Background
Nigeria is endowed with a large hectare (14 million) of land consisting of reservoirs, lakes and rivers, capable of producing over 980,000 metric tonnes of fish annually (Federal Department of Fisheries (FDF) 2007). Statistical surveys have shown that the demand for fish in the country exceeds supply due to ever-increasing human population (currently over 150 million) (Tsadu et al. 2006; National Population Commission of Nigeria (NPC) 2006).
African pike, Hepsetus odoe, (Bloch 1794), commonly referred to as ‘Ẹgá’ in Yoruba-land, Nigeria, belongs to the family Hepsetidae and is a diurnal, pelagic, oviparous predatory and piscivorous species inhabiting highly vegetated backwater, large floodplain, lagoon and swamp systems, but has been reported absent in the River Nile (Idodo-Umeh 2003; Oso et al. 2011; Ezekiel and Abowei 2014). Both male and female species mature at 140 and 160 mm, respectively (Merron et al. 1990). They constitute the major catches and landings of fishermen, and they are biologically and economically important in Nigerian and other West African coastal waters (from Senegal to Botswana) because of their roles in pelagic food chain and diet component of livestock and humans (Winemiller and Kelso-Winemiller 1994; Akponine and Ayoade 2012; Ezekiel and Abowei 2014; Kareem et al. 2015).
Fish from inland waters such as Hepsetus odoe is known to be a highly demanding, valuable and important source of rich protein to man and also contributes immensely to the nation’s gross domestic product (GDP) (Bolarinwa 2015). In addition, it generates income and employment for the vast majority of the populace in terms of fishing, fish-smoking, processing etc., and hence serves as a source of livelihood in tropical regions (Adedokun and Fawole 2012). Hepsetus odoe bears a striking resemblance to the European pike. They are torpedo-shaped, predatory fish. The body form is elongate with a pronounced snout. The dorsal surface is usually dark brown or green while the ventral surface is silvery. The face is often light green or brown with prominent dark brown or black stripes radiating from the eye. The coloration of the adults is relatively uniform, but colouration of young is extremely variable (Jubb and Manning 1961). The scales of the African pike are rough to the touch and number 49–58 along its lateral line. The most striking feature of H. odoe is their dentition. Both upper and lower jaws are filled with sharp pointed teeth, but the lower jaw has two rows while the upper only has one. There are also two large canines in each jaw. Two pairs of dermal flaps can be found on the upper and lower jaws (Barnard 1971). The folds on the upper jaw are smaller than those on the lower jaw. They were once thought to be sensory organs, but are now thought to be used in building nests for breeding (Merron et al. 1990). They are multiple spawners, breed over the summer months and are relatively short-lived, with a life span of only 4–5 years (Skelton 1993). They build a free-floating bubble-nest which suspends the eggs to receive enough aeration, acts as a deterrent to predators due to its placement in highly vegetated areas and provides an anchor for the young to attach themselves to. Hepsetus odoe seems to prefer the upper courses of small rivers where Hydrocynus spp. (their main predators) are absent or less abundant (Entsua-Mensah et al. 2010). The onset of the spawning season varies depending upon the region, the method is constant across Hepsetus odoe populations. Spawning season usually begins with rainy season when flood and water level have risen (Merron and Bruton 1988; Merron et al. 1990). It is suspected that flood waters may actually stimulate the maturity of gonads of H. odoe. However, water temperatures play a critical role as well, and it is not until August that the water temperatures are likely to reach optimal levels for the eggs and developing embryos. Females are repeated spawners and produce about 6440 eggs in a season. These eggs vary in size from 2.5–2.9 mm in diameter, and the adults are known to guide and show some parental cares for the eggs and hatched embryos (Merron et al. 1990).
Length-weight relationship (LWR) indicates the average weight of fish at a given length by making use of a mathematical equation to show relationships between the two (Beyer 1987). Fish can attain either isometric or allometric growth (Gayanilo and Pauly 1997; Sakar et al. 2013). Isometric growth indicates that both length and weight of the fish are increasing at the same rate. Allometric growth can be either positive or negative. Positive allometric implies that the fish becomes stouter or heavier or deeper-bodied as its length increases. Negative allometric implies the fish becomes slender or lighter as its length increases (Wootton 1998; Zafar et al. 2003). For stock assessment purpose, LWR is assumed to be uniform for an entire fish stock (Gerritsen and McGrath 2007). Knowledge of the relationship between the length and weight of a given species is useful to transform the length structures obtained into the weights of fish captured and to monitor health status of a population (Cone 1989; Ecoutin et al. 2005). Fluctuation in fish growth is a common phenomenon in tropical and subtropical waters because of environmental variations, spawning and dynamics of food composition (Adeyemi et al. 2009).
Condition factor (CF) is an estimation of general well-being of fish (Oribhabor et al. 2011) and is based on the hypothesis or assumption that heavier fish (at a given length) are in better condition than the lighter ones (Bagenal and Tesch 1978; Ogamba et al. 2014). The condition factor of 1.0 or greater indicates the good condition of fish while less than 1.0 shows bad condition (Abobi 2015). Condition factor is a useful index for estimating growth rate and age and for assessing environmental quality (Navarro et al. 2010; Olopade and Tarawallie 2014). The condition factor can be influenced by season, sex, type of food organism consumed by fish, age of fish, amount of fat reserved and environmental conditions (Bagenal and Tesch 1978; Anene 2005; Abowei 2009).
Reproduction is an important physiological system that is crucial in the life cycle of fish. Fecundity is the number of ripened or vitellogenic eggs or oocytes in the female prior to the next spawning period and varies intra- and interspecifically (Bagenal 1978; Wootton 1990; Akponine and Ayoade 2012). Lagler et al. (1977) stated that the number of eggs produced by fish in a spawning season is species-specific and also varies with genetic characteristics, size, age, environmental factors and physiological conditions of the fish. For instance, large female fish in better condition tend to exhibit higher fecundity than those in poor condition. This information is useful in estimating spawning stock biomass and management (Idowu 2017). The gonadosomatic index, (GSI), is described as gonad mass as a percentage of total body weight and is widely used as a simple measure of the extent of reproductive investment, gonadal development and maturity of fish in relation to spawning (Quyyam and Quasim 1961; Bagenal and Tesch 1978; Fagbenro et al. 1991; Cubillos and Claramunt 2009). Gonadosomatic index of fish increases with maturity and abruptly declines after spawning. GSI is particularly helpful in identifying season of spawning as the female gonads increase in size prior to spawning (Biswas 1993; Idowu 2017). Study on reproductive biology of fish is essential for conservation and selecting fish candidates of aquaculture from the wild (Muchlisin 2013).
Fish production in Nigeria comes from three main sources: artisanal (coastal and inland waters such as lakes), aquaculture and industrial fishing, but the majority of the supply comes from the artisanal sub-sector (of about 500, 000 metric tonnes per annum) (Adekoya and Miller 2004 as reported by Chilaka et al. 2014). Because fish demand (1.5 million metric tonnes) exceeds supply, local fish production has failed to meet the country’s domestic demand. Nigeria has the resource capacity to produce 2.4 million metric tonnes of fish every year, yet the country, as of 2007 (over a decade), is still a large importer of frozen fish annually from all sources (Fasakin 2008). Fish production is significant to the Nigerian economy (contributing over US$ 1billion to its gross domestic product) in view of its role in providing livelihoods (employment and income generations) and a very cheap source of proteineous foods (fish such as Hepsetus odoe) to a riparian community (such as the study area, Eleiyele) and over 3 million people inhabiting Ibadan metropolis (where the study area is located) (Food and Agricultural Organization (FAO) 2004). Nowadays, coastal and inland waters, especially oceans, lakes and reservoirs, have come under increasing threats of human activities such as industrial, municipal, agricultural and domestic pollutions which could alter environmental quality and have biological, ecological and societal implications (Muyodi et al. 2011). Most inland water-bodies in Nigeria are also overfished as a result of increasing rate of fishing efforts being employed for exploitation of aquatic resources (Faturoti 2000 as reported by Adedokun et al. 2017). In the same vein, fish resources are susceptible to induced environmental stress, which can deteriorate and affect their biological production (Chilaka et al. 2014). Olaniran (2003) had previously reported five dominating and mostly landed families in the lake system of Eleiyele: Cichlidae (Sarotherodon spp.), Centropomide, Hepsetidae (H. odoe), Gymnachidae and Bagridae.
Although, many studies have been conducted on some aspects of the ecology of fishes in Nigeria, there is a dearth of reports of this species (H. odoe) in Lake Eleiyele (Offem et al. 2009; Kareem et al. 2016), which necessitates this research. The objective of this study is to determine the length-weight relationship, condition factor, fecundity and gonadosomatic index of Hepsetus odoe in Lake Eleiyele, which would serve as important tools in describing the status of fish population, predicting the potentials of fisheries such as knowledge of recruitment pattern from fecundity and gonadosomatic index, selective exploitation, effective conservation and management of this species in the water-body (Abowei and Hart 2009; Arawomo 1982 cited in Idowu 2013; Kareem et al. 2016; Mireku et al. 2016).
The following are the research questions:
What kind of growth pattern did Hepsetus odoe exhibit in the lake system of Eleiyele Reservoir?
Were the investigated fish species in good condition in the ecosystem despite pollution from anthropogenic sources?
What reproductive indices, in terms of fecundity and gonadosomatic index, did the fish species exhibit in the aquatic ecosystem?
Methods
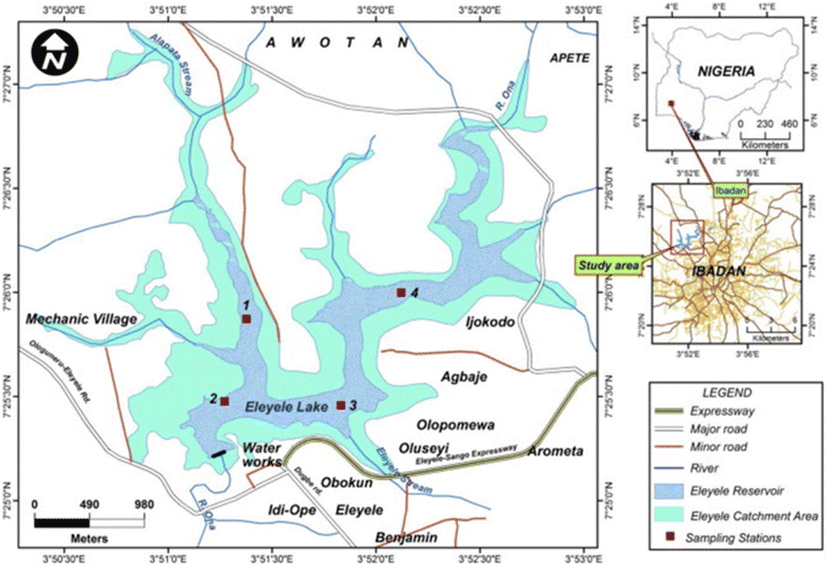
Eleiyele Lake is a freshwater ecosystem basin located in the northwest part of Ibadan and lies on the latitude 7° 25′ 0″ to 7° 27′ 0″ N and longitude 3° 50′ 30″ to 3° 53′ 0″ E (Fig. 1) and altitude 125 m above sea-level. It has an impoundment area of 5.46 km2, storage capacity of 704 million litres of water (now increased to 900 million), depth of 12 m (Imevbore 1965 cited in Akponine and Ayoade 2012) and receives water principally from the Ona river, part of the dense network of inland water courses that flow southwards into the Lagos Lagoon. A number of stream channels serve as tributaries to the Eleiyele wetland basin. Along the sides of the lake is a forest reserve with stretches of vegetation such as elephant grass, giant-star grass, trees, shrubs and spear grass. The lake serves as the major source of freshwater for town and domestic uses and fishery products: fin and shellfish. Along its banks or catchment areas, various domestic and industrial activities are carried out: dumping of refuse and sewage, agricultural activities such as crop farming, agro-processing, mechanic workshops, sand mining, water transportation/navigation, spiritual exercise such as water baptism and unregulated discharge of industrial wastes, which may lead to metal, polychlorinated biphenyl, PCB and hydrocarbon contaminations of the aquatic ecosystem and therefore can have detrimental effects on the biological components (Olaifa et al. 2004; Ajani and Balogun 2015; Olayinka et al. 2017).
Fresh specimens (90 samples in total based on financial resources available) of Hepsetus odoe were collected from fishermen at Eleiyele Lake from June to August 2010 as this period marks the end and beginning of gonad maturation and spawning respectively, and rainy season when the level of water rises (Merron et al. 1990; Idowu 2017). The fish were caught using gill nets (mesh sizes ranging from 3 to 8 cm) set overnight and were retrieved in the morning of the following day. They were identified to species and sex level using the fish catalogue of FAO (Fischer et al. 1981; Food and Agricultural Organization (FAO) 1990; Schneider 1990) and sex ratio (male to fecund female, 1:1.8) determined according to the method described by Oso et al. (2011). Biometrics, including standard length (STL), total length (TL) and body weight (BWt) were measured at the landing site using a calibrated plastic board and a sensitive weighing balance (Moruf and Lawal-Are 2015). All lengths and weights were measured in centimetres and grams respectively. Gonads were dissected, weighed and preserved in 5% formalin and Gilson’s fluid (Akponine and Ayoade 2012; Mireku et al. 2016). Gonad maturity (stage IV) was visually and microscopically observed and characterised by large ovaries, yellowish or milky-whitish in colour, visible opaque oocytes without bruised areas and occupying three quarter of the entire body cavity with blood capillaries (Bagenal 1978).
The raw data of total lengths (STL), total lengths (TL) and body weights (BWt) of the H. odoe collected were used to compute the length-weight relationship with the formula (Le Cren 1951; Ricker 1973; Hayes et al. 1995);
where W = body weight, L = total length, a = intercept on the length axis and b = slope or regression coefficient which usually ranges from 2 to 4.Equation (1) is log transformed to give a linear relationship (Le Cren 1951; Parson 1988);
When Log W is plotted against Log L, the regression coefficient or growth exponent, b, and intercept, a, are obtained. The value of exponent, b, provides biological information on the pattern of growth of fish (Abobi 2015). For the species, the compiled growth exponent (b) was compared to 3 using Student’s t test to ascertain whether species grew isometrically or not (Sokal and Rohlf 1987). This was achieved by using the formula and statistical link (Zar 1984; Morey et al. 2003);
http://www.socscistatistics.com/pvalues/tdistribution.aspxwhere ts = Student’s t test, b = slope and sb = standard error of the slope. The b value of 3 was chosen for comparison and a standard because it indicates that the fish grows isometrically.
This was computed for the species with Fulton’s equation (Ricker 1971; Pauly 1983);
where K = condition factor, W = body weight, L = total length and b = compiled growth exponent.The gravimetric method was used for estimating fecundity (ripe ovary). This involves subsampling of eggs by weighing from the gonad for fecundity estimation. Preserved eggs were washed with Gilson’s fluid and freshwater to drain excess preservative and, after removing any remaining ovarian tissue, were left on filter paper for about 5–10 min to dry. A subsection of the ovaries or gonads was weighed (1 g) and number of eggs in it was counted and recorded. The total number of eggs in the ovary was calculated as a proportion. Fecundity (F) is finally extrapolated using the formula: F = nxW where W = total weight of ovary in grams and n = number of eggs per 1 g of subsection ovary or gonad (Bagenal 1978; Sevaraj et al. 1972; Hunter and Goldberg 1980; Oso et al. 2011). This method of estimating fecundity has been reported accurate to about 1% (Simpson 1959). The linearity of the fecundity-weight and fecundity-length relationships was determined using the equation (Akponine and Ayoade 2012);
F = fecundity estimate, W = body weight (g), TL = total length (cm) and ‘a’ and ‘b’ are regression constants.
Gonadosomatic indices were calculated using the formula (Dadzie and Wangila 1980);
The data obtained from the morphometric and reproductive variables were subjected to statistical analysis using R version, 0.98.1083 (2009–2014) and Excel. Analysis of variance (ANOVA) was used to test whether the calculated regression line was significant (Ogbeibu 2005). All statistical analyses were considered at significant level of 5% (p < 0.05).
Results and discussion
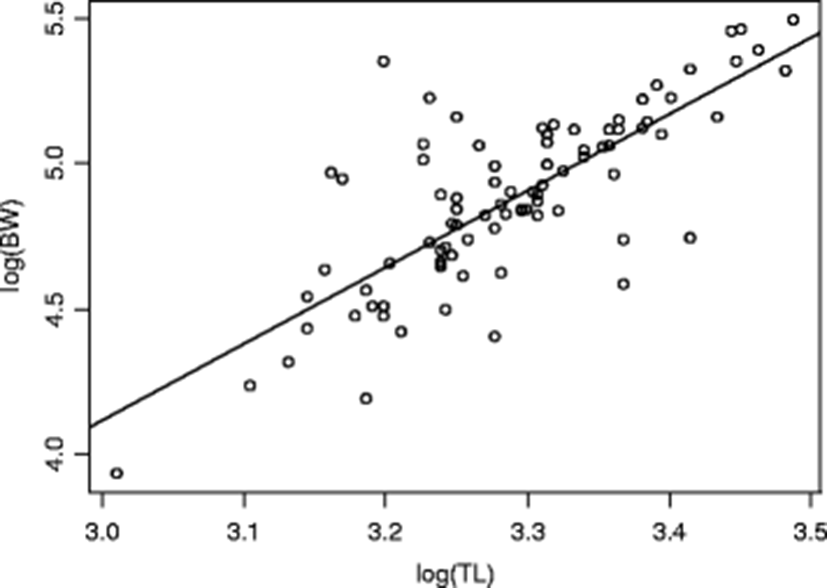
Growth is an important component of biological production. The LWR of Hepsetus odoe of 90 samples in this study was examined. The mean total length (TL) was 26.89 ± 2.47 cm, and the mean body weight (BW) was 137.11 ± 41.05 g. Table 1 shows the regression analysis. The relationship between the total length (TL) and body weight (BW) shows a highly significant positive correlation, R2 = 0.61 (p = 2 × 10− 16). This indicates that BW of the species increases as the TL increases (Ezekiel and Abowei 2014). Gomez and Gomez (1984) reported that high correlation coefficient value, r, validates or explains the quality and reliability of the LWR model or linear regression. The scatter plot or regression graph of the total length and weight relationship of the species is shown in Fig. 2. The b, growth exponent value, for this species was 2.64 which is not statistically different from 3 (p = 0.5625, power of statistical test (1−ß error prob) = 0.8666, using small effect size of 0.1 and sample size of 90 on G*Power 3.1.9.2 version) when tested with Student’s t test to ascertain if the species grew isometrically or not. This indicates that the H. odoe in this study grew isometrically although this growth pattern is rarely or occasionally observed (Thomas et al. 2003; Bolarinwa 2015). This indicates that the fish will not change its shape as it grows. Froese (2006) reported that if b = 3, then small number of specimens in the fish sample have the same form and condition as large specimens. Le Cren (1951) and Fagbenro et al. (1991) stated that conformance with the cube law (isometric growth, b = 3) was rare in the majority of fish species and this was not true for H. odoe in this study because there was no deviation (p > 0.05 of 2.64) from the cube law (b = 3) when tested with Student’s t test. This value was within those (2–4) mostly reported for tropical freshwater fish species (Tesch 1971; Thomas et al. 2003). This is also in agreement with Carlander (1969) and Gayanilo and Pauly (1997) who postulated that exponent b normally falls between 2.5 and 3.5 for most tropical fish species. The regression equation for length-weight relationship of H. odoe shows that the species exhibited an isometric growth pattern. This indicates that there was dimensional proportionality (in body weight and total length) of the species at the same rate. Gonzales et al. (2000) reiterated that it is desirable to use already established growth exponents for the same species in other sites or country, but local data are better for quantitative assessments in the area because this parameter can have spatial disparities (Sparre et al. 1989; Kulbicki et al. 1993; Duarte et al. 1999). The growth pattern (b exponent) obtained for H. odoe in this study is in agreement with Offem et al. (2009), and Abdul et al. (2016) who observed similar value (isometry) for 76% of all the species investigated in Cross River wetlands in the Niger-Delta and Ogun coastal estuary respectively, but contradicts positive allometric pattern (b > 3) observed for this species in Epie creek in the Niger-Delta (Elakhame and Sikoki 2001), Ogbomoso Reservoir (Adedokun et al. 2013), Ado Ekiti Reservoir (Oso et al. 2011; Idowu 2013), Amassoma floodplain in the Niger-Delta (Ezekiel and Abowei 2014), Lake Eleiyele (Kareem et al. 2016) and River Sô, Benin Republic (Hazoume et al. 2017) and negative pattern (b < 3) in Ogbomoso Reservoir, Buyo Reservoir, Côte d’Ivoire, Lake Eleiyele, Ibadan and Lapai-Agaie Reservoir, Niger-State (Olaniran and Adeniran 2009; Tah et al. 2012; Akponine and Ayoade 2012; Baba-Usman 2017). Armstrong et al. (2004) and Gerritsen et al. (2006) were of the opinion that many biological parameters do vary over small geographical ranges. It has been shown that differences in the growth pattern of fish could be attributed to sampling season, number of individuals in the sample, location specificity (fishing area or habitat), genetic, biological and environmental factors such as temperature, pollution, nutritional status, feeding rate, stomach fullness, gonad maturity, spawning conditions, fishing pressure, sex, age, presence or absence of disease and parasite as well as differences in energy allocation (Bagenal and Tesch 1978; Lowe-McConnell 1987; King 1994; King and Udo 1998; Sikoki et al. 1998; Yaragina and Marshall 2000; Chouinard and Swain 2002; Slnovcic et al. 2004; Hossain et al. 2006, 2009, 2011; Sarkar et al. 2013). The observed growth pattern (isometric) in this study could be linked to feeding behaviour or intensity of the species which is determined by the abundance of food or prey items and the low or absence of competitors and predators such as Hydrocynus forskalii and Gymnarchus niloticus (Welcomme 2001; Pervin and Mortuza 2008). During rainy season, flooding exists, water level rises, food resources become abundant and fish density is at its lowest favouring intensive feeding (Welcomme 2001). When predator density is low, prey species like Hepsetus odoe will be less attentive and increase their time for foraging (Bayley 1988; Belk 1993). Fish generally forage to maximise growth rate rather than growth efficiency. The weight of fish increases when they utilise food items available for growth (Kamaruddin et al. 2012). Growth is also seasonal, being fastest during the flood or high-water season (Bayley 1988). The rapid growth during the high-water period generally corresponds to periods when food resources are abundant (as a result of flood promoting primary and secondary productions and bringing in large quantities of allocthonous food inputs into the system), temperature is at its highest (moderate) and density (competition) is at its lowest (Welcomme 1985, 2001; Junk et al. 1989; Quarcoopome 2016).
Species | Mean TL (cm) | Mean BWt (g) | a | b | Type of growth | R2 | p value of r | Mean K | t value |
|---|---|---|---|---|---|---|---|---|---|
Hepsetus odoe | 26.89 ± 2.47 | 137.11 ± 41.05 | − 3.80 | 2.64 | I | 0.61 | 2.2 × 10–16*** | 2.6 | − 1.6 |
BW body weight, TL total length, a intercept of the regression, b slope of the regression (growth exponent), I Isometric growth, r correlation coefficient of length-weight relationship, p value of r significance of correlation, K condition factor, t value absolute value of t test parameter to compare calculated slope to 3, p > 0.05
***highly significant
The condition factor, K, was used to assess the degree of well-being of H. odoe in the study area which provides information on the environmental quality and suitability (polluted or non-polluted of the ecosystem (Le Cren 1951; Bolger and Connolly 1989 cited in Guidelli et al. 2011). Wootton (1996) and Bagenal (1978) reported that fish with higher K values (> 1) are in a better condition than fish with lower K values (< 1). In this study, K values ranged from 1.6 to 5.2 with a mean value of 2.6 (Table 1) which indicates that fish were in good condition during the study period in the aquatic ecosystem. This range was within and over 2.9–4.8 reported for mature freshwater fish by Bagenal and Tesch (1978). Similar pattern (K > 1) was obtained for this species in Epe creek, River Zambezi, Ado Ekiti, Ogbomoso, Ogun coastal estuary and Eleiyele Reservoirs (Balogun 1980; Winemiller and Kelso-Winemiller 1994; Idowu 2013; Adedokun et al. 2013; Kareem et al. 2016) respectively. In contrast to this, lower K values (< 1) were obtained for H. odoe in Epie creek floodplain, Cross River inland waters, Ado Ekiti Reservoir and Amassoma floodplain (Elakhame and Sikoki 2001; Offem et al. 2009; Oso et al. 2011; Ezekiel and Abowei 2014) respectively. High K values have been attributed to food or prey availability and abundance and feeding intensity of the fish, good environmental conditions such as high dissolved oxygen, moderate temperature, low or absence of predators, low fishing pressure, genetic and immune and self-regulatory systems (Lagler et al. 1977; Bagenal 1978; Idowu 2007; Guidelli et al. 2011; Abdul et al. 2016). The good condition of this species could be due to good environmental or water quality parameters of temperature, 27.7 °C; pH, 6–7.5; and dissolved oxygen, 6.0 mg/L commonly reported for this lake system by Olayinka et al. (2017) which are ideally recommended and in agreement with 28–30 °C, 6.5–8.5 and ≥ 5.0 mg/L, respectively, for fish production in tropical waters by Boyd (1979), Boyd and Lichtkoppler (1985), Food and Agricultural Organization (FAO) (1993), Bhatnagar et al. (2004) and Bhatnagar and Singh (2010). This study did not investigate and confirm the evidence and absence of water quality parameters and chemical pollution (contaminants) of the lake which could have been flushed out of the aquatic basin by flood after raining. Furthermore, Rohde (1993) reported that the immune system of the fish could be enhanced when the species feeds adequately on food resources in its environment which could make individuals in better, healthier conditions to fight infestations and adapt to harsh environmental vagaries. The influence of condition factor, CF, on body weight, BW, and total length, TL, of Hepsetus odoe was carried out to ascertain significant relationships. There is a positive significant influence of CF on the BW (p = 2.2 × 10− 16, R2 = 0.30) but not on TL (p = 0.8528, R2 = 0.20).
The perpetuation of fish is dependent upon the process of reproduction, the success of which relies on resource allocation, location and timing of reproduction (Lagler et al. 1977). Spawning is a metabolically demanding activity and an important aspect in the biology and life history of fish (Ellis et al. 2012; McBride et al. 2015). Fecundity is seen as an adaptation to life in a certain environment. The formation of fecundity begins in early ontogeny after the gonadal differentiation. Fecundity has been estimated for many tropical fish species, but rarely with Hepsetus odoe. The fecundity estimates of Hepsetus odoe ranged from 243 to 3670 eggs with a mean value of 1349. The estimated fecundity range and mean values obtained in this study are within the ranges 245–3920, 387–4996, 556–2365, 2769–6669, 800–6155 and 1372–4853 eggs obtained from this species in Ado Ekiti Reservoir, Epie creek floodplain and Eleiyele Reservoir as reported by (Anetekhai et al. 1990; Elakhame and Sikoki 2001; Oso et al. 2011; Akponine and Ayoade 2012; Kareem et al. 2016; Idowu 2017). As a rule, the fecundity of the fish is inversely proportional to the degree of parental care it exhibits (Nikolsky 1963; Adebisi 1987). The low fecundity (mean of 1349 eggs) obtained in this study could be the confirmation of parental care exhibited by the fish. Being a batch or multiple spawners, Hepsetus odoe often exhibits parental care and a more protracted breeding period with only a proportion of eggs within the gonad becoming ripe at any one spawning (Lowe-McConnell 1987). Furthermore, generally, fish species with large egg diameter (size) are often less fecund (Ekpo 1982; Oben et al. 2000). The increase in the egg size is accompanied by a higher degree of parental care (Sargent et al. 1987). The basic idea proposed is that egg size and fecundity tend to be inversely related (Blaxter 1988). According to Elgar (1990), for any body size of fish, increases in clutch size are accompanied by decreases in egg and vice-versa. This also could explain the reason for the data obtained though not measured. It has been widely reported that the fecundity of fish could also be governed by its age, growth rate, size, genetic make-up, adequate food resource supplies and environmental factors such as temperature (Lagler 1956; Schmitt and Skud 1978; Fagade et al. 1984; Arawomo 1998; Adams et al. 2015). For instance, in majority of fishes, the number of eggs or fecundity at first, gradually increases with age and then as the individual approaches senility, it begins to decrease (Adams et al. 2015). Water temperature also plays a major role in influencing spawning periodicity and subsequent recruitment success (Morgan 2008). Under extreme temperature condition, adult female fish is forced to budget between survival and reproductive capacity (Pörtner et al. 2010). The low fecundity obtained in this study could also be traced to this environmental variable (though not measured) which is in agreement with Pike in Lake Liambezi where the water temperature changes had a profound influence on spawning or fecundity of the fish (Van der Waals 1985). Although not measured or quantified, low fecundity could be traced to abrupt changes in water level and pollutants (Wootton 1990). Gerking (1980) suggested that abrupt changes in water levels may also cause reduced fecundity in fish. The study area is known to have varied water levels during the rainy season. Koshelev (1984) revealed that in water-body subjected to anthropogenic pressure, the conditions for reproduction are the most important for the life cycle of the species. Jones and Reynolds (1997) reported that environmental pollutants such as heavy metals, pesticides and polycyclic aromatic hydrocarbons, could have a delayed or reducing effect on the reproduction of fish. Mansour and Sidky (2002) revealed that trace metals such as cadmium is known to cause impaired fecundity in fish. This statement is also supported by Scott and Sloman (2004) and Shobikhuliatul et al. (2013) that toxicant exposure of fish could disrupt effective performance of behaviours associated with all reproductive stages and thus reduces their reproductive success. Benejam et al. (2010) reported similar low fecundity of all the fish species investigated in a pollution-impacted reservoir. This low fecundity of fish could be as a result of low level of secretion of gonadotropins, hormones that regulate fish reproduction (gonadal development and production of steroids), which could have been affected by in vivo exposure to metals, pesticides and hydrocarbons (Adams et al. 1992; Ma et al. 1995; Johnson et al. 1997). Ambani (2015) affirmed that water pollutants, agricultural wastes, pesticides and trace metals can have histopathological effects on the reproductive tissues of fish gonads, thereby declining the ability of the fish to reproduce. Other effects include asymmetrical development and morphological deformation of the gonads, early sexual maturity, increase in the number of fish omitting spawning and destruction and resorption of sex cells (Akimova and Ruban 1992; Shatunovskii et al. 1996). Fecundity in a single population which has a greater food supply usually has a higher fecundity and vice-versa. Fecundity of fishes may undergo considerable fluctuations in relation to the supply of nourishment, i.e. food resources (Nikolsky 1963; McBride et al. 2015). Feeding condition can directly influence individual’s condition and energy expenditure into reproduction (Somarakis et al. 2012; McBride et al. 2015). This statement is strongly supported by Offem et al. (2011) that differences (low or high) in fecundity could be attributed to environmental factors such as differential abundance of food and water quality. This implies that shortage of food through intra- and interspecific competitions can cause low fecundity (Arawomo 1998). Hepsetus odoe have been reported to compete for food items among themselves and with tiger-fish (Hydrocynus spp.) (Winemiller and Kelso-Winemiller 1994). Furthermore, fecundity may also be reduced if individual fish matures at a smaller size (Offem et al. 2011). Smaller fish (such as Hepsetus odoe) are expected to be less fecund than larger ones (Musallam et al. 2006). Another important intrinsic factor which could regulate and responsible for this observable fecundity is species specificity (Alam and Pathak 2010). Bagenal and Braum (1978) and Khallaf and Authman (1991) had previously showed that fecundity is not a constant parameter, but fluctuates with variation in environmental conditions and is governed by species specificity. This means that they determine and regulate the amount of eggs to spawn at a given time as they have a flexible spawning time because the bubblenets they make allow them to reproduce at anytime even when the level of dissolved oxygen in their habitat is low. They do not have a distinct breeding season (Mospele et al. 2009). Nikolsky et al. (1973) proposed that fecundity is a specific fish species population regulatory mechanism in response to a certain level of egg and offspring mortality. This species specificity could also traced to a factor called ‘maternal effect’. Fecundity and egg quality of the fish are determined by the genotype and phenotype of the parents. Nikolsky (1962) reported that maternal effects could be responsible for fecundity, egg size and quality and spawning period of the fish which are all attributed to the size and condition of the mother. The relationships between the fecundity, fish body weight and total length are shown in Figs. 3 and 4 respectively. The regression equations and basic statistics for these relationships are as follows:
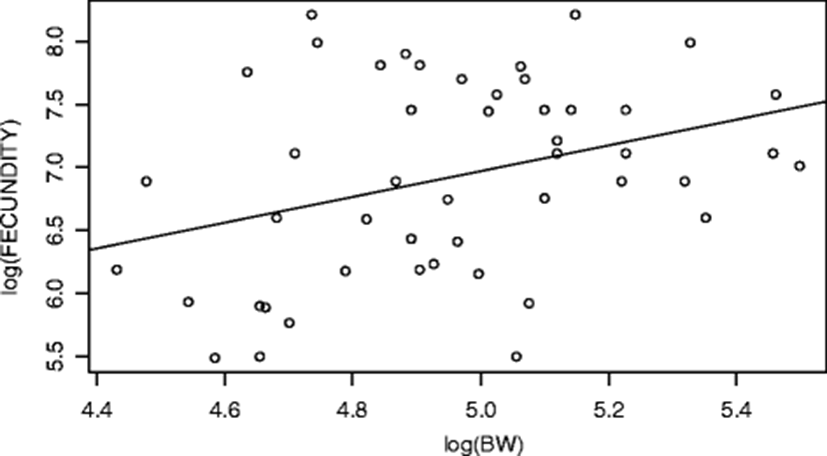
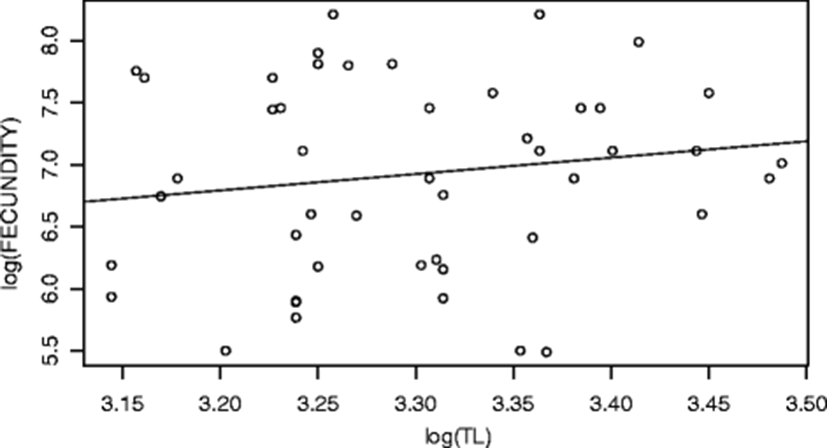
In the equations above, there is a highly positive significant influence (p < 0.001) of the total length (TL) and the body weight (BW) of the fish on its fecundity. Linear relationships of fecundity with body measurements were reported by Musallam et al. (2006). Reproductive output increases with body size. Trella (1998) revealed that fecundity increases with length faster than weight does. The low positive correlation coefficients, R2, obtained in this study are similar to that of Anetekhai et al. (1990), Oso et al. (2011) and Akponine and Ayoade (2012) and indicate that the fecundity is not only size dependent, but could also be governed by utilisation of food resources. Wootton (1973) and Ali and Kadir (1996) revealed that fecundity is more likely to be closely correlated with weight than length. Olurin and Savage (2011) did not find any relationship between fecundity and fish length. Jonsson and Jonsson (1997) were of the opinion that increase in body size of fish will increase body cavity to accommodate more eggs and more energy to produce more eggs. Jakobsen et al. (2009) argued that the whole body weight of fish is a significantly better predictor of fecundity than total length as it reflects surplus energy more realistically than the latter. Generally speaking, fecundity increases with increasing brood-fish size and weight and the number of eggs contained in a specific volume may also be different (Bromage et al. 1990; Bone et al. 1996; Carrillo et al. 2000). A significant relationship (p = 2.2 × 10–16, R2 = 0.37) was found between the fecundity and condition factor. This reveals that when the fish were in good condition (with K > 1), they spawned.
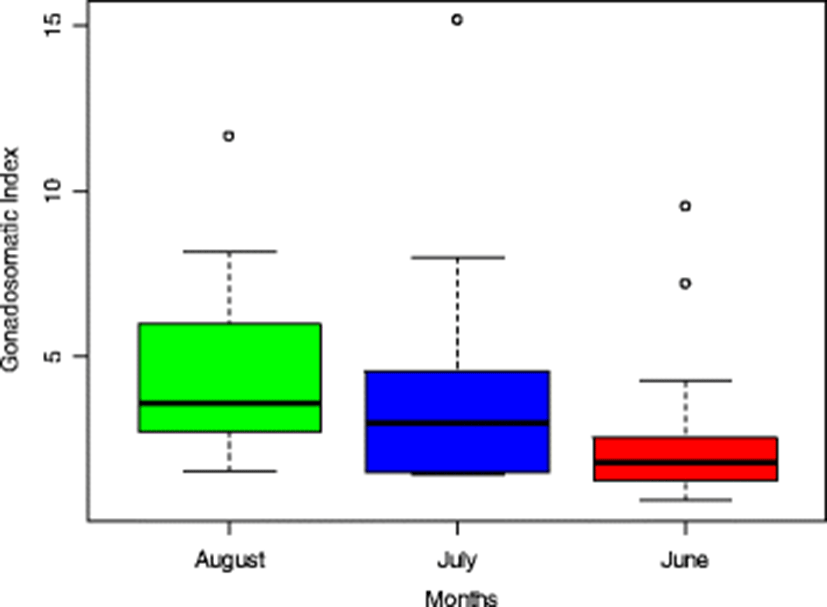
GSI is a common metric of reproductive allocation and condition in fisheries biology. There have been numerous studies in which GSI has been used as an indicator of gonad changes and development of fish, i.e. ovary and testis in terms of maturity and denotes the phase of the reproductive cycle (Yeldan and Avsar 2000). Gonadosomatic index is generally used to assess the maturation and spawning biology of fish. It is also used to assess the degree of ripeness of the ovary (Nandikeswari et al. 2014). The seasonal timing of reproduction is often identified from changes in the gonadosomatic index (Welcomme 1985; Biswas 1993; King 1995; Pieterse 2004). The GSI of Hepsetus odoe showed highest peak in August (4.53 ± 2.66) and lowest (2.63 ± 2.44) in June as illustrated in the box plot (Fig. 5). There was a steady increase in the GSI of the fish from June to August. These 3 months fell within the flooding period and condition (due to heavy rainfall) which is an important exogenous cue that stimulates gonadal development of the fish (Bruton and Jackson 1983; Van der Waals 1985; Merron et al. 1990). The highest peak value obtained in August is close to September reported by Idowu (2017) as the peak breeding month of the species. The influence of months (representing wet season) on GSI was tested. The result showed a significant influence (p = 0.0137, R2 = 0.12) of months on GSI. This shows and validates gonadal maturation during this period (rainy season).
The GSI range and mean values calculated for Hepsetus odoe were 0.64–15.15 (3.53%) respectively. Similar values (0.64–15.15 with mean 4.4%) were reported for this species in the same lake by Kareem et al. (2015) but higher mean value of 5% was reported by Anetekhai et al. (1990). The average value (3.53%) obtained in this study falls within (< 5 up to 30%) of several peaks for multiple (batch) spawners during breeding season as explained by Wootton (1990). These differences are governed largely by the temporal pattern of egg development and spawning. The gonadosomatic index increases as the fish matures and reaches to its maximum at the peak period of maturity. Increases in GSI during the reproductive cycle reflect the growth of developing oocytes during vitellogenesis. Its abrupt decrease denotes the beginning of spawning (Nandikeswari et al. 2014). Buxton (1990) pointed out that low value of GSI in case of female fish is due to low energy investment in gamete production. This could have accounted for the values obtained in this study. Likewise, a low GSI could be due to pollutants or contaminants received by the inland water. Kime (1995) and Pieterse (2004) reported that a long-term exposure of fish to pollutants could lead to a decrease in GSI, more smaller, less developed oocytes and fewer large, mature oocytes. Louiz et al. (2009) were of the opinion that a low GSI in fish has often been linked to gonad disturbance in polluted sites which could be probably due to atresic oocytes and disintegration of cytoplasm that are often found in their ovaries. Morley et al. (2010) reported a low GSI for fish investigated in polluted water of Cranleigh Waters but were in good condition (with K > 1).
GSI is influenced during the breeding period of fish by the amount of yolk accumulation and uptake of fluid by ripe oocytes and environmental variables such as pollutants like industrial effluents (Idowu 2007; Offem et al. 2011). Because GSI value was obtained from gonad weight (GW) and body weight, it is statistically important to know the relationship (Fig. 6) between these two variables. When calculated, it was observed that there was a positive significant relationship (p < 0.05) between GW and BW. As the BW increases, GW increases. This is expressed in the equation:
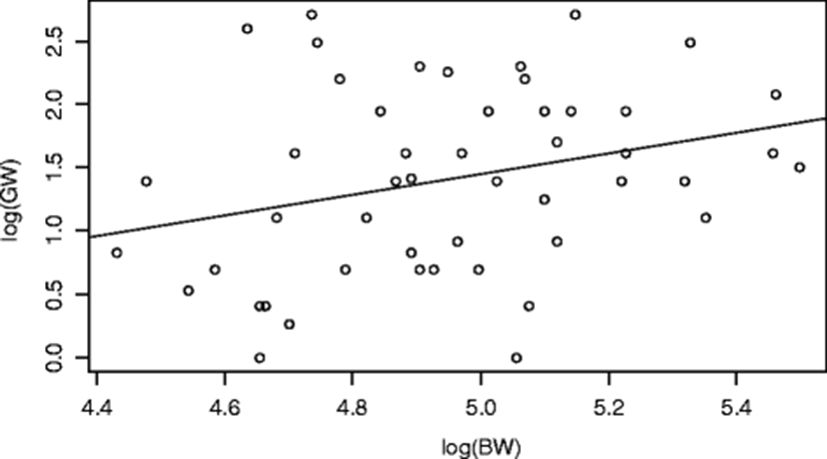
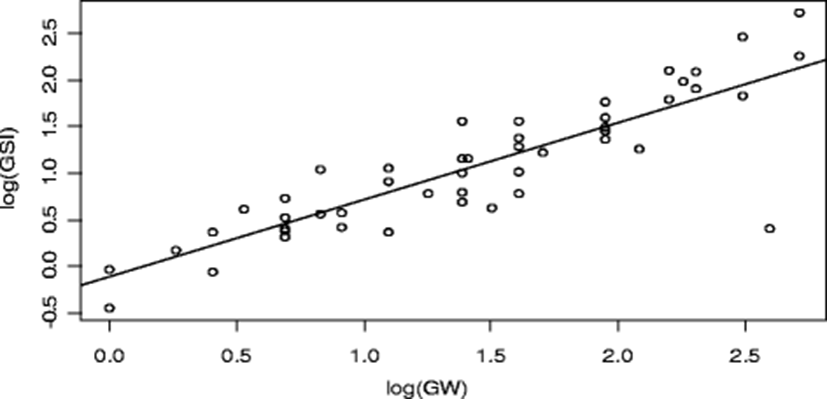
Furthermore, there was a highly positive significant (p = 3.25 × 10− 16, R2 = 0.76) influence of GW but not BW and their interaction (GW:BW) on GSI. This shows that GW contributed more to GSI of the fish than its BW. As the GW increases, GSI increases (Fig. 7).
Conclusions
Based on visual observation of fish landings while on the field in this season, Hepsetus odoe was considered to be the third most targeted species (after Tilapia and Chrysichthys nigrodigitatus) among other pelagic fishes and their total landings may fluctuate from region to region along the inland water areas. This study of the length-weight relationship, condition factor, fecundity and gonadosomatic index of Hepsetus odoe in Lake Eleiyele provides information on the growth pattern (isometric), not commonly observed in tropical fishes, well-being (good condition) and reproductive indices of the species. Our major concern is the isometric growth pattern (in contrary to negative allometry expected) displayed by the species (although a very high statistical power of 0.866 was obtained when tested); this indicates that the fish became robust (heavy) proportionately as its length increased (Bagenal and Tesch 1978). This would command a good size for the landed fish and market price that could benefit the fishermen from the sales. The good condition observed for this species in the lake system would have a potential significant role of enhancing the spawning, recruitment, growth, survival and abundance of the species which could support and favour the population of predators, Gymnarchus niloticus, Hydrocynus forskalii, but a declining and deleting effects on the demography of prey organisms, tilapia, mormyrids, Synodontis spp., Barbus spp., Schilbe mystus, as a result of intraspecific competition along the food chain (Adedokun and Fawole 2012). Hepsetus odoe usually grow large, heavy, abundantly in inland waters, consequently, can reduce the abundance and species richness (loss in biodiversity) of prey-sized fish such as cichlids, thereby ecologically affecting important herbivorous species. This could threaten the livelihoods within riverine communities. In response to this, management efforts should be geared and directed to control their population to a level that will minimise ecological and economic impacts. Additionally, comprehensive research should be conducted on aquaculture of the species as it is a good alternative candidate (to catfish) that can be used to depopulate ponds invaded and overcrowded with cichlids such as tilapia which have been regarded as a potential pest that could disrupt the ecosystems in waters where it is introduced. (Pullin and Lowe-McConnell 1982).
Individual or combination of many factors such as sex, season, size, food availability, competition, genetics, gonad development and maturation, environmental or water quality etc. could have contributed immensely to the observed parameters on the biology of the investigated fish species. This study could not account, clarify and ascertain which factors were responsible due to time and budget constraints. The findings of this research demonstrate that the isometric growth and well-being of H. odoe in Lake Eleiyele, but poor fecundity and gonadosomatic index, which might be attributed to anthropogenic activities that have led to reservoir pollution and resultant low reproductive indices of the investigated fish therein. Kime (1995, 1998) revealed that fecundity, gonadosomatic index and early life stages are the most sensitive indicators of long-term impact of pollutants on reproduction of fish as it affects the number of eggs spawned due to reduced availability of vitellogenin for yolk production. Barton et al. (2002) and Benejam et al. (2008) affirmed that long exposure of fish to chemical pollutants can ultimately have detrimental effects on their reproduction. This means that inhibition of normal reproductive capacity by pollution might have serious implication for fish population, as a decrease in fecundity and GSI can result in low recruitment and decline in size of the community. Quantifying the reproductive potential of H. odoe is needed to understand the population dynamics of this species and for stock assessment purposes. Therefore, it is necessary to control the disposal of domestic sewage and industrial effluents into the lake to prevent metals and other pollutants from getting into the lake.
Paradoxically, it can be concluded that the fish (Hepsetus odoe) exhibited some degree of tolerance (isometric growth pattern and in good condition) in the degraded ecosystem (Eleiyele Lake), but there was an adverse effect of the pollution on its reproductive indices. It is therefore recommended that concerted efforts should be made to monitor, improve, maintain and sustain the environmental conditions for continuous, thriving of this species and other aquatic resources inhabiting the lake system. Furthermore, the spawning ground area of the fish species must be closed down to fishing during the peak breeding season, which often corresponds with the period of the high flood. This will allow successful breeding activities and recruitment in the subsequent season. The knowledge of the reproductive and growth biology of the species can be utilised to compare and ascertain the time and the number of recruitments and population dynamics from spawning events, enhance effective fisheries management, sustainable exploitation of this fish resource and model the lake system. Further research needs to be conducted in order to validate and obtain more information about the investigated fish biology, especially in the dry season as season plays a significant role in the growth pattern, fecundity, life cycle, feeding habits and physiology of fish (Tesch 1971; Bagenal 1978; Froese 2006; Atama et al. 2013).