Introduction
Marine snails are popular seafood in Asian countries, and a certain number of poisoning cases have been reported due to eating this animal group (Ha & Sato, 2010; Ha et al., 2023; Hwang et al., 1995, 2005; Shiu et al., 2003; Shui et al., 2003; Sui et al., 2002; Yang et al., 1995). Several marine snail’s families were known as causative organisms for these poisonings (Noguchi et al., 2011). It was indicated that tetrodotoxin (TTX) and saxitoxins are responsible toxins in these poisonings (Ha et al., 2020; Hwang et al., 2004, 2007; Liu et al., 2004; Narita et al., 1984; Taniyama et al., 2009, 2013).
TTX, a powerful sodium channel blocker of excitable membranes, is one of the marine toxins linked to human poisonings. The origin of TTX contamination in marine environments is still under investigation, with various hypotheses proposed (Varini et al., 2025). It has been known to spread widely, but only in a few and specific species of freshwater and marine cold-blooded organisms (Hwang et al., 1992; Kim et al., 1975; Lin & Hwang, 2001; Miyazawa & Noguchi, 2001; Mosher & Fuhrman, 1984), bacteria (Noguchi et al., 1986; Yasumoto et al., 1986) and macroalgae (Yasumoto et al., 1988). TTX poisoning has been reported mainly in Southeast Asia, China, Korea, Japan, where the potential toxic animals such as puffer fish, horshoe crab, toxic goby, toxic snails and others are eaten (Hashimoto, 1979; Mosher & Fuhrman, 1984).
In Viet Nam, poisoning incidents by eating marine snails have been happened, sometimes, mostly in coastal areas (Ha & Sato, 2010; Ha et al., 2020). In our preminary studies, 05 marine Nassaridae snail species (Nassarius conoidalis, Nassarius glans, Nassarius papillosus, Nassarius pullus, and Nassarius siquijorensis) were found to contain TTXs (Dang et al., 2015; Ha et al., 2020). This data indicated the potential threat to human health from marine snails in Viet Nam. However, in almost of all case, causative toxin(s) in poisoning incidents have not been confirmed due to lacking of specimen collection, excepted in the incident caused by eating Nassarius glans in Khanh Hoa 2020 (Ha et al., 2020).
In March 2021, there was a poisoning incident involving 05 family members as result of eating marine snails in Phan Thiet City, Binh Thuan Province, Viet Nam. About 30 min after eating, four out of five persons were experienced with typical neurological symptoms such as tingling on lips, touge and limbs. This paper presents result of toxin analysis in the implicated snail samples, which was later identified as Phos senticosus (Linnaeus, 1758) (Fig. 1) collected in the incident. Also, the toxicity variation of 29 collected snail individuals was documented. It is the first scientific report on P. senticosus causing poisoning in Viet Nam, therefore, it is important for public awareness on human health risk from this marine species.

Materials and Methods
As leftover food, 29 snail individuals were collected in the poisoning incident in Phan Thiet City, Binh Thuan Provice in March 2021 and sent to the laboratory in cool condition. Each snail specimen was cleaned outside, identified scientific name, measured and deshelled to collect soft tissue (Table 1) for toxin analysis.
| n | Length (cm) | Width (cm) | Whole body weight (g) | Weight of soft tissue (g) |
|---|---|---|---|---|
| 29 | 3.86 ± 0.29 | 1.98 ± 0.18 | 5.79 ± 1.03 | 2.70 ± 0.63 |
Formic acid, acetic acid and TTX (1 mg) were Wako pure chemicals (FUJIFILM, Osaka, Japan) products. Ammonium hydroxide of 25% (liquid chromatography-mass spectrometry [LC-MS] grade) was a Sigma-Aldrich (Tokyo, Japan) product. Acetonitrile was Kanto Chemicals (Tokyo, Japan) product. 4-epiTTX and anh-TTX were a gift from Dr. Shigeru Sato, Kitasato University, Japan.
For food safety consideration, all snail soft tissue that is frequently consumed by people was used for toxin analysis. Additionally, the soft tissue of each individual snail was only around 2 g (Table 1) due to the snail species’ small size, replicates were not applied during the extraction procedure. Since TTXs was the predominant toxin in a number of marine toxic species in Viet Nam, this toxin was the focus of this investigation. TTXs in the soft tissue was extracted following to Brillantes et al. (2003). Briefly, the soft tissue was homogenized with 1% acetic acid (1:4 w/v), boiled for 5 min, cooled down at room temperature and centrifuged at 11,000×g, 25°C for 10 min to collect the extract, which one ml was equivalent to 0.2 g of the soft tissue. The extract was then passed through an ENVI-Carb SPE cartridge (250 mg, 3 mL–1, Sigma-Aldrich, St. Louis, MO, USA), eluted by acetonitrile 25%. TTXs in the eluates were then dertemined by the hydrophilic interaction liquid chromatography-tandem mass spectrometry (HILIC-MS/MS) method following to Boundy et al. (2015) with some modifications using the liquid chromatography (LCMS 8040, Shimadzu, Kyoto, Japan) coupled to a triple quadrupole/linear ion trap mass spectrometer DUIS-8040 (Shimadzu). The HILIC separation was carried out on a Waters Xbrige (HILIC) Amide column (4.6 mm I.D. × 150 mm, 3.5 μm, WatersTM, Milford, MA, USA) at 60°C with injected volume of 5 μL. Water/formic acid/ammonium hydroxide (500:0.075:0.3 v/v/v) (A) and acetonitrile/water/formic acid (700:300:0.1 v/v/v) (B) were set in a flow rate of 0.6 mL min–1 as mobiphases. The chromatographic conditions were initial condition of 100% B for 20 min; following of a linear gradient of 50:50 A and B within 15 min, held for 9.90 min.
The ion source parameters of the MS spectrometer were 10 V of entrance potential, 30 psi of curtain gas; 4,500 V of ion spray voltage; 250°C of source desolvation temperature; 400°C of source ion block temperature; 1,000 Lh–1 of desolvation gas flow; 2 L min–1 of nebulizer gas flow; and 0.15 mL min–1 of collision gas flow rate. Multiple reaction monitoring (MRM) was performed in a positive electrospray ionization. Due to the limitation of reference material (TTX standard), single-point calibration was applied in the analysis, also recovery test with spiked samples were not carried out in this study. Practically, the limit of detection and the limit of quantitation were 38 nM and 126 nM for TTX analysis on our HILIC-MS/MS system. MS/MS spectra were obtained with 40 eV of collision energy for the precursor of m/z 320.0 within a range of m/z 50–350 for confirmation of TTX in the snail extract.
TTX levels were calculated from HILIC-MS/MS data by comparing with TTXs standard. Toxicity was expressed in mouse unit (MU/g) in which one MU is the dose of toxin that will kill a male mouse (ddY, 20 ± 2 g body weight) within 30 min. One mg TTX is corresponding to 4,500 MU, one mg 4-epiTTX to 709 MU and one mg anh-TTX to 92 MU of toxicity (Nakamura & Yasumoto, 1985).
Results
The HILIC/MRM chromatogram revealed that the retention times (Rt) for the anh-TTX, 4-epiTTX and TTX standards were 24.17, 25.58 and 26.50 minutes, respectively (Fig. 2). The corresponding peaks that were seen in all extracts from soft tissue of P. senticosus indicating the presence of TTXs. The MS/MS spectrum at m/z 162, 178, 256, 284, 302 and 320; which match to the indentical MS/MS spectrum of the TTX standard were detected in the snail extract, confirmed TTX in the sample (Fig. 3).
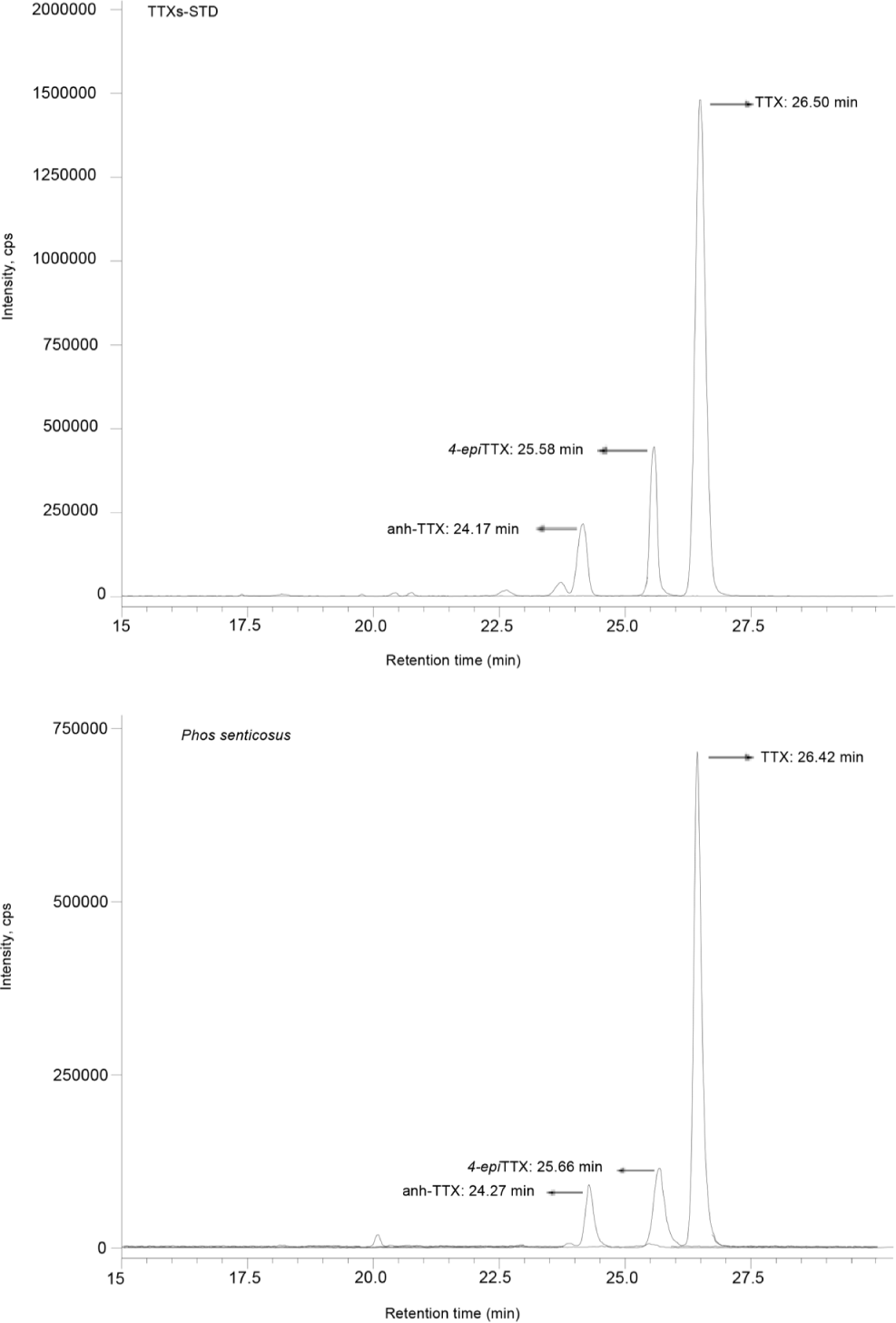
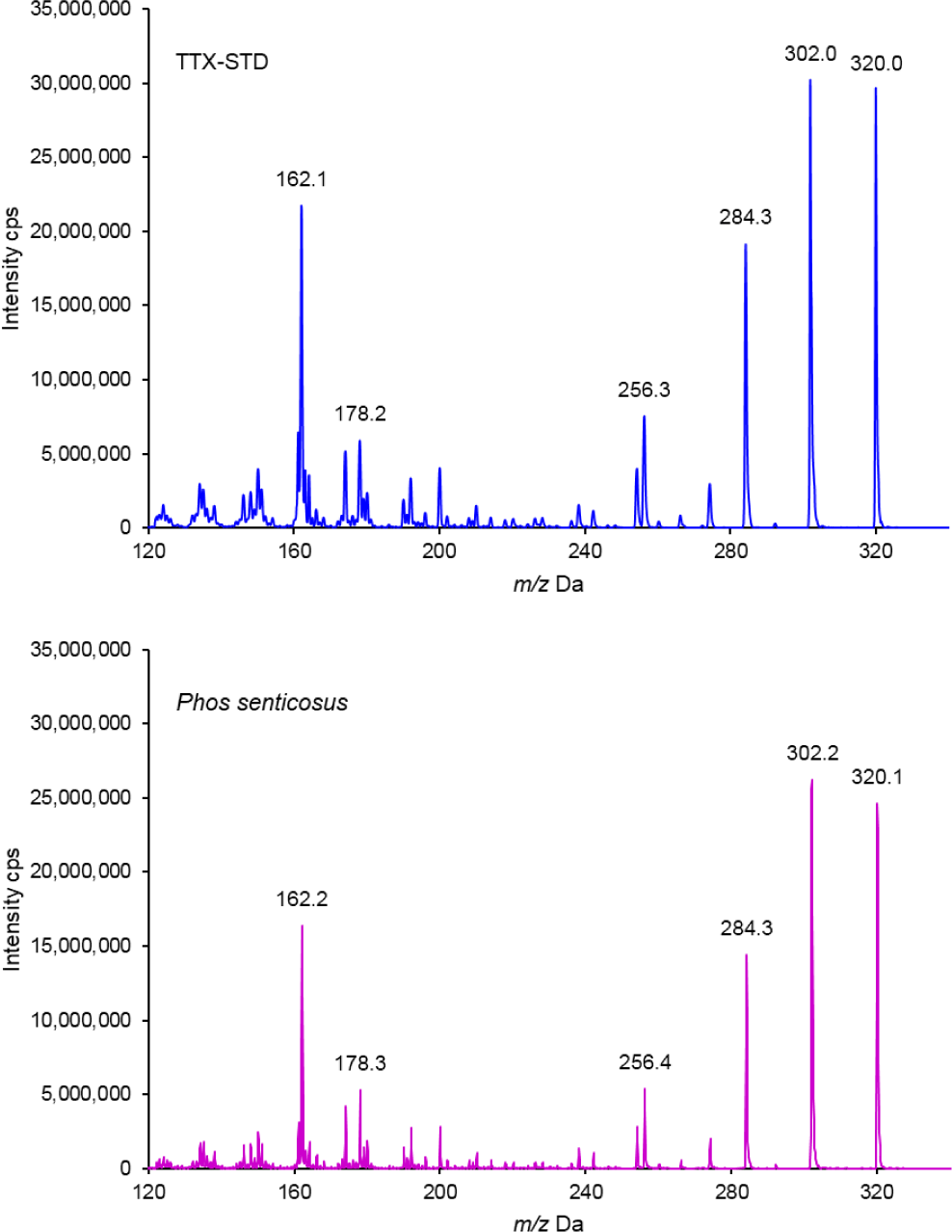
Table 2 presents level of TTXs and overall toxicities (MU/g) calculated based on the specific mouse toxicity of each toxin component (Nakamura & Yasumoto, 1985) in 29 specimens of P. senticosus. At 60.7 ± 126.5 µg g–1, anh-TTX was detected in the most dominant level, followed by TTX (46.0 ± 107.6 µg g–1) and then 4-epiTTX (23.3 ± 24.3 µg g–1). The overall toxicity in these specimens was estimated as 229 ± 526 MU g–1 with the range from 10 to 2,672 MU g–1, with 90% was responsible from TTX due to it strong potency.
| Value | Level of TTXs (µg/g) | Overall toxicity (MU*/g) | ||
|---|---|---|---|---|
| anh-TTX | 4-epiTTX | TTX | ||
| Range | 5.3–603.9 | 1.9–233.7 | 1.7–544.5 | 10–2672 |
| Mean ± SD | 60.7 ± 126.5 | 23.3 ± 24.3 | 46.0 ±107.6 | 229 ± 526 |
One MU is the dose of TTXs which kills a 20 g male mouse (ddY) in 30 min (Nakamura & Yasumoto, 1985).
All specimens were recognized as toxic, with 75.8% exhibiting toxicity in range of 10–100 MU g–1, as shown in Table 3. In particular, 17.3% specimens had TTX toxicity between in range 100–1,000 MU g–1, whereas 6.9% displayed toxicity that was exceptionally high (> 1,000 MU g–1) (Table 3).
| TTX toxicity range (MU/g) | 10–100 | 100–1,000 | > 1,000 |
|---|---|---|---|
| Number of specimens | 22 | 5 | 2 |
| Frequency (%) | 75.8 | 17.3 | 6.9 |
Discussion
The amount of TTX found in P. senticosus specimens was less than the amount of anh-TTX, as shown in Table 2. However, TTX was responsible for most of the total toxicity because its toxic potency (4,500 MU mg–1) was over 50 times higher than that of anh-TTX (92 MU mg–1) which noted by Nakamura and Yasumoto (Nakamura & Yasumoto, 1985). It shows that the toxin that caused this poisoning incidence was TTX.
Among more than 15 Nassarius snail species known to be distributed in Viet Nam (Hylleberg & Kilburn, 2003), 5 species were reported to contain certain levels of TTX (Dang et al., 2015; Ha et al., 2020) and later, one of them, Nassarius glans was confirmed to cause the poisoning incident in Khanh Hoa Province, 2020 (Ha et al., 2023). P. senticosus (Linnaeus, 1758), which is also member of Nassariidae family, known to distribute widly in Indo-West Pacific (Abbott, 1991; Cernohorsky, 1972; Okutani, 2000; Wilson, 1994); however, there is little information available regarding TTX this species. The present data is the first report on the presence of TTX in P. senticosus, also it supports our previous data that TTX is a dominant toxin in marine snails in Viet Nam (Ha et al., 2020). According to the findings, it is suggested that Nassarius snails pose a health concern to people in Viet Nam.
The toxicity level detected in the specimens was all beyond the safe level of consumption (10 MU/g) for puffer (fish) suggested in Japan (Kodama & Sato, 2005) and the European Food Safety Authority (EFSA) for TTX (which sets a safety limit of 44 µg/kg TTXs in shellfish meat) (EFSA et al., 2017). The highest TTX toxicity in this study was higher than that detected in some other Nassarius species in Viet Nam in our earlier report (Ha et al., 2020) and comparable with that detected in Nassarius glans in Taiwan (Hwang et al., 2005). It is claimed that P. senticosus in Viet Nam is unsafe for human consumption, even if this level is still lower than that of Nassarius glans from Viet Nam in our latest report (Ha et al., 2023) and Japan (4,290 MU g–1) (Taniyama et al., 2009). The minimum human lethal dose of TTX is estimated to be approximately 10,000 MU (Noguchi et al., 2011). Similar to many other marine snails, P. senticosus is frequently eaten by not only locals but also foreign tourists in Viet Nam. According to the present data, only 10 g soft tissue (equivalent to 4 specimens) containing maximum toxicity (2,672 MU g–1) may cause death for people if consumed. In addition to that, frequency of toxic specimens of P. senticosus in this study was quite high (100%) with a certain number of extremely toxic specimens (6.9%). Although the number of specimens in this study was small, the results indicate that P. senticosus at least in Viet Nam is unsafe, even quite dangerous for human food. More attention has to be paid to this possible source of poisoning.
Certain snail species, particularly marine snails like the trumpet shell, can accumulate high concentrations of TTX (Costa et al., 2021). This accumulation can occur through the food chain, where snails ingest TTX-bearing organisms like certain starfish or dead puffers, leading to trophic transfer of the toxin (Noguchi & Arakawa, 2008). There was a hypothesis that the origin of TTX in the small necrophagous snails including Nassariidae species originate from their food, however still uncertain due to geographical dispersion and seasonal variation (Noguchi et al., 2011). For instance, in contrast to spring to early summer in China and Taiwan or late summer in Japan (Noguchi et al., 2011), snail poisoning incidents were observed in Viet Nam in autumn (Ha et al., 2023). TTX in P. senticosus together with their food sources would be an interesting for understanding the mechanism of toxin contamination in this marine animal’s group.
Conclusion
For the first time, TTX was verified as the responsible toxin in the marine snail P. senticosus specimens causing the poisoning incident in Binh Thuan Province, Viet Nam in March 2021 by HILIC-MS/MS analysis. Given the high level and wide range of TTX toxicity found in the specimens, it is unsuitable, even dangerous, for human consumption, at least in Viet Nam. There should be a more stringent warning about this species’ potential to cause poisoning. To further understand the genesis of the toxin and its accumulated mechanism, more research is required on the geographical and seasonal variation of TTX in P. senticosus.
