression induced by T. nigra in catfish (C. fuscus) yielded significant results. MDA and H2O2 levels in catfish tissues (gill, liver, kidney, and heart) significantly decreased after treatment with MAEE. Organs severely damaged by T. nigra invasion exhibited a substantial reduction in antioxidant enzymes, including glutathione (GSH), AsA, CAT, GPx, SOD, GR, and TAC. However, after MAEE treatment, these parameter concentrations increased significantly. Particularly, for all parameters, the efficacy of MAEE at a concentration of 1,000 ppm was nearly equivalent to formalin. A significant reduction in WBC and a substantial increase in ROS, PA, TP, and TI in catfish blood were also observed after treatment. The use of MAEE demonstrated the catfish’s recovery ability. Ethanol extract from M. azedarach leaves showed the potential to cope with oxidative stress and immune suppression caused by T. nigra in catfish, with effectiveness comparable to formalin. This study opens prospects for practical applications in managing and protecting catfish health.
