Introduction
The Nile tilapia (Oreochromis niloticus) is a vital freshwater species, significantly contributing over 60% of Ethiopia’s annual fish yields and supporting both subsistence and commercial fisheries (Golubtsov & Mina, 2003). Its success is attributed to rapid growth, tolerance to varied water quality, and flexible reproductive strategies influenced by local conditions (Froese & Pauly, 2023).
The feeding ecology of O. niloticus has been extensively studied across both native and introduced ranges, revealing a highly adaptable omnivorous feeding strategy that enables the species to exploit diverse food resources depending on environmental availability and ontogenetic stage (Getabu et al., 2003). Moreover, dietary studies have shown that juvenile O. niloticus typically consume higher proportions of animal protein in their diets, including zooplankton, benthic invertebrates, and detritus-associated microorganisms, whereas adult individuals increasingly incorporate plant materials such as phytoplankton, macrophytes, and organic detritus (Moriarty & Moriarty, 1973). This ontogenetic dietary shift reflects morphological and physiological adaptations, including changes in gut length, digestive enzyme production, and mouth morphology that enhance the efficiency of plant material processing in larger individuals (Moran et al., 2005).
Seasonal variations in feeding patterns have been documented across multiple African lake systems, with studies demonstrating that O. niloticus diet composition responds dynamically to temporal changes in prey availability, water level fluctuations, and reproductive cycles (Getabu et al., 2003). Research in Lake Victoria, Lake Turkana, and other lakes have revealed that phytoplankton typically dominates the diet during periods of high primary productivity, while detritus and macrophyte consumption increases during periods of reduced planktonic abundance or when fish move to littoral zones for breeding activities (Jul-Larsen et al., 2003). These feeding pattern variations have significant implications for understanding population dynamics, growth rates, and carrying capacity in different aquatic systems.
The reproductive biology of O. niloticus shows considerable variation across geographic regions and environmental gradients, with studies demonstrating that factors such as water temperature, nutrient availability, and population density significantly influence spawning timing, fecundity, and reproductive success (Trewavas & British Museum [Natural History], 1983). Research conducted in Ethiopian lakes has revealed species-specific adaptations to local conditions, with populations in different water bodies exhibiting distinct patterns of sexual maturation, breeding seasonality, and reproductive output (Wakjira & Getahun, 2017). However, comparative studies across Ethiopian lake systems remain limited, particularly regarding the integration of reproductive and feeding ecology data that would provide comprehensive understanding of population dynamics and ecosystem functioning.
Lake Tinishu Abaya, located in the Central Ethiopian, represents a relatively understudied freshwater ecosystem that supports diverse fish communities including O. niloticus as a dominant species (Enawgaw & Lemma, 2018). Previous research in Lake Tinishu Abaya has provided preliminary insights into O. niloticus biology, with Asaminew et al. (2011) documenting basic growth parameters and reproductive patterns, while Enawgaw & Lemma (2018) conducted a seasonal analysis of diet composition covering only three dry and three wet months of observation.
Review of existing literature shows several significant knowledge gaps that limit our understanding of O. niloticus ecology in Lake Tinishu Abaya and hinder the development of evidence-based management strategies. First, comprehensive annual data on reproductive parameters including length at first sexual maturity, fecundity, and detailed breeding seasonality remain unavailable, despite their fundamental importance for establishing minimum harvest sizes and closed season regulations (King, 2013). Second, the limited temporal scope of previous dietary studies (Enawgaw & Lemma, 2018) provides insufficient understanding of seasonal feeding pattern variations and ontogenetic dietary shifts that are crucial for assessing habitat requirements and ecosystem carrying capacity (Hart, 1997).
Furthermore, the morphometric characteristics and condition factor dynamics of O. niloticus in Lake Tinishu Abaya remain poorly documented, despite their importance as indicators of population health, growth performance, and environmental stress. The lack of comprehensive morphometric data from Lake Tinishu Abaya represents a significant limitation for comparative studies and regional fisheries management planning. Given these knowledge gaps and the increasing pressure on Ethiopian inland fisheries from population growth, agricultural intensification, and climate variability, there is an urgent need for studies that provide detailed understanding of key species biology in understudied water bodies (Reyntjens & Wudneh, 1998). Such research is essential for developing sustainable management strategies that can maintain fisheries productivity while preserving ecosystem integrity and supporting local livelihoods that depend on aquatic resources (Jul-Larsen et al., 2003).
Therefore, this study aims to examine the reproductive and feeding biology of O. niloticus in Lake Tinishu Abaya through systematic monthly sampling over a complete annual cycle. The specific objectives were: (i) determining length-weight relationships and condition factor dynamics to assess population health and growth patterns; (ii) analyzing reproductive parameters including sex ratios, length at first sexual maturity, absolute fecundity, breeding seasonality, and gonadosomatic index variations; and (iii) characterizing diet composition, ontogenetic dietary shifts, and seasonal feeding pattern variations through detailed stomach content analysis. This comprehensive approach will generate essential baseline data for Lake Tinishu Abaya while contributing to broader understanding of O. niloticus ecology in Ethiopian highland aquatic systems.
Materials and Methods
Lake Tinishu Abaya is located approximately 160 km southwest of Addis Ababa, Ethiopia, in the Siltie Zone of Central Ethiopia. Situated about 15 km east of Kebet, it lies at an elevation of 1,835 meters above sea level, with coordinates 7.48°N and 38.05°E and covering an area of 12.53 km2. It is a shallow freshwater system with a maximum depth of 3.7 m and a mean depth of 2.9 m. The lake is oval in shape and is fed by two perennial rivers: Dacha in the north and Boboda in the south. River Badober serves as the lake’s outlet. It hosts various fish species, including O. niloticus, Tilapia zillii, and Labeobarbus intermidius species (Fig. 1; Enawgaw & Lemma, 2018).
The specimens for this study were collected monthly from February 2023 to January 2024 using various sizes of gillnets (3, 4, 6, 8, and 10 cm mesh). The gillnets were deployed at 5:00 PM and retrieved the following morning at 7:00 AM. Immediately after collection, samples were transported to the Hawassa University laboratory in an icebox. In the laboratory, total weight and length were measured to the nearest 0.1 g and 0.1 cm using a balance and measuring board, respectively. Following morphometric measurements, specimens were dissected with a surgical blade to extract gonads and stomachs for reproductive biology and feeding habit analyses. The gonads and stomachs were preserved in a 5% formaldehyde solution until further analysis.
The length-weight relationship was determined using the equation proposed by Panase & Mengumphan (2015):
Where W is the body weight of the fish (g), L is the total length of the fish (cm), and a is the scaling constant determined empirically b = growth coefficient/ or pattern. If the value of b is equal to 3, the growth is considered isometric. If b is not equal to 3, the growth is allometric, which can be either positive (b > 3) or negative (b < 3).
According to from the b value of this length weight relationship result, the well-being of the fish species was assessed using the relative condition factor (Kn), as described by Le Cren (1951). The interpretations of the the fish species health condition depend on the exponent (b) value generated from the length-weight relationship. Fulton’s condition is considered for isometric growth (b = 3), whereas, for non-isometric growth (b ≠ 3), and statistically significant difference from 3, the relative condition factor is considered. Then the relative condition factor (Kn) was calculated using the formula:
Where Kn-relative condition factor, W is weight of the fish (g), L is total length of the fish (cm), a and b are the intercept and slope, respectively, derived from the logarithmic length-weight relationship. The condition factor provides insights into the general fitness and nutritional status of the fish species within their habitat. Higher values of the condition factor indicate better overall health and well-being of the fish.
The sex of each considered species was identified using Holden & Raitt (1974) keys. After catagorized the sex of O. niloticus in each sampling month and size clasess, the sex ratio was calculated using Chisquare test (χ2) (Vazzoler, 1996) as shown in the Equation (3):
For identifying length at first maturity (Lm50), first the numbers of mature (III−V) and immature (I and II) O. niloticus were identified after modal class (mid-length) grouping into 2 size classes. The average length at which 50% (Lm50) of the fish had mature gonads stage (III−V) was estimated for the fish using a logistic relationship established between the proportion of mature fish per length class and fish length (Holden & Raitt, 1974) as follows:
Where, PM is the proportion of mature fish in the length groups and L is the length groups (cm), and α and β are the intercept and the slope of the relationship parameter estimates for the above relationship were obtained by fitting a logistic regression procedure as shown below:
Where, Mn is number of matured fish in each size class and Tn is the total number of fish in each size class. The average length at which 50% (Lm50) of the fish possessed mature gonads was estimated by dividing the intercept (α) by the slope (β) of the above relationship and grouped in to 2 cm size class.
For evaluating fecundity, only rip female fish were considered. The total number of eggs per ovary was determined by counting that were preserved in plastic veal. The longitudinally preserved samples of each ovary were gently shaken and washed with tap water continuously to remove the ovarian membrane. The relationship between fecundity and total weight as well as total length was determined using the following formula developed by Crim & Glebe (1990).
Where F is fecundity, TL is total length and TW is total weight, a indicates constant and b indicates exponent.
The breeding season was determined from the seasonal catch of fish with ripe gonads (stage IV) in each month of the year. The frequency of males and females of O. niloticus with ripe ovaries was plotted against the sampling month in percentage to show their peak and non-peak breeding seasons (Admassu, 1996).
The gonad somatic index (GSI) was calculated as the ratio of gonad mass to total body mass and the seasonal variation in the GSI was used to define the peak spawning periods, as indicated in (Oso et al., 2013).
Where GW is gonad weight and TW is total weight.
After the morphometric measurements, the fish were dissected by a dissection kit. Following this, the full stomachs of O. niloticus individuals were isolated and pressed to extract the stomach contents. Furthermore, the stomachs were dissected the stomach contents were removed and preserved in a 5% formalin solution for more study. Finally, all specimens were labeled (date of sampling, length and weight measurements, location of sampling, fish species, etc.) and transported to Aquatic Science, Fishery, and Aquaculture laboratory at Hawassa University for additional analysis.
During laboratory analysis, stomach contents were transferred from plastic veal transferred to a known volume of a graduated cylinder test tube. Next to this overall volume was known and the samples were shaken to homogenize the stomach contents. And then larger specimens were identified using a dissecting microscope (LEICA MS), and smaller prey items were identified using a compound microscope (LEICA DME) with a magnification power of 100X. Finally the results were interpreted based on the frequency of occurrences, volumetric contributions, and index of preponderances. As described by Hyslop (1980), Food items were categorized to the lowest taxonomic level using keys and literature. In the frequency of occurrence method (%Fi), the number of specific preys per non-empty stomach was counted to understand how much it occurred in the total non-empty sample. The occurrences of each prey were expressed as a percentage and is calculated using the following formula:
Where Fi is frequency of occurrence of specific prey from non-empty O. niloticus stomach, Ti total number of non-empty O. niloticus considered for feeding habit analysis and Si total number of O. niloticus stomach with specific prey item. In the volumetric analysis, food items that were found in the stomachs were sorted out into different taxonomic categories. The water displaced by a group of items in each category was measured in partially filled graduate cylinder and expressed as a percentage of total volume of the stomach contents.
where Vi is the volume contributhion of of prey item i in percentage, i, Vn is the volume of a specific prey item found in all specimens, i and Tn is the volume of all food items found in all non-empty stomachs. The index of food preponderance (Ipi), which estimates the importance of each food item was calculated using the frequency of occurrence (%Fi) and volumetric contribution (%Vi) (Tomojiri et al., 2019). The calculation is as follows:
Where IPi is index of preponderance.
The diet compositions were once again grouped and categorized based on rain and dry months. Frequency of occurrences, volumetric contributions, and IP methods were used to compare the diet composition variations among seasons. To determine the size-based ontogenetic dietary shift, the samples were grouped into different size classes depending on their TL, and the percentage of volumetric contribution was calculated (Schoener, 1970). The volumetric contributions of major prey items were calculated in each size class, plotted with a stacked chart to indicate their contributions, and expressed as percentages. The four size classes considered for calculation were 12.0–14.9 cm, 15.0–17.9 cm, 18.0–20.9 cm, and > 21.0 cm TL.
Data were analyzed using Microsoft Excel and descriptive statistics. One-way analysis of variance (ANOVA) assessed monthly variations in the breeding season andGSI. The relationship between F and TL and TW was analyzed using regression. Lm50 was calculated via logistic function based on mature fish percentages in size classes. The Chi-square test determined the sex ratio, while descriptive statistics evaluated food item contributions. The IP was used to grade food items from stomach analyses.
Results
A total of 648 O. niloticus specimens from Lake Tinishu Abaya from February 2023 to January 2024, including 452 males and 196 females, were collected, resulting in an overall male-to-female ratio of 1:0.43. The results revealed significant deviations from the expected 1:1 ratio across different size classes and temporal periods (Tables 1 and 2). In the smallest size class (12.0–13.9 cm TL), females were more numerous than males (7 females versus 3 males), though this difference was not statistically significant (χ2 = 1.6, p > 0.05). However, in all larger size classes (≥ 14.0 cm TL), males significantly outnumbered females, with chi -square values ranging from 7.66 to 40.96 (p < 0.05 to p < 0.01). The overall sex ratio showed a highly significant deviation from unity (χ2 = 101.14, p < 0.001), indicating a pronounced male-biased population structure in Lake Tinishu Abaya.
Our temporal sex ratio analysis results revealed considerable monthly variation in the relative abundance of males and females (Table 2) where in the females slightly outnumber males (34 females versus 31 males) in February, though this difference was not statistically significant (χ2 = 0.12, p > 0.05). Similarly, in November, males were slightly more abundant than females, but the difference remained non-significant (χ2 = 3.63, p > 0.05). In contrast, all other months demonstrated statistically significant male dominance, with chi-square values ranging from 6.15 to 31.50 (p < 0.05 to p < 0.01). The most pronounced male bias occurred in September (χ2 = 31.50, p < 0.01), where males outnumbered females by a ratio of 1:0.14.
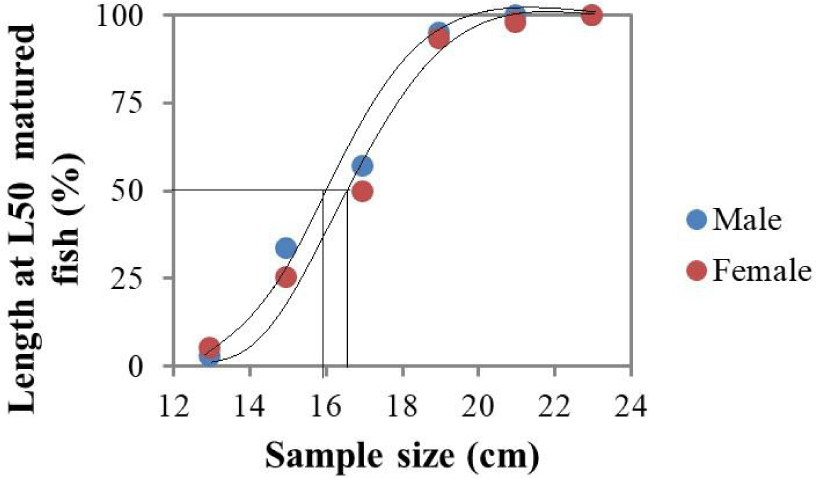
We conducted sexual maturity analysis based on macroscopic gonad examination showing that the smallest sexually mature male and female O. niloticus captured measured 12.5 cm TL (50.1 g TW) and 14.3 cm TL (65.6 g TW), respectively. As shown in Fig. 2, the logistic regression result of the proportion of mature individuals across different length classes yielded length-at-first-maturity (Lm50) values of 15.9 cm TL for males and 16.7 cm TL for females. These results demonstrate that male O. niloticus in Lake Tinishu Abaya attain sexual maturity at smaller sizes than females, with an approximate 0.8 cm difference in Lm50 values. The logistic curves fitted to the maturity data showed strong goodness-of-fit, indicating reliable estimates of the length at which 50% of individuals reach sexual maturity for both sexes.
F analysis was conducted on 41 gravid female O. niloticus specimens ranging from 13.8 to 22.6 cm TL and weighing between 51.0 and 197.4 g TW. Absolute F ranged from 110 to 493 eggs, with a mean of 353.63 ± 97.37 eggs per female. Regression analysis revealed a strong positive relationship between F and both TL and TW. The relationship between F and TL exhibited a curvilinear pattern with a correlation coefficient of R2 = 0.90, while the relationship with TW was linear with R2 = 0.8771. Statistical analysis indicated no significant difference between the strength of correlation for F with TL versus TW (p > 0.05), suggesting that both parameters are equally reliable predictors of reproductive potential in this population.
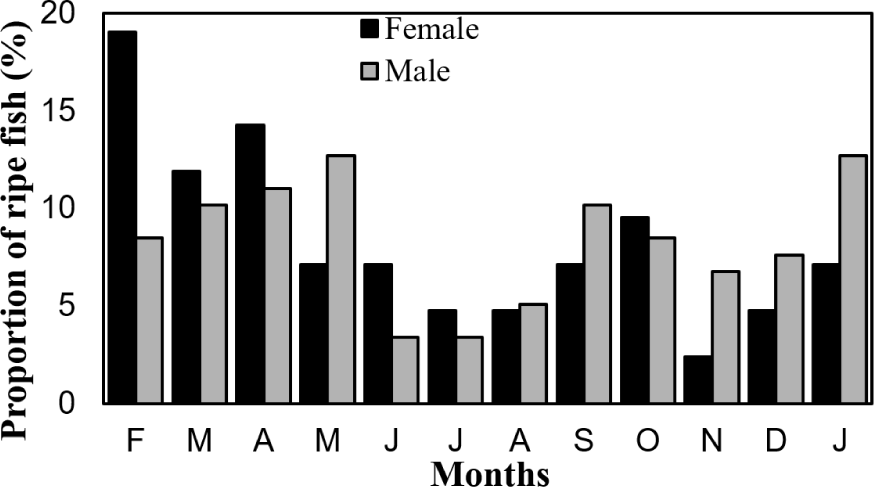
Temporal analysis of gonadal development revealed that O. niloticus in Lake Tinishu Abaya exhibits year-round reproductive activity, with distinct seasonal peaks in breeding intensity (Fig. 3). Fish with ripe gonads were captured throughout the entire sampling period, indicating continuous spawning activity. However, the proportion of individuals with ripe gonads varied significantly among months, with relatively high frequencies observed from January to May for both sexes. The peak reproductive activity occurred in January for males and February for females, when the highest proportions of ripe individuals were recorded. Conversely, the lowest reproductive activity was observed during October and November, when minimal numbers of fish with ripe gonads were captured. The temporal pattern of gonadal development was remarkably similar between males and females, suggesting synchronized reproductive cycles within the population.
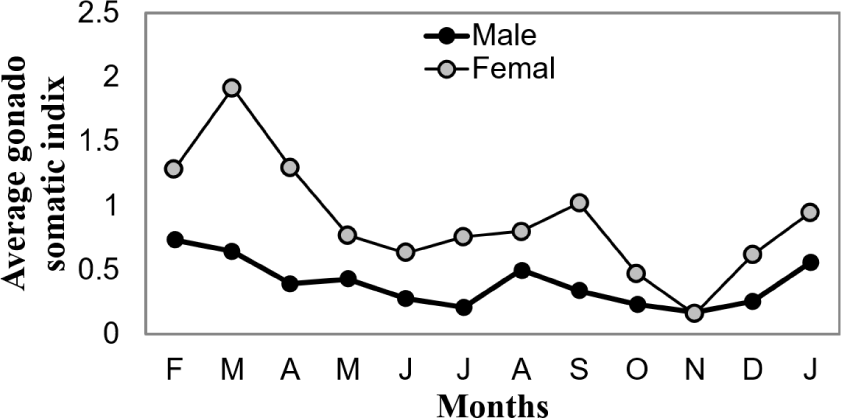
Monthly variations in the gonadosomatic index provided quantitative confirmation of the seasonal reproductive patterns observed through gonadal staging (Fig. 4). For males, GSI values ranged from 0.17 ± 0.26 in November to 0.73 ± 0.4 in February, while females exhibited a broader range from 0.16 ± 0.27 in November to 1.91 ± 3.0 in March. The highest GSI values, indicating peak reproductive condition, were recorded in February for males and March for females, closely corresponding to the periods of maximum ripe gonad frequency. The lowest GSI values for both sexes occurred in November, confirming this as the period of minimal reproductive activity. Statistical analysis revealed significant monthly variation in GSI values for both sexes (p < 0.05), with females showing greater variability in reproductive investment than males, as evidenced by the higher standard deviations and maximum GSI values.
The GSI data show clear seasonal variations, and understanding how TL and linear metre (Lm) relate to the GSI changes is crucial for understanding the reproductive strategy, identifying spawning stocks, fisheries management, and ecological insights. That is, a positive correlation between TL and GSI would suggest that larger individuals tend to have larger gonads, which is often the case as fish allocate more energy to reproduction as they grow. However, the rate at which GSI increases with TL can vary and might indicate different reproductive strategies. Similarly, Lm is inherently linked to the onset of significant GSI increases.
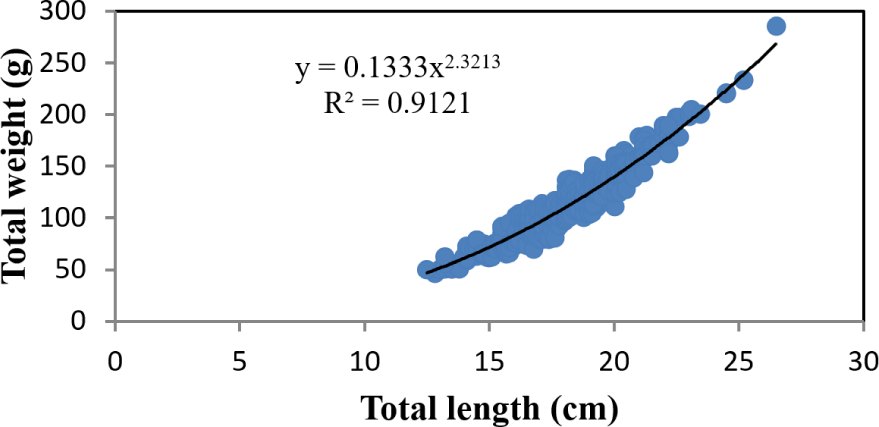
The length-weight relationship analysis encompassed the entire size range of captured specimens, with TLs ranging from 12.5 to 26.5 cm and corresponding weights from 46 to 285 g. Regression analysis revealed a curvilinear relationship described by the equation W = 0.1333 L^2.3213 (R2 = 0.9121), where W represents TW in grams and L represents TL in centimeters (Fig. 5). The allometric coefficient (b = 2.3213) was significantly less than 3 (p < 0.05), indicating negative allometric growth, whereby fish become relatively thinner as they increase in length. This growth pattern was consistent across both sexes, suggesting that O. niloticus in Lake Tinishu Abaya exhibits a characteristic elongation of body form with increasing size rather than proportional increases in all body dimensions.
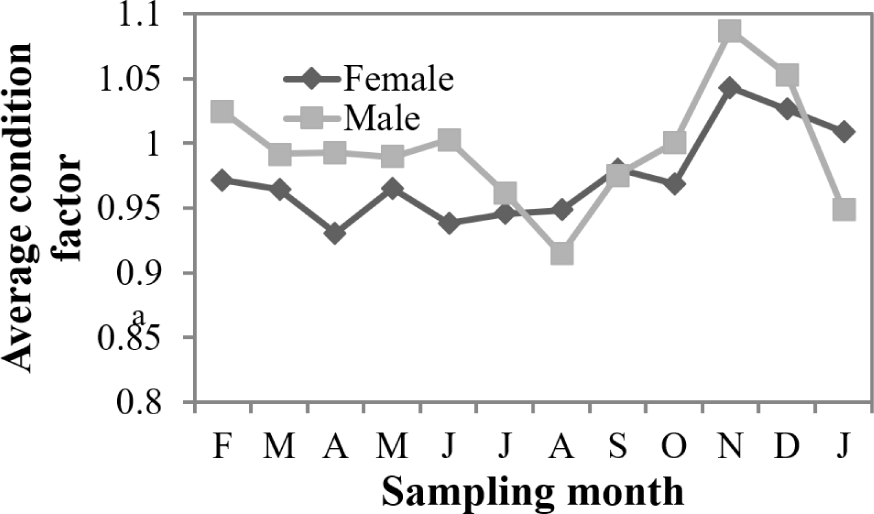
Analysis of the relative condition factor (RCF) revealed temporal variations in fish condition throughout the sampling period (Fig. 6). Monthly RCF values for males ranged from 0.91 ± 0.06 in August to 1.09 ± 0.08 in November, while females exhibited values from 0.93 ± 0.07 in April to 1.04 ± 0.08 in November. The annual mean RCF was 1.00 ± 0.08 for males and 0.97 ± 0.07 for females, with no statistically significant differences observed between sexes (p > 0.05). These values indicate that both sexes maintained relatively stable body condition throughout the year, with slight improvements during November corresponding to the post-reproductive period when energy allocation shifts from gonadal development to somatic growth and condition restoration.
Stomach content analysis was conducted on 648 O. niloticus specimens, of which 575 (88.7%) contained food material while 73 (11.3%) had empty stomachs. The specimens analyzed for dietary composition ranged from 12.5 to 26.5 cm TL and weighed between 46 and 285 g TW. Microscopic examination of stomach contents revealed nine major food categories: phytoplankton, zooplankton, detritus, macrophytes, insects, fish parts, fish eggs, ostracods, and nematodes (Table 3). Phytoplankton emerged as the dominant dietary component, occurring in 574 (99.8%) of the examined stomachs and constituting 58.41% of the total food volume. The phytoplankton assemblage was primarily composed of blue-green algae (26.93% by volume), diatoms (15.56%), green algae (13.55%), and euglenoids (2.38%).
Detritus represented the second most important food category, occurring in 571 (99.3%) of stomachs and accounting for 18.63% of the total volume. Macrophytes were present in 449 (78.09%) of the examined stomachs, contributing 19.09% of the total food volume. Zooplankton, despite occurring in 233 (40.5%) of the stomachs, contributed only 2.55% of the total food volume, with rotifers being the most abundant component (1.85% by volume) followed by copepods (0.69%). Insects were found in 235 (40.87%) of the stomachs but represented only 0.90% of the total volume, with Diptera being the most frequently encountered order. The IP analysis confirmed phytoplankton as the most important dietary component (38.78% IP), followed by detritus (12.31% IP) and macrophytes (9.91% IP). All other food categories exhibited IP values below 1%, indicating their relatively minor contribution to the overall diet.
Generally, there are roles of fishes as plankton, detritus, and macrophyte feeders, and they have their correlation to lake ecosystems. The fish as plankton feeders (the crucial role of phytoplankton as the primary producer and a fundamental nutritional source for fish, directly supporting the conclusion that the fish is a plankton feeder) and the fish as detritus feeders (has the importance of detritivory in freshwater fish, has its role in energy flow and nutrient cycling, and reinforces the importance of detritus feeders in aquatic environments).
Analysis of dietary composition across four size classes (12.0–14.9, 15.0–17.9, 18.0–20.9, and ≥ 21.0 cm TL) revealed significant ontogenetic shifts in feeding preferences (Fig. 7). In the smaller size classes (12.0–20.9 cm TL), phytoplankton consistently dominated the diet, with volumetric contributions ranging from 55.04% to 60.31%. For the fishes in the size classes of 12.0–14.9 cm TL, 15.0–17.9 cm TL, and 18.0–20.9 cm TL, detritus makes up a regular and reliable part of their diet, ranging from 20.71% to 25.01%. However, in the largest size class (≥ 21.0 cm TL), macrophytes became the main food type, providing 50.4% of the total volume, while phytoplankton contributed only 16.4%.
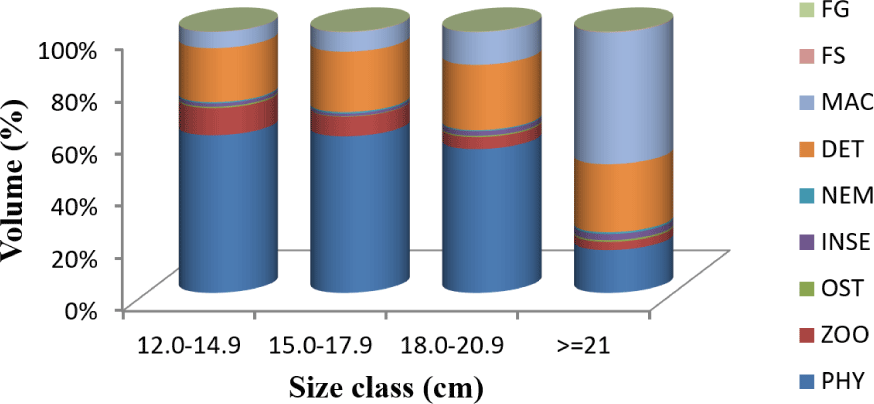
Zooplankton showed a clear decreasing trend with increasing fish size of classes from 12.0 TL to 20.9 cm TL, declining from 7.54% in the smallest size class to 3.15% in the largest class. Conversely, detritus consumption increased slightly with size, reaching 26.04% in the largest fish. Minor food categories (insects, ostracods, nematodes, and fish scales) consistently contributed less than 3% of the total diet volume across all size classes, indicating their supplementary role in the feeding ecology of O. niloticus. This ontogenetic shift in diet reflects the species’ adaptive feeding strategy and morphological constraints related to mouth gape size and digestive capacity. Specifically, smaller fish (within the 12.0–14.9 cm TL and 15.0–17.9 cm TL size classes) exhibit a phytoplankton-dominated diet, which then transitions to a macrophyte-dominated diet in larger fish (those in the 18.0–20.9 cm TL and ≥ 21.0 cm TL size classes).
Seasonal analysis of dietary composition was conducted on 575 specimens with stomach contents, comprising 295 individuals from the dry season (October to March) and 280 from the wet season (April to September) (Table 4). The primary food categories (phytoplankton, detritus, and macrophytes) were present in substantial quantities during both seasons, though their relative importance varied significantly. Phytoplankton maintained the highest frequency of occurrence in both seasons (99.1% in dry season, 96.64% in wet season) but showed notable seasonal variation in volumetric contribution. During the dry season, phytoplankton constituted 60.09% of the total food volume compared to 48.18% in the wet season. The IP confirmed phytoplankton as the most important dietary component in both seasons, with values of 39.63% in the dry season and 34.73% in the wet season.
Detritus and macrophytes exhibited opposite seasonal patterns, with both showing increased importance during the wet season. Detritus increased from 17.91% (dry season) to 22.70% (wet season) by volume, while macrophytes increased from 18.34% to 23.30%. The IP values for these components were correspondingly higher in the wet season (detritus: 16.25%, macrophytes: 13.61%) compared to the dry season (detritus: 11.81%, macrophytes: 9.18%). Zooplankton, insects, and other minor food categories maintained relatively consistent contributions across seasons, with slight increases during the wet season. However, their overall contribution remained minimal, with IP values below 1% for most categories. The combined contribution of phytoplankton, detritus, and macrophytes accounted for 94.18% of the diet in the wet season and 97.15% in the dry season, confirming their status as the primary food sources for O. niloticus in Lake Tinishu Abaya throughout the year.
Discussion
In this study, the length-weight relationship of O. niloticus exhibited a curvilinear and significant association, with a length-weight coefficient (b) value of 2.3213 in Lake Tinishu Abaya. This indicates a negative allometric growth pattern, suggesting a decrease in body condition or elongation in body form as length increases. This result similar finding with previous studies (Kebede et al., 2018). However, these findings differ from prior research on O. niloticus conducted by Enawgaw & Lemma (2018) in the same lake with similar species. This is probably due to result from sample size, food availability, gonad development stage, and spawning period (Bagenal & Tesch, 1978).
The current study showed, O. niloticus had average RCF values of 1.00 ± 0.07 for males and 0.97 ± 0.08 for females. These figures are notably lower than those reported in previous studies, which ranged from 1.43 to 1.81(Assaminew et al., 2011). This is due to habitat quality, prey availability, water temperature fluctuations, and the stage of gonad development (Abujam & Biswas, 2016).
The sex ratios of O. niloticus captured in Lake Tinishu Abaya differed significantly from the expected 1:1 sex ratio (χ2 = 101.14, p value < 0.05). The male O. niloticus were more numerous than females at a ratio of 1:0.43. The imbalanced male-to-female ratio of 1:0.43 in Lake Tinishu Abaya may be due to several ecosystem factors in fish populations, like: temperature, food availability and diet, predation, fishing pressure and selective harvesting, habitat degradation and water quality, reproductive biology and life history, and stocking practices (Anteneh et al., 2023). Similar founding was reported by scholars Asaminew et al. (2011) and Enawgaw & Lemma (2018) in the same lake with similar species. Even if further specific research would be needed, the imbalanced male-to-female ratio of 1:0.43 in the Lake Tinishu Abaya may be due to several ecosystem factors in fish populations, like: temperature (temperature-dependent sex determination and sex-specific mortality due to temperature stress), food availability and diet (nutritional stress and dietary shifts), predation (sex-selective predation), fishing pressure and selective harvesting (size-selective fishing and sex-specific vulnerability), habitat degradation and water quality (pollution/contaminants and habitat loss/degradation), reproductive biology and life history (differential longevity and sex change), and stocking practices (Anteneh et al., 2023). Understanding these factors is paramount to developing effective and sustainable management strategies to restore a balanced sex ratio and ensure the long-term viability of the O. niloticus fishery in Lake Tinishu Abaya. Without intervention, this significant sex imbalance could lead to a substantial decline in fish populations, impacting both the ecosystem and the communities reliant upon it. This result will be due to the reason the earlier sexual maturity of females and the faster growth rate of males, leading to males quickly growing out of this size bracket while females remain within it as they mature and reproduce. Environmental factors like temperature during early development could also play a significant role (Asaminew et al., 2011).
The higher number of females than males in O. niloticus in February is the result of seasonal breeding patterns and spawning aggregations. Since February is a period of high reproductive activity for females, their increased numbers in the sample could reflect their aggregation for spawning. This is supported by studies that show seasonal variations in sex ratios linked to breeding cycles (Shishitu et al., 2021). Similarly, in November, males were slightly more abundant than females, but the difference remained non-significant (χ2 = 3.63, p > 0.05). This may be due to the reason that intrinsic factors (genetic predisposition, differential growth/maturity, sex-specific mortality, life history) (Wootton & Smith, 2014) and lake ecosystem factors (temperature, food availability and quality and predation pressure) (Ospina-Álvarez & Piferrer, 2008) are involved.
In Lake Tinishu Abaya, the length at Lm50 of sexual maturity was found to be 15.9 cm for males and 16.7 cm TL for females of O. niloticus. This is similar to the findings of Kebede et al. (2018) in Lake Langano (17.4 cm TL for females and 17.2 cm TL for males). However, the present results showed lower values than those found in other locations such as Ribb reservoirs (24.0 cm for females and 25.4 cm for males) (Tesfahun & Alebachew, 2023). This suggests that O. niloticus in Lake Tinishu Abaya tends to mature at an early age, possibly due to stress and variations in habitat that affect food availability.
The total F range of O. niloticus observed in the study area was between 110 and 493, with a mean of 353.63 ± 97.37 eggs. Comparable findings were reported by researchers Gómez-Márquez et al. (2003) (104 to 373) and Abarike & Ampofo-Yeboah (2016) (104 to 373). The F of O. niloticus in Lake Tinishu Abaya showed a curvilinear relationship with TL and a linear relationship with TW. In many tropical freshwater fish species, F increased in proportion to the 2.81–3.96 power of TL (Lowe-McConnell, 1975). Additionally, there was a strong positive correlation between F and the length of O. niloticus (R2 = 90). This result is consistent with findings by Gómez-Márquez et al. (2003), who showed a linear relationship between F and weight (b = 1) for the O. niloticus study. This low F of O. niloticus observed in Lake Tinishu Abaya may be associated with various environmental pollutants resulting from anthropogenic factors, habitat variations, low-quality food resources, water pollution, agricultural waste, and pesticides (Tesfahun & Alebachew, 2023).
The main breeding season of O. niloticus in Lake Tinishu Abaya for both sexes occurs between January and May. Similar results were reported by Asaminew et al. (2011) in the same lake and Kebede et al. (2018) in Lake Langano. Accordingly, the main breeding season of O. niloticus in Lake Tinishu Abaya might be triggered by the lake ecosystems. The monthly variation of the gonadosomatic index (GSI) for O. niloticus revealed that the lowest GSI for both sexes was observed in November. But the highest average GSI was observed in February for males and March for females, which could be taken as the peak breeding season of O. niloticus. The findings of the current study align closely with the findings of Asaminew et al. (2011) in the same lake and Kebede et al. (2018) in Langano. Examining the relationship between GSI and length across different size classes helps validate the estimated Lm and provides insights into the size at which individuals contribute to reproduction (Diana & Höök, 2023; King, 2013).
The diet of O. niloticus consisted primarily of phytoplankton, detritus, and macrophytes, with phytoplankton being the most consumed food item. This finding is consistent with studies by Enawgaw & Lemma (2018), indicating the high availability of phytoplankton in tropical lakes. The consumption of macrophytes and detritus was also significant, reflecting the diverse feeding habits of this species. Variations in dietary composition between lakes are influenced by numerous factors, including seasonality, spatial variations, and ontogenetic dietary shifts (Bozza & Hahn, 2010).
Additionally, the trophic roles of fish in freshwater ecosystems demonstrate how fish (particularly planktivorous fish) can exert top-down control on lower trophic levels (zooplankton and phytoplankton), directly impacting lake productivity and water quality. That is top-down (fish predation) and bottom-up (nutrient availability) controls on plankton communities (Amadeu Santana et al., 2015). Phytoplankton is the dominant dietary component in the examined stomachs, and this is because of its high occurrence and highest volume contribution, and it also makes up the largest bulk of the diet compared to other food items. This significant volumetric contribution makes it the dominant dietary component in this study (Lobus & Kulikovskiy, 2023). As O. niloticus matures, there is a notable shift in dietary preferences. Younger fish primarily consume animal-origin foods, while larger fish increasingly include macrophytes and detritus in their diet. This change is attributed to the wider mouth gapes and enhanced digestive capabilities of adult fish, allowing them to process more complex food items (Temesgen et al., 2022). Several studies have reported similar feeding variations in different size groups of O. niloticus in various water bodies (Engdaw, 2023; Tesfahun & Alebachew, 2023).
Seasonal variations in diet composition were evident, with phytoplankton being predominant during the dry season (October to March), likely due to increased turbidity from flooding and nutrient influx. In contrast, during the wet season (April to September), macrophytes and detritus became more important, as fish moved to shallower waters for breeding and feeding. This shift in available food resources aligns with findings from Enawgaw & Lemma (2018), suggesting that the fish’s diet adapts to seasonal changes in habitat and food availability.
This study faced several limitations that may have influenced the results. Data collection was constrained by seasonal variations, which could impact the assessment of reproductive and feeding biology. The local community’s lack of awareness regarding fishery production hindered cooperation and access to certain areas of the lake. Additionally, the transport of fish samples was challenging due to inadequate facilities, potentially affecting sample integrity. The anthropogenic factors on the lake are households, businesses, community wastes or domestic waste, detergents, fertilizers, food scraps, rapid and excessive growth of algae, plastic bags, bottles, wrappers, fishing lines, and other debris directly dumped into lakes or carried by wind/rain. Furthermore, the absence of professional fishery experts limited the depth of analysis and the implementation of best practices in sampling and data collection. Future studies should focus on a broader temporal scope to capture seasonal variations more comprehensively. Investigating the impact of anthropogenic factors on fish health and habitat quality would provide valuable insights. Engaging local communities through education and awareness programs about sustainable fishery practices could enhance cooperation and data collection efforts. Additionally, establishing partnerships with professional fisheries experts would improve methodological rigor and data accuracy. Future research should also explore the socio-economic factors influencing local fishing practices and their impact on fish populations, ultimately contributing to more sustainable management strategies for Lake Tinishu Abaya.
Conclusion
The study found that male O. niloticus were more abundant than females in Lake Tinishu Abaya, with year-round breeding peaking during the rainy season, influenced by food availability. The species reached sexual maturity earlier and exhibited lower F compared to similar species in other waters. Their diet included phytoplankton, detritus, macrophytes, insects, and zooplankton, with dietary composition varying by size and season. As fish size increased, the importance of macrophytes and detritus grew, while phytoplankton and zooplankton decreased. Monthly fluctuations impacted food availability, indicating a potentially stressful environment due to natural or human factors. Therefore, it is recommended to stop harvesting of O. niloticus below 16.4 cm and further study sex ratio assesment. Additionally, it is also advised to close fisheries during the peak breeding season (between January and May). Phytoplankton and detritus are a dominant prey and it is recommended to construct buffer zone to maintain the fisheries resource from the Lake.