Introduction
Tofu is a traditional oriental food made by grinding soybeans with water to precipitate and solidify into a gel form, consisting of soluble protein glycinin and minerals such as Ca2+ and Mg2+ (Choi et al., 2020; Kim, 2016). Soybeans, as the raw material for tofu are inexpensive (Kim & Yoon, 2024), highly digestible and absorbed well in the body (Kim et al., 2021). They are rich in essential amino acids, which are often lacking in the grain-based diet of Koreans, making them a valuable supplement (Jung & Kim, 2016). Moreover, soybeans are notably abundant in lysine, comparable to animal protein sources (Sim et al., 2024). Tofu is known to have various health benefits, including cholesterol reduction, cardiovascular health promotion, cancer prevention, osteoporosis inhibition, and acquired immunodeficiency syndrome (AIDS) infection suppression (Jung & Kim, 2016). To enhance the taste and nutritional content of tofu, many studies have reported the addition of ingredients such as Gloiopeltis furcata (Kim et al., 2021), Codium fragile (Choi et al., 2020), barley sprout powder (Park & Yoo, 2023), redbeat (Beta vulgaris L.) (Lee et al., 2019), and green tea powder (Jung & Kim, 2016).
Collagen is a fibrous protein that accounts for about 30% of body proteins. About 40% of collagen in the body is contained in bones and cartilage in the skin epidermis, and is distributed in various places such as blood vessels and tooth tendons (Kim & Shin, 2021; Lee & Cho, 2010). Collagen is a major component of connective tissue, connecting bones and ligaments (Choi, 2015). Collagen peptides derived from protein hydrolysates increase the diameter of collagen fibers in the dermis, increasing density and enhancing skin moisture and elasticity, making them widely used in cosmetics (Kim et al., 2011). Additionally, oral consumption of collagen has been shown to prevent aging and hair loss, positively impact ligaments, bones, and cartilage, and improve the condition of hair, nails, and toenails (Kim et al., 2020; Lee et al., 2013). The collagen commonly consumed by humans is extracted from the skin and bones of cow or pigs, which are large molecules with a molecular weight of about 300 kDa, resulting in low absorption rates in the body (Aguirre-Cruz et al., 2020). Therefore, low molecular fish collagen (LMFC) with a molecular weight of 1,000–5,000 Da has been developed through research (Kim et al., 2020). The demand for LMFC, produced from sources such as pollock (Lee et al., 2022), shark (Sukumaran et al., 2013), flatfish (Martins et al., 2022), salmon (Tarigan & Tarigan, 2022), and jellyfish (Kim et al., 2016), is increasing. Jo & Choi (2023) found that reactive oxygen species generated through respiration and UV exposure specifically activate the proteolytic enzyme matrix metalloproteinase (MMP)-1, which accelerates skin aging. Consequently, recent studies have reported that LMFC effectively inhibits MMP-1, proving its antioxidant activity (Lee, 2018). Furthermore, LMFC exhibits antioxidant effects in cartilage and joints by promoting synovial fluid secretion, cartilage metabolism, and anti-inflammatory effects, thereby suppressing joint inflammation (Sukumaran et al., 2016).
With the proven antioxidant efficacy of fish collagen, it is widely available in powder form; however, research on food manufacturing using fish collagen is scarce. Therefore, in this study, we added LMFC powder to tofu, a traditional food that we commonly consume, in various proportions to easily obtain its antioxidant properties. We also analyzed the general components (crude ash, crude protein), antioxidant activity (2,2’-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) [ABTS], 1,1-diphenyl-2-picrylhydrazyl [DPPH]), quality characteristics (yield, texture, pH, color), aiming to demonstrate the superiority of LMFC and provide basic data for the development of tofu products supplemented with LMFC.
Materials and Methods
The manufacturing process of tofu with LMFC is illustrated in (Fig. 1). The soybeans used in this study were harvested from Gangwon-do Province, and the LMFC used was a product containing 98% fish collagen in powder form. Tofu was manufactured with LMFC content of 0%, 10%, and 20%. The total weight of soybeans and powder was maintained at 200 g. For tofu with 0% collagen, 200 g of soybeans were used; for 10% collagen, 180 g of soybeans and 20 g of powder; and for 20% collagen, 160 g of soybeans and 40 g of powder were used. The soybeans were washed and soaked in room temperature water for about 20 hours. After soaking, the soybeans were ground using a blender with the addition of 150 mL of water. The resulting mixture was then filtered through a cloth to separate the soybean pulp (okara) from the soybean milk. The soybean milk was heated to 80 ± 3°C, stirring until foamy, and then coagulated by adding magnesium chloride (MgCl2·6H2O). After allowing it to set, the coagulated mixture was placed in a mold and pressed at 75 psi for 30 minutes to shape it. The resulting tofu was then cut into uniform dimensions of (12 × 12 × 3 cm).
The yield of tofu with LMFC was calculated using the method described by Park & Yoo (2022). The yield (%) was determined by dividing the weight of the manufactured tofu by the weight of the raw soybeans and then multiplying by 100 to express it as a percentage.
10 g of tofu sample was homogenized with 20 mL of distilled water and the filtered liquid was measured using a YSI 63 pH meter (Orion Star A211, Thermo Fisher Scientific, Waltham, MA, USA).
The tofu was cut into uniform pieces (2 × 2 × 2 cm) and the hardness, chewiness, cohesiveness, and springiness were measured in Texture Profile Analysis (TPA) mode using a CT3 texture analyzer (Brookfield Engineering Laboratories, Middleboro, MA, USA). The measurement conditions were as follows: probe, stainless steel probe-type TA18 12.7 mm; test speed, 1.0 mm/s; force threshold, 20 g; distance threshold, 0.50 mm; compression limit, 50% deformation.
The color of tofu with added LMFC was measured using a colorimeter (NE-4000, Nippon Denshoku, Tokyo, Japan). The standard plate values for CIE L* (lightness), a* (redness), and b* (yellowness) were 97.32, –0.37, and 0.33, respectively.
The general components of tofu with added LMFC were analyzed according to the Association of Official Analytical Chemists International (AOAC) (1995) method: crude protein content was determined using the Micro-Kjeldahl method, and crude ash content was calculated using the dry ashing method at 550°C.
The ABTS radical scavenging activity of tofu with added LMFC was measured using the method of Kim et al. (2021). ABTS 7.4 mM and potassium persulfate 2.6 mM were mixed in equal proportions and left in the dark for 12–16 hours to form ABTS cations. The solution was then diluted with distilled water until its absorbance at 734 nm was 1.5–1.6. Tofu samples and the ABTS solution were added in varying concentrations to a total volume of 200 μL, and the absorbance at 734 nm was measured after 10 minutes. The radical scavenging activity was calculated and expressed as a percentage using the following formula.
The DPPH radical scavenging activity of tofu with added LMFC was measured according to the method of Choi et al. (2020). A mixture of 0.003 g of DPPH reagent (Sigma-Aldrich, St. Louis, MO, USA) and 50 mL of 99.9% ethanol was left in the dark for 30 minutes. Tofu samples and the DPPH solution were added in varying concentrations to a total volume of 200 μL, then left in the dark for 30 minutes. The absorbance was measured at a wavelength of 517 nm. DPPH radical scavenging activity was calculated using the formula below and expressed as a percentage (%).
The yield, antioxidant activities (ABTS radical scavenging activity, DPPH radical scavenging activity), pH, general components (crude ash, crude protein), and texture of tofu with added LMFC were analyzed using the mean values and standard deviations obtained from three replicates per sample for all experiments. Statistical analysis was conducted using the SPSS version 12.0 software program (SPSS, Chicago, IL, USA), employing one-way analysis of variance (one-way ANOVA) and Duncan’s multiple range test to verify significant differences at the 5% level (p < 0.05).
Results and Discussion
The measurement values for yield of tofu with added LMFC are presented in Table 1. The yield of tofu was highest in the control group (tofu without added LMFC) at 164.79%, followed by 147.70% for 10% collagen content, and 140.49% for 20% collagen content, indicating that as the amount of LMFC increased, the yield decreased (p < 0.05). Generally, the yield of tofu is reported to be directly influenced by its moisture content and soluble protein content (Choi et al., 2020). Lee et al. (2019) reported that the presence of natural substances could interfere with soy protein binding, resulting in lower yields as the content of natural substances increases. Consistent with our findings, previous studies have reported decreased tofu yields when additives such as Salicornia herbacea L. (Kim et al., 2010), Enteromorpha intenstinalis powder (Chung, 2010), and Gloiopeltis furcata (Kim et al., 2021) were added. According to Rekha & Vijayalakshmi (2013), factors such as insufficient grinding of soybeans during tofu manufacturing, thermal denaturation of soybean proteins due to excessive grinding, improper coagulation, and excessive agitation during coagulation directly affect yield reduction. Additionally, Sim et al. (2014) reported that an excess of coagulant could lead to the leakage of uncoagulated parts or impair coagulation, thereby affecting yield. However, in this study, we maintained a consistent amount of coagulant, and therefore, differences in yield due to coagulant quantity were not observed. Thus, the decrease in yield is attributed to the addition of LMFC affecting the yield of tofu.
The pH measurement values of tofu with added LMFC are presented in Table 1. The pH of tofu was 6.37 in the control group and 6.32–6.33 in the LMFC addition groups, showing a significant decrease (p < 0.05). However, there was no significant difference between 10% and 20% additions (p > 0.05). In a study by Lee & Jeong (2009), the pH of collagen peptides extracted from fish scales was reported to be 5.7, indicating slight acidity. Similarly, in a study by Yoo et al. (2013) on tofu supplemented with puffer fish powder, it was reported that the pH decreased due to the acids generated when the puffer fish powder was added while the soy milk was boiling. According to Musyoka et al. (2018), foods with lower pH have higher preservation effects than those with higher pH. Therefore, it is inferred that adding LMFC can contribute to improved storage stability due to the decrease in pH.
The texture and color analysis values of tofu with added LMFC are shown in Table 2. Hardness ranged from 268.33–172.33 g, decreasing with higher content of LMFC (p < 0.05). Chewiness also decreased with higher collagen content, ranging from 134.00–51.00 g (p < 0.05). There was no significant difference in springiness (p > 0.05). These differences in physical properties are considered to be due to the natural materials used in tofu production, the physical state (liquid, solid, etc.), the solid content, the amount of coagulant added, and the protein content and composition (Choi et al., 2020).
The appearance and color values of tofu with added LMFC are presented in (Fig. 2) and Table 3, respectively. As the amount of LMFC increased, the color of the tofu became progressively more yellow (Fig. 2). The L value, representing lightness, significantly decreased from 87.78%–82.43% (p < 0.05). The a value, representing redness, and the b value, representing yellowness, showed no significant differences (p > 0.05). However, the ΔE, significantly increased from 19.00%–23.26% (p < 0.05). According to Lee & Jeong (2009), LMFC has a pale yellow color, and when natural substances are added, they impart their inherent color to the product. Therefore, it is considered that the collagen powder decreased the lightness and increased the ΔE of the tofu.
The values for crude protein and ash content in tofu with the addition of LMFC are shown in Table 1. The crude protein content was highest at 11.38% in the 20% collagen group (p < 0.05), while the control group (10.77%) and the 10% collagen group (10.78%) showed no significant difference (p > 0.05), although the 10% collagen group had a slightly higher value compared to the control group. According to Lapi et al. (2021) fish collagen is rich in amino acids and minerals, indicating that fish collagen contains protein. The ash content was highest in the control group at 1.60% (p < 0.05), with the 10% (1.37%) and 20% (1.44%) collagen groups showing no significant difference (p > 0.05). According to Nóbrega et al. (2024) the ash content of collagen from peacock bass skin is 1.5%. The difference may be due to the varying amounts of LMFC added; however, since the amount of soybean also changed when adjusting the collagen content, it is presumed that the reduction in ash content was due to the decreased amount of soybean.
Antioxidant activity refers to the extent to which redox molecules in food and biological systems can neutralize free radicals that cause cellular damage (Floegel et al., 2011; Kim et al., 2021). According to Zhang et al. (2016), collagen peptides extracted from tilapia skin have been shown to increase the scavenging rates of ABTS and DPPH free radicals.
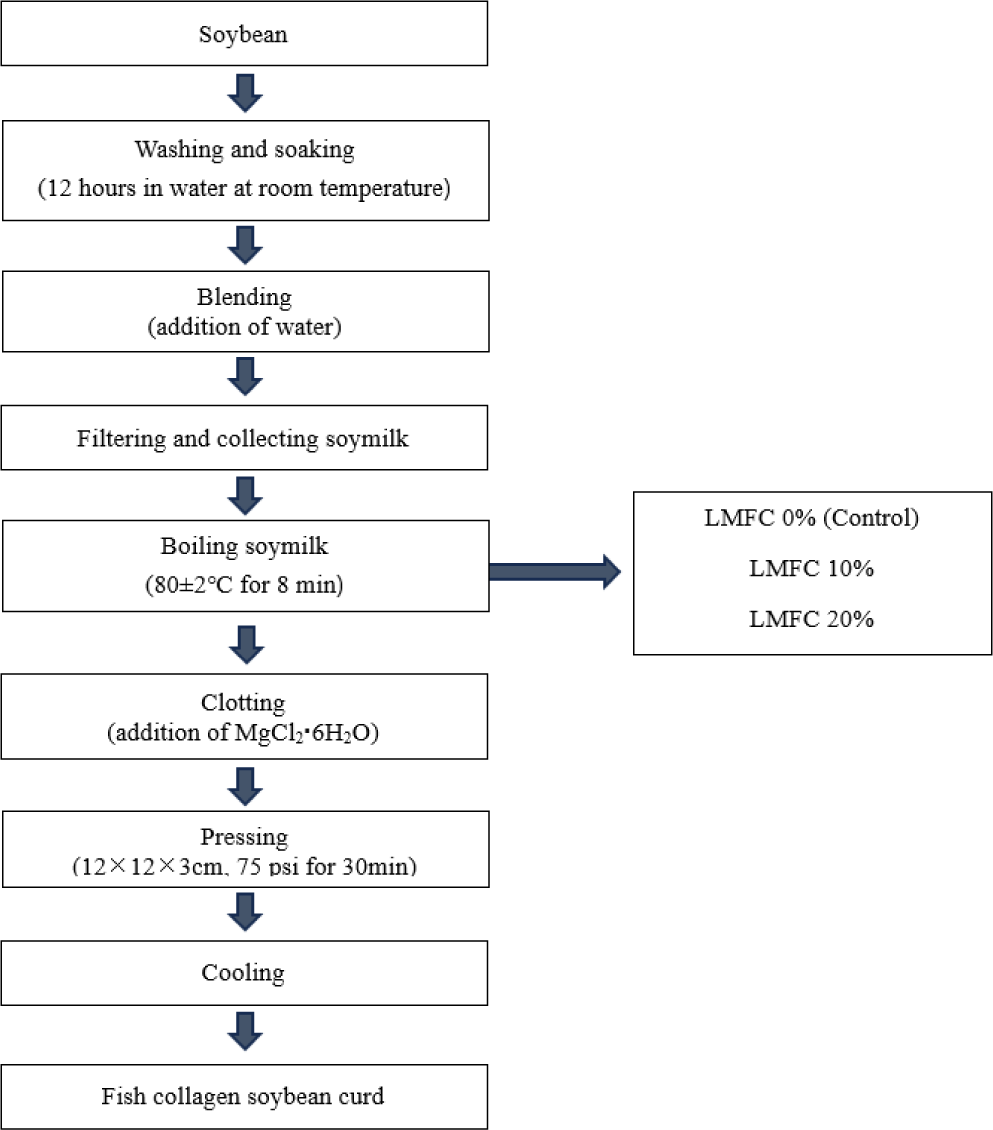
The DPPH radical scavenging activity of tofu with LMFC is presented in (Fig. 3). The control group had the lowest activity at 56.50%, while the 10% collagen group showed 74.49% and the 20% collagen group showed 87.00%. It is evident that the DPPH radical scavenging activity significantly increases with the addition of LMFC (p < 0.05).
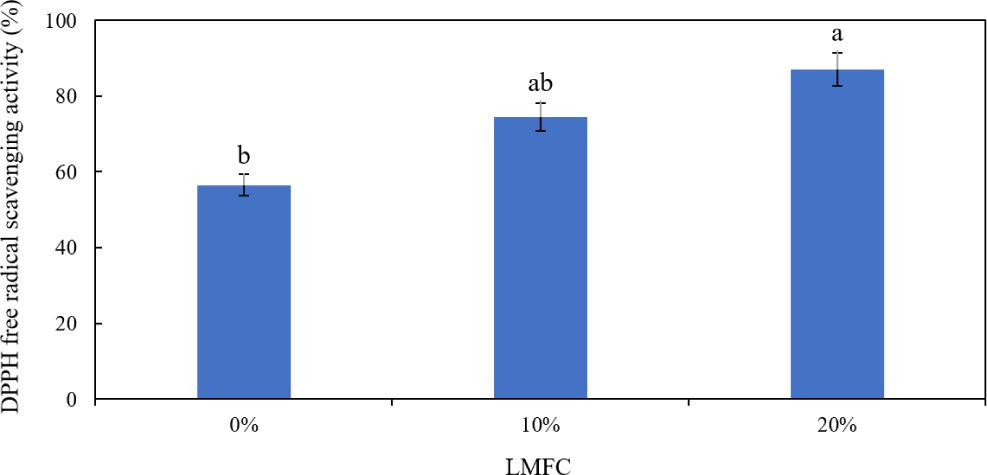
The ABTS radical scavenging activity of tofu with LMFC is presented in (Fig. 4). The control group had the lowest activity at 57.39%, while the 10% collagen group showed 74.15% and the 20% collagen group showed 88.96%. It is evident that the ABTS radical scavenging activity significantly increases with the addition of LMFC (p < 0.05).
Palamutoğlu & Kasnak (2019) reported that the addition of LMFC to meatballs does not alter sensory characteristics and exhibits superior antioxidant activity, suggesting the promising potential of LMFC in the food industry. Similarly, consistent with the results of this study, the addition of collagen powder to noodles (Kwon et al., 2013), collagen powder to Yanggaeng (Choi, 2015), and LMFC to yogurt (Ayati et al., 2022) has been reported to enhance antioxidant activity. In the present study, the significant increases in ABTS and DPPH values with the addition of low-molecular-weight fish collagen (p < 0.05) indicate that supplementation with low-molecular-weight fish collagen improves antioxidant capacity.
Conclusion
The research results showed that tofu manufactured by adding 10% and 20% LMFC powder exhibited a significantly increased antioxidant effect compared to the control group (tofu without added LMFC). Additionally, as the amount of LMFC increased, the yield and pH decreased, and the texture and color of the tofu changed according to the collagen addition. This is attributed to the unique characteristics of collagen. There were also fluctuations in the general components of tofu, such as total protein and ash content, which increased as the collagen content increased.
Therefore, in this study, we examined the antioxidant activity and physicochemical quality characteristics of tofu made from soybeans (without fish collagen) and tofu supplemented with 10%–20% LMFC. By doing so, we aimed to demonstrate the commercial potential of LMFC—added tofu and the feasibility of developing various collagen—processed food products using LMFC. However, further research on sensory evaluation and storage stability is necessary to popularize LMFC tofu.

