Introduction
Eel aquaculture is a pivotal sector in the global aquaculture industry, with a specific emphasis on species such as the Japanese eel (Anguilla japonica L.). The demand for eel products is escalating, particularly in Korea, China, Taiwan, and Japan. However, the availability of elvers (also known as glass eels or juvenile eels), which relies on natural recruitment, has been in decline since the 1960s (Dekker, 2003; Shiraishi & Kaifu, 2024). To overcome these challenges, extensive research on artificial seed production has been continuously conducted since the 1970s (Okamura et al., 2020; Tanaka, 2015). While significant improvements have been technically successful, commercial scalability remains constrained, typically yielding from thousands to ten thousands of elvers annually (Okamura et al., 2020). Given these constraints, there is an increased focus on optimizing technologies and enhancing productivity within eel aquaculture to maintain its sustainability.
Recent trends in aquaculture have seen a rise in the use of functional additives in feed formulations aimed at improving growth performance, health, and stress resilience of cultured species (Hossain et al., 2024). These additives encompass a wide range of bioactive compounds, including nucleotides (Hossain et al., 2020), organic acids (Ng & Koh, 2017), prebiotics (Guerreiro et al., 2018), probiotics (El-Saadony et al., 2021), immunostimulants (Vijayaram et al., 2022), and enzymatic hydrolysates, which provide specific health benefits beyond basic nutrition (Gómez & Balcázar, 2008).
Liver hydrolysates, in particular, are valued for their rich concentration of bioactive peptides, essential amino acids, vitamins, and minerals, which enhance the attractiveness and palatability of diets and support various physiological functions including immune modulation, antioxidant activity, and growth promotion (Alves et al., 2019, 2020). Chicken liver hydrolysate (CLH), produced from the enzymatic hydrolysis of human food-grade chicken livers, consists mainly of oligopeptides and free amino acids (FAs) with minimal carbohydrates and fats (Neklyudov et al., 2000; Wu et al., 2003).
Despite the widespread documentation of CLH’s benefits in aquaculture (Gu et al., 2024; Hao et al., 2020; Kureshy et al., 2000), its application in eel culture remains underexplored. This gap highlights the need for detailed research on the potential advantages and appropriate inclusion levels of CLH in eel diets. This study aims to assess the impact of Harim-CLHTM supplementation, a commercial CLH, in diets of A. japonica eels on their growth performance, biochemical parameters, attractancy/palatability, and transcriptomic profiles through next-generation sequencing (NGS) analysis.
Materials and Methods
Juvenile A. japonica eels, approximately 20 g each, were sourced from the Kofec Central Research Institute. Each 60 L tank housed 25 fish, with three replicates per treatment. Diets were administered twice daily, at 07:30 and 14:30, targeting an intake of 3% to 7% of body weight. Uneaten feed was collected, dried, weighed, and deducted from the total feed quantity 20–30 minutes after feeding. Aquaculture conditions were maintained at 29 ± 1°C, with dissolved oxygen levels at 6.5 ± 0.5 mg/L and pH at 5.9 ± 0.5. Water changes of 90% were conducted daily, four hours post-feeding, and tanks were cleaned weekly.
After eight weeks of feeding trials, each fish was individually counted and weighed. Growth performance metrics were calculated as follows:
Final Weight (FW): Weight of fish at the end of the trial.
Weight Gain (WG): Final Weight – Initial Weight.
Weight Gain Rate (WGR): (Final Weight – Initial Weight) × 100/Initial Weight.
Specific Growth Rate (SGR): [ln(Final Weight) – ln(Initial Weight)]/Days × 100.
Total Dry Feed Intake (DFI): Total feed consumed on a dry matter basis.
Feed Efficiency (FE): (Wet Weight Gain/Dry Feed Intake) × 100.
Survival Rate: (Number of Fish at End of Experiment/Number of Fish at Start of Experiment) × 100.
The attractancy test used a specific tank structure (Fig. 1). A total of 770 A. japonica eels, averaging 1.02 g, were acclimatized to the test feed by alternating between CTL and CLH-1 diets twice daily for three weeks. Adapted fish were transferred to the test tank for the attractancy test, with a three-day adaptation period to acclimate to the new conditions. Water flow was maintained using a 5 W submersible pump, cycling from the water tank to the experimental tank and back, with nets installed to prevent fish from escaping. For testing, 20 g of CTL and CLH-1 diet mixed with 140% water was placed in the food nets of each test tank. After opening the pathway between the tanks for 10 minutes, the number of eels that had moved was counted upon closing the pathway. This procedure was repeated daily for three consecutive days to minimize stress impacts on the fish.
A total of 160 Japanese eels, averaging 3.75 ± 0.02 g, were utilized. These eels were evenly distributed, with 80 individuals per tank. The system employed a water recirculation filtration method. Initially, there was a 7-day acclimatization period to the water temperature, aquarium environment, and diet. During this period, commercial feed (Kofecs, Korea) constituting approximately 6.3% of the fish body weight was administered twice daily at 08:00 and 17:00. Following acclimatization, feeding was adjusted to 5 g per session, twice daily. Subsequently, during the evaluation period, the control diet (CTL) and the chicken liver hydrolysate diet (CLH-1) were provided once daily for seven days, each constituting an appropriate percentage of the fish body weight based on previous feeding practices. Twenty minutes post-feeding, any uneaten feed was collected, dried, weighed, and its weight deducted from the total diet supply to accurately measure feed intake and assess palatability. This method ensured precision in quantifying the actual feed consumption and provided insights into the relative palatability of the two diets under examination.
The proximate composition of the experimental diets was determined according to the Association of Official Analytical Chemists (AOAC, 2000). Moisture content was assessed by drying samples at 105°C until constant weight was achieved, typically around 6 hours. Crude protein was analyzed using the Kjeldahl method, and ash content was determined by incineration in a furnace at 550°C for 6 hours. Crude lipids were extracted using a modified acid-base method described by Duongbia et al. (2018). Gross energy content was calculated using conversion coefficients of 23.6 kJ/g for protein, 38.9 kJ/g for fat, and 16.7 kJ/g for carbohydrates (Miglavs & Jobling, 1989). Amino acid composition was analyzed by the ninhydrin post-column derivatization method using ion exchange chromatography, with modifications from Peace & Gilani (2005). For the analysis of 16 amino acids, samples (0.2 g) were hydrolyzed with 6N HCl at 110°C under an aerobic condition for 24 hours. The hydrolyzed samples were concentrated under reduced pressure, diluted in 50 ml of 0.2M sodium citrate buffer, filtered, and analyzed. Methionine and cysteine were analyzed using the performic acid oxidation method, and tryptophan via alkali hydrolysis. Lipid composition was determined following the method of Folch et al. (1957) using gas chromatography (HP 6890, Hewlett Packard , Palo Alto, CA, USA) with an HP-88 column. The samples underwent transesterification using 14% BF3-MeOH and were analyzed under specific temperature conditions: initial 140°C for 5 minutes, then ramping at 4°C/min to 240°C and holding for 20 minutes. Injector and detector temperatures were set at 260°C and 270°C, respectively.
Lysozyme activity was assessed using a modified turbidimetric method (Koskela et al., 2004). The assay was conducted in a 96-well plate, where each reaction mixture (1 mL) included 50 μL of serum, 0.05 M sodium phosphate buffer (pH 6.2, Sigma-Aldrich, Ronkonkoma, NY, USA), and Micrococcus lysodeikticus (0.2 mg/mL). Turbidity was measured at 530 nm with a microplate reader (Molecular Device, San Jose, CA, USA) at 20°C after 0.5 and 5 minutes. Lysozyme activity was expressed in units per mL, defined as a decrease in absorbance rate of 0.001 per minute. Phagocytic activity was determined via a modified spectrophotometric method (Seeley et al., 1990). Serum and Nitro Blue Tetrazolium (NBT) solution (Sigma-Aldrich) were mixed in a 1:1 ratio (50 μL each) and incubated at 25°C for 30 minutes. After incubation, the reaction mixture was transferred to a glass tube, and 1 mL of dimethylformamide (Sigma-Aldrich) was added to stop the reaction and dissolve the formazan product. The supernatant was collected post-centrifugation at 2,000 g for 5 minutes, and the reduction of NBT was measured at 540 nm using a microplate reader, with dimethylformamide serving as the blank control.
Following the feeding trials, blood samples were collected from the caudal vein of 10 anesthetized fish per group (CTL and CLH-1) using heparinized syringes. Samples were centrifuged at 500×g for 15 minutes at 4°C. The serum was then stored at –20°C for further biochemical analyses. Serum parameters analyzed included total protein (TP), total cholesterol (TCHO), triglyderides (TG), glucose (GLU), glutamic oxaloacetate transaminase (GOT), and alkaline phosphatase (ALP), using commercial enzymatic kits and a blood analyzer (NX500i, FUJI FILM, Tokyo, Japan). The condition index (CI) was estimated using K = W/L3 × 100, where W is the weight of the fish (g) and L is the length of the fish (cm). Hepatosomatic index (HIS) was estimated using LW × 100/BW, where LW is the fresh weight of liver (g) and BW is the weight of fresh body weight (g).
Intestine, liver and distal intestine samples were collected and pooled from 5 eels of two test groups. In homogenized tissues, total RNA was extracted by using TRIzol Reagnet (Ambion) according to the manufacturer’s instructions. RNA was determined Agilent Bioanalyzer (Agilent Scientific, Santa Clara, CA, USA), and the concentration was measured using a spectrophotometer. cDNA library of RNA was determined using QIAGEN’s QIAseq FX single cell RNA library kit. Library concentration was measured by LightCycle qPCR and library size was checked using Agilent TapeStation HS D5000 Screen Tape (Agilent Scientific). Sequencing was performed using Illumina HiseqX (Illumina, San Diego, CA, USA) using HiSeq × HD Reagent Kit v2.5. Low quality reads (the percentage of the low quality bases of quality value ≤ 30) were also removed. The clean reads were aligned to reference sequence by Bowtie2 aligner.
The differentially expressed genes (DEGs) between spot and non-spot tissues were identified and filtered with the R package DEGseq based on Q-value < 0.05, normalized|log2 (FC)| > 1. The heatmap displays of the Trimmed read count normalized against the Fragment Per Kilobase of transcripts per Million (FPKM) mapped reads were performed using the R package heatmap.2 function. The DEGs were imported into Blast2GO software v5.2.5 and in-house R scripts for gene ontology (GO) term analysis.
Results
Compared to the fishmeal used in this experiment, Harim-CLHTM has lower crude protein (fishmeal, 71.54 ± 0.33%; Harim- CLHTM, 15.99 ± 0.27%), crude lipids (fishmeal, 8.86 ± 0.08; Harim-CLHTM, 0.94 ± 0.01%), and carbohydrates (fishmeal, 0.4 ± 0.01; Harim-CLHTM, 73.90 ± 3.24%) and ash (fishmeal, 13.00 ± 0.08%; Harim-CLHTM, 1.50 ± 0.11%) (Supplementary Table S1). The CTL and the CLH-1 where 1% of the fishmeal was replaced with Harim-CLHTM, exhibited no significant differences in crude protein (CTL, 47.08 ± 0.33%; CLH-1, 45.91 ± 0.27%), crude lipid (CTL, 6.61 ± 0.07%; CLH-1, 6.77 ± 1.66%), carbohydrates (CTL, 24.40 ± 0.01%; CLH-1, 24.42 ± 0.08%) and ash (CTL, 16.17 ± 0.25%; CLH-1, 17.54 ± 1.71%) (Table 1). Amino acid analysis revealed no significant differences in the structural amino acid composition between eels fed CTL and those fed Harim-CLHTM. However, slight variations were observed in specific amino acids such as glutamic acid, glycine, cysteine, methionine, leucine, phenylalanine, and histidine (Supplementary Table S2). Notably, there were significant differences in free amino acids between fishmeal and Harim-CLHTM. For instance, CTL had higher levels of taurine (14 g/kg dw), histidine (25 g/kg dw), and GABA (33 mg/kg dw) compared to Harim-CLHTM, which contained none of these compounds. However, Harim-CLHTM exhibited substantially higher levels of almost all structural amino acids, such as aspartic acid (Harim-CLHTM: 5.79 g/kg dw vs. CTL: 0.39 g/kg dw) (Table 2). Fatty acid analysis showed that compared to fishmeal, Harim-CLHTM had higher contents of stearic acid, oleic acid, and linoleic acid but lower levels of polyunsaturated fatty acids such as EPA and DHA (Supplementary Table S3).
Vitamin and mineral are composed of following composition. Vitamin (VA, 20,000 IU; VD3, 40 mg; VE, 40 mg; VK, 50 mg; VB1, 25 mg; VB2, 40 mg; VB6, 20mg; VB12, 0.1 mg; Niacin, 125 mg; Calpan, 65 mg; Folic acid, 7.5 mg; Biotin, 1.5 mg; Inositol, 200 mg; VC, 600mg; antioxidant, 30 mg; some excipient) and mineral (Fe, 150 mg; Zn, 180 mg; Mn, 150 mg; Cu, 18 mg, Co, 0.9 mg; I(Ca), 1.8 mg; Mg, 240 mg; some excipient).
After 8 weeks of feeding the CTL and CLH-1 diets, significant differences in growth performance metrics were observed. The WGR for eels fed the CLH-1 diet was significantly higher at 306.7 ± 12.4%, compared to 267.5 ± 17.5% for those fed the CTL diet. Similarly, the DFI was significantly greater for the CLH-1 group at 204.1 ± 9.1%, vs. 175.5 ± 4.8% for the CTL group. FE was slightly higher in eels fed the CLH-1 diet (62.3 ± 6.2%) compared to those fed the CTL diet (53.9 ± 7.4%); however, this difference was not statistically significant. Additionally, CI and HIS were assessed for 10 randomly selected eels from each group after the feeding trial. BMI showed no significant difference between the groups, with eels fed CLH-1 averaging 8.07 ± 1.7 compared to 7.45 ± 1.7 for the CTL group. HIS also did not reveal significant differences, with a slightly lower average in the CLH-1 group (1.08 ± 0.1) compared to the CTL group (1.14 ± 0.2) (Fig. 2). These findings suggest that the CLH-1 diet enhances growth performance metrics such as WGR and DFI without significant impacts on body indices like BMI and HIS.
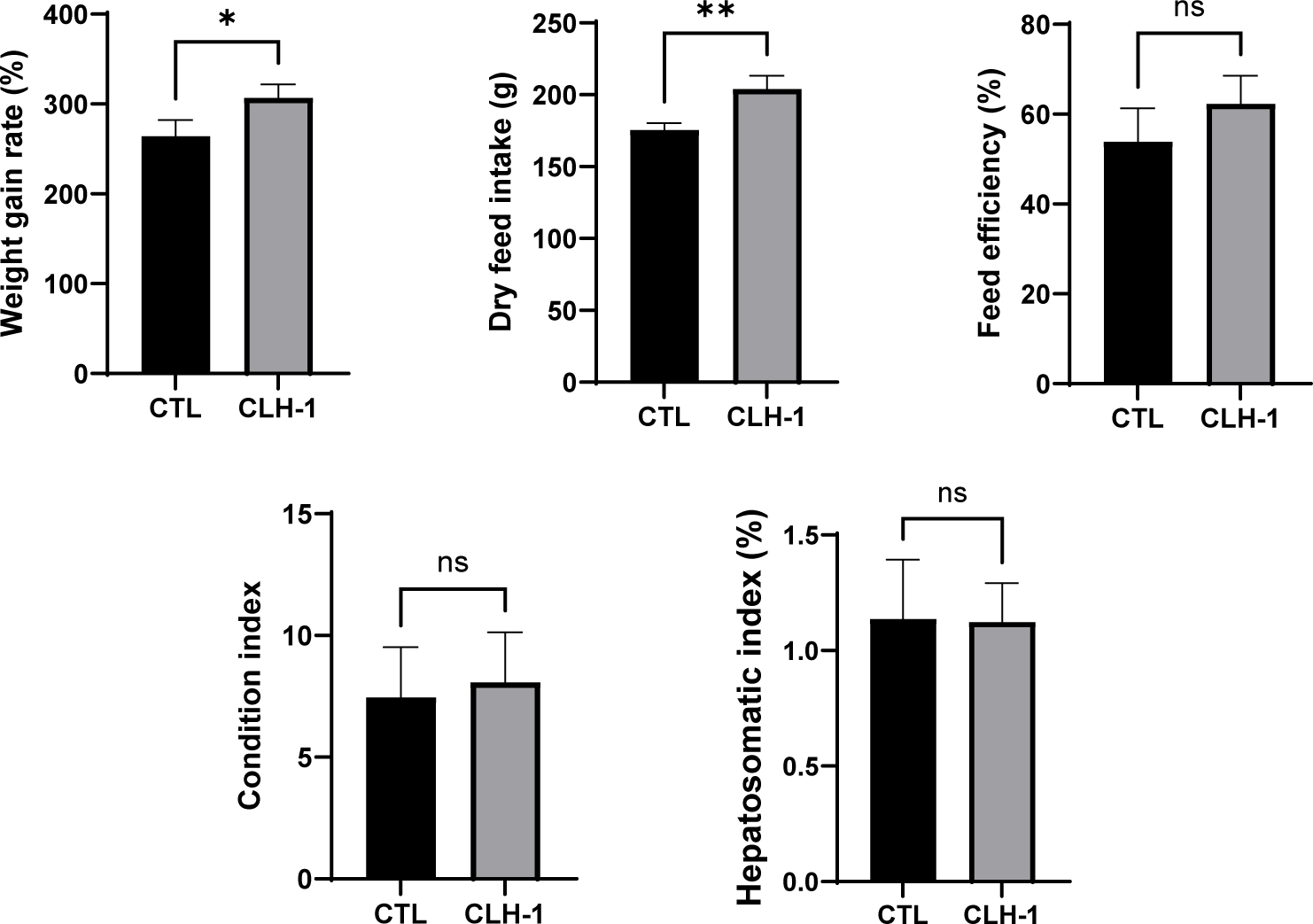
Non-specific immune activity and blood chemical elements of eels fed CTL and CLH-1 diets for 8 weeks were evaluated. Lysozyme activity (CTL, 0.04 ± 0.00 unit; CLH-1, 0.03 ± 0.01 unit) and phagocytic activity (CTL, 0.08 ± 0.00 unit; CLH-1, 0.08 ± 0.00 unit) were both evaluated at almost the same value. Similarly, no significant changes in all blood biochemical factors examined, including GOT (CTL, 133.00 ± 38.64 U/L; CLH-1, 157.00 ± 54.63 U/L), HDL-C (CTL, 391.00 ± 70.35; CLH-1, 428.00 ± 46.82), TP (CTL, 4.40 ± 0.41 U/L; CLH-1, 4.80 ± 0.17 U/L), ALP (CTL, 1632.00 ± 323.33 U/L; CLH-1, 1668.50 ± 512.40 U/L), GLU (CTL, 97.50 ± 13.05 U/L; CLH-1, 130.00 ± 23.54 U/L) and TCHO (CTL, 417.00± 79.56 U/L; CLH-1, 437.00 ± 77.03 U/L), between eels fed CTL and CLH-1 diet (Table 3). These results indicate that the replacement of 1% fishmeal with Harim-CLHTM in the diet does not significantly affect the non-specific immune responses or the major blood biochemical markers in eels.
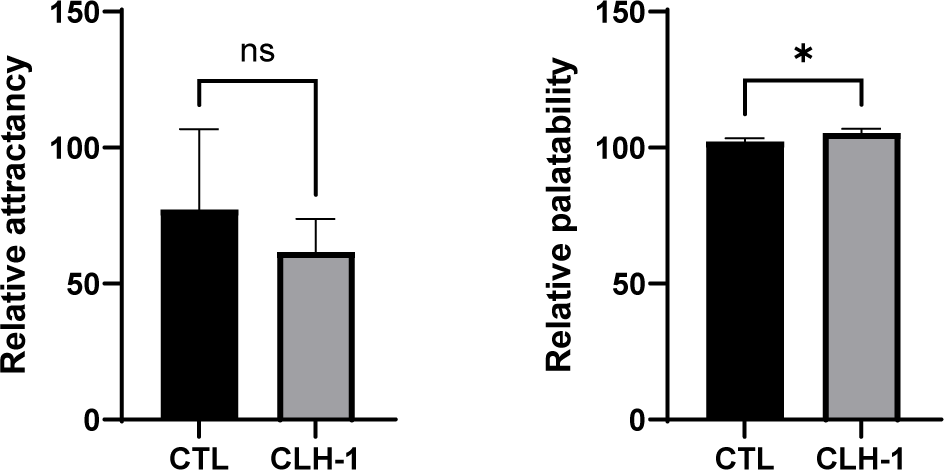
The attractancy and palatability of CTL diet and CLH-1 diet were evaluated. While attractancy did not show statistically significant difference between diets, the CTL diet was higher average attractancy (77.3 ± 24.1 eels) compared to the CLH-1 (61.7 ± 9.9 eels), indicating that CLH-1 did not surpass the attractancy of the traditional fishmeal-based diet. In contrast, palatability exhibited a significant difference, with CLH-1 diet showing a notably higher intake (106.9 ± 1.2, measured over 7 feeding instances) compared to the CTL diet (102.3 ± 1.0, measured over 7 feeding instances) (Fig. 3). This result underscores that CLH-1 effectively enhances palatability, encouraging greater feed consumption in eels.
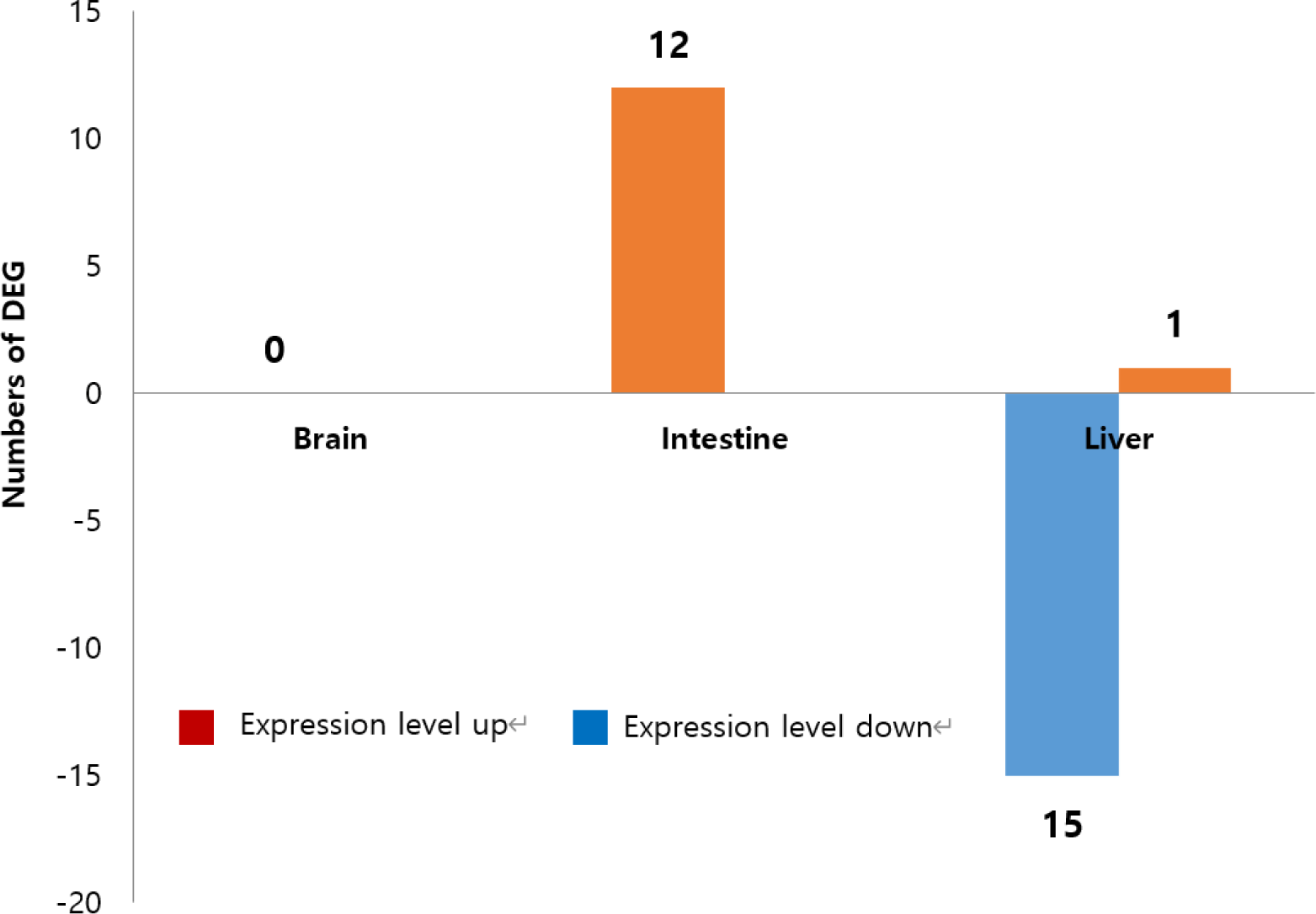
NGS was employed to analyze gene expression patterns in the brain, liver, and intestine tissues of eels fed with CTL (control) and CLH-1 diets for 8 weeks. No statistically significant DEGs were identified in brain tissue. On the other hand, in liver tissue, a total of 26 DEGs were identified, including 17 types of non-coding RNAs and 9 types of mRNAs (Fig. 4). The expression levels of 25 genes were decreased, except for one gene (XR_004762851.1), which showed an increase. In intestinal tissue, all 12 identified DEGs were non-coding RNAs, with all exhibiting an increase in expression levels. Interestingly, these 12 types of genes were also expressed in liver tissue but showed a down-regulation in the intestine and up-regulation in the liver. Specific findings in liver tissue included non-coding RNAs that are transcript variants of genes with currently unknown functions. Additionally, all nine types of mRNA specifically expressed in the liver showed down-regulation, including five variant genes (X1, X2, X3, X4, X5) derived from the gene LOC118226149. The functions of three genes (XM_035392166.1, XM_035415567.1, XM_035412709.1) remain unspecified (Table 4). These findings underscore the complex regulatory mechanisms influenced by diet in eel tissues and highlight potential areas for further research into the metabolic and functional implications of diet on gene expression in eels.
Discussion
Protein hydrolysates are increasingly recognized for their significant impacts on both palatability and functionality in food products. The process of hydrolysis breaks down proteins into smaller peptides and amino acids, enhancing flavor profiles and improving nutrient digestibility and absorption. Notably, protein hydrolysates often impart a savory or umami taste, significantly enhancing the overall flavor profile of food products. Beyond taste, some hydrolysates exhibit bioactive properties, including antioxidant, antihypertensive, and antimicrobial activities, which contribute to the health benefits of the foods they are added to (Neklyudov et al., 2000; Yang et al., 2024). Whey protein hydrolysate, soy protein hydrolysate, casein protein hydrolysate, and collagen hydrolysates are prominently used across various sectors. In sports nutrition and infant formulas, these hydrolysates provide high-quality plant proteins. They are also used in medical nutrition and contribute to joint health, skin health, and possess functional properties in food and beverages, reflecting their versatile applications (Li & Zhu, 2024; Qian et al., 2024). Furthermore, the physiological activities of various hydrolysates are well-documented, with many nearing commercialization due to their proven benefits (Das et al., 2021; Neklyudov et al., 2000).
CLH particularly stands out due to its rich nutrient composition and array of bioactive compounds. CLH finds extensive use in pet food as a flavor enhancer and nutritional supplement, showcasing its dual role as a functional ingredient. In human nutrition, it serves as a basis for functional foods that offer specific health benefits and is used as a flavor base in a variety of foods (Pinto et al., 2022; Wu et al., 2024). This multi-functionality of CLH underscores its potential for broader applications in both pet and human food industries.
Harim, a prominent company in Korea, operates extensively across various sectors including chicken feed production, breeding, processing, and distribution. It also ventures into innovative product development such as the creation of liver hydrolysate from broiler chicken by-products. Harim-CLHTM, developed by the company, serves primarily as a feed additive designed to enhance palatability and functionality rather than acting as a direct substitute for traditional protein sources like fishmeal. Notably, Harim-CLHTM is characterized by its low protein content and high carbohydrate levels, used as excipients to enhance taste and functional properties (Supplementary Table S2). While commonly utilized to improve the palatability of pet and poultry feeds, Harim-CLHTM has not traditionally been used in fish feed. This product exhibits a distinct composition, especially in terms of FAs, which are substantially higher than those found in fishmeal. These amino acids are crucial for imparting umami flavor, a key factor in enhancing feed attractiveness across animal species, including fish. Such umami taste-stimulating additives are known to play roles in digestive and absorptive functions and activate gut-brain axis mechanisms. These mechanisms are crucial for regulating feed intake through the transcriptional activation of amino acid transporters, taste receptors, signaling molecules, and hormones (Calo et al., 2024). In general, chicken meat protein hydrolysate, which includes peptides such as anserine and carnosine, exhibits various physiological activities known to benefit health (Intarapichet & Maikhunthod, 2005). However, these specific peptides were not detected in either Harim-CLHTM or fishmeal, and detailed information on the product’s manufacturing process and peptide content remains undisclosed (Table 2). There is also a notable difference in the fatty acid profiles between fishmeal and Harim-CLHTM. Essential fatty acids like DHA and EPA, critical in aquaculture, are present in higher amounts in fishmeal. In contrast, Harim-CLHTM contains higher levels of linoleic acid but shows a low overall composition ratio (0.94 ± 0.01%). While fatty acids are important, their role in influencing palatability in fish is minimal, suggesting that their presence in the CLH-1 diet, which includes 1% Harim-CLHTM, is likely not a major factor affecting feed preference (Supplementary Table S3).
Prior evaluations have recognized CLH as a palatable or functional additive in various animal diets, yet its application in eel (A. japonica) farming had not been previously explored until this study. Preliminary tests determined that a 1% concentration of Harim-CLHTM, the specific CLH product used, was optimal for feeding, based on undisclosed preliminary data. Over an 8-week feeding trial, the CLH-1 diet (containing 1% Harim-CLHTM) improved growth rates compared to the CTL, though it did not significantly enhanceFE. However, both total feed intake (TFI) and weight growth rate (WGR) exhibited almost proportional increases (Fig. 1).
In terms of attractancy and palatability compared to CTL diet, CLH-1 diet performed lower in attractancy (with no significant difference) but significantly higher in palatability (Fig. 2). This enhancement of palatability parallels findings from dog diets where CLH also showed positive effects (Hao et al., 2020), likely attributable to the high concentration of umami-affecting amino acids in the hydrolysate. Unlike the neutral effects on growth performance in juvenile tilapia (Gomes et al., 2023) and the reduced palatability at a 5% inclusion level in Nile tilapia diets, our findings suggest species-specific responses to CLH. The differences in response may be influenced by variations in the manufacturing process of Harim-CLHTM, its amino acid and peptide composition, and the proportions used as an additive.
Despite these growth and palatability outcomes, the effects on non-specific immunity and major blood health parameters in eels fed with the CLH-1 diet did not differ significantly from those on the CTL, indicating negligible physiological impacts. Moreover, while the liver weight index in the CLH-1 fed group was slightly lower than in the control group, it was not statistically significant. This observation aligns with previous studies that suggest CLH might influence liver function or fatty acid metabolism, noted in other species for its hepatoprotective functions, such as enhancing antioxidant activity and reducing lipid accumulation in high-fat feeding scenarios (Chen et al., 2017; Wu et al., 2024; Yang et al., 2024).
Overall, our results and those from previous studies indicate that CLH, independent of its production specifics, may offer beneficial effects on liver function and fatty acid metabolism. Future studies should consider varying inclusion levels, detailed biochemical analyses, and a broader range of aquatic species to fully elucidate the potential of CLH in aquaculture.
To extend our understanding beyond palatability, we investigated the potential functional effects of Harim-CLHTM on A. japonica eels by examining changes in gene expression across major tissues using NGS. Transcriptome analysis is increasingly utilized in experimental animals, including humans, to assess the dietary impacts of various chemical and natural additives (Khwatenge et al., 2020; Wang et al., 2022). Similarly, in aquaculture, transcriptomic profiles alongside microbiota studies are commonly employed to evaluate factors such as growth performance, antioxidant capacity, and intestinal histology (Lv et al., 2024; Sun et al., 2024).
In our study, we specifically analyzed DEGs in the brain, liver, and intestinal tissues to discern potential changes in gene expression that could affect physiological functions such as antioxidant activity, lipid metabolism regulation, liver function, and WG. Our DEG analysis revealed no significant changes in gene expression between eels fed the CTL and those fed the CLH-1 diet in the brain tissue. However, significant DEGs were observed in the liver and intestinal tissues, indicating alterations in the expression of non-coding RNA and some genes with currently unclear functions (Fig. 3 and Table 4). Interestingly, some DEGs were common between liver and intestinal tissues, exhibiting opposite expression patterns—upregulated in the liver and downregulated in the intestine. This suggests tissue-specific responses that might influence diverse physiological pathways. However, most identified DEGs were non-coding RNAs or genes with unknown functions, or were variants of some known genes, such as betaine-homocysteine 5-methyltransferase and fucolectin. This ambiguity in gene function makes it challenging to conclusively determine the impact of Harim-CLHTM on eel growth, antioxidant enhancement, or lipid accumulation reduction. The current findings underscore the complexity of dietary impacts at the molecular level and highlight the need for further investigation. Future studies should focus on exploring the specific roles of these DEGs in tissue-specific functions, alongside additional biochemical assays, to build a clearer picture of how dietary additives like Harim-CLHTM influence the health and performance of aquaculture species.

