Introduction
About 60 seagrasses are distributed worldwide except for the Antarctic (Den Hartog, 1970; Phillips & Menez, 1988; Short et al., 2007). Among them, 9 species (five species in genus Zostera, two species in genus Phyllospadix, one species in genus Ruppia and one species in genus Halophila) are found on the Korean coast (Kim et al., 2009; Lee & Lee, 2003; Lee et al., 2018; Shin et al., 2002).
Zostera caespitosa has been considered one of the endemic species in this region (Omori, 1993). This species can easily be distinguished from other species genus of Zostera by their non-generated creeping rhizomes, extremely shortened internodes, obcordate leaf apex and separately tufted appearance (Kuo & Den Hartog, 2001; Shin & Choi, 1998). Leaf blades of Z. caespitosa grow up to 70 cm tall and 2.5–4.5 mm wide and 6–10 male and female flowers are arranged per spadix (Kuo & Den Hartog, 2001).
Seagrasses reproduce in two ways, sexually by flowering and asexually by clonal growth (Den Hartog, 1970; Eriksson, 1989; Eriksson & Froborg, 1996). Asexual reproduction has been considered the dominant process for the establishment and maintenance of seagrass meadows (Hemminga & Duarte, 2000). Sexual reproduction is an important mechanism to maintain the genetics and regeneration of many seagrass populations (Kendrick et al., 2012, 2017; Orth et al., 2000; Plus et al., 2003).
Seagrasses are considered to be one of the most important shallow marine ecosystems by various roles that provide habitats and nursery grounds for fish and invertebrates as well as filtering coastal waters and anchoring sediments (Costanza et al., 1997; Green & Short, 2003; Orth et al., 2006).
Seagrass meadows have been disappearing at a rate of 110 km2 a year since 1980 (Short & Wyllie-Echeverria, 1996; Waycott et al., 2009). This is a result of disturbances by nature and human activity such as grazing herbivores, hurricanes, harbor construction, and sewage disposal.
There have been few studies on Z. caespitosa in Korea (Lee et al., 2002, 2005b, 2005c). In this study, to clarify the phenology of Z. caespitosa, we investigated the variation of morphological features, density, biomass and characteristics of reproductive shoots of Z. caespitosa in Tongyeong, Korea. We should provide not only valuable information on the biology and ecology of this species but also more detail characteristics on reproductive shoots.
Materials and Methods
This study was carried out in Tongyeong on the southern coast of Korea (Fig. 1). Three seagrasses (Z. caespitosa, Z. marina and H. nipponica) were found in this area. These seagrasses had different distributed depths; H. nipponica was found in shallow water at a depth of 1–2 m. Z marina and Z. caespitosa were found at depths of 1–3 m and 3–5 m, respectively. The meadow of Z. caespitosa showed a monotypic form at a depth of 5 m and mixed form at a depth of 3 m depth with Z. marina. This coast is open sea, not sheltered by wave action, and the sediment has a sand-muddy composition. Water temperature and salinity data were obtained from the Korea Hydrographic and Oceanographic Administration (http://www.khoa.go.kr).
In this study, Z. caespitosa was collected in the study area from January to December 2016 by skin scuba diving. To determine density and biomass, a 50 cm × 50 cm quadrat was used, which contained three tufts in a quadrat. Above ground morphological features such as leaf, sheath length, leaf width and density, and biomass were measured monthly. The number of leaves and plant biomass in the quadrat were measured and converted to unit area values. Shoot height and the length and width of reproductive shoots were also measured during the reproductive shoot period.
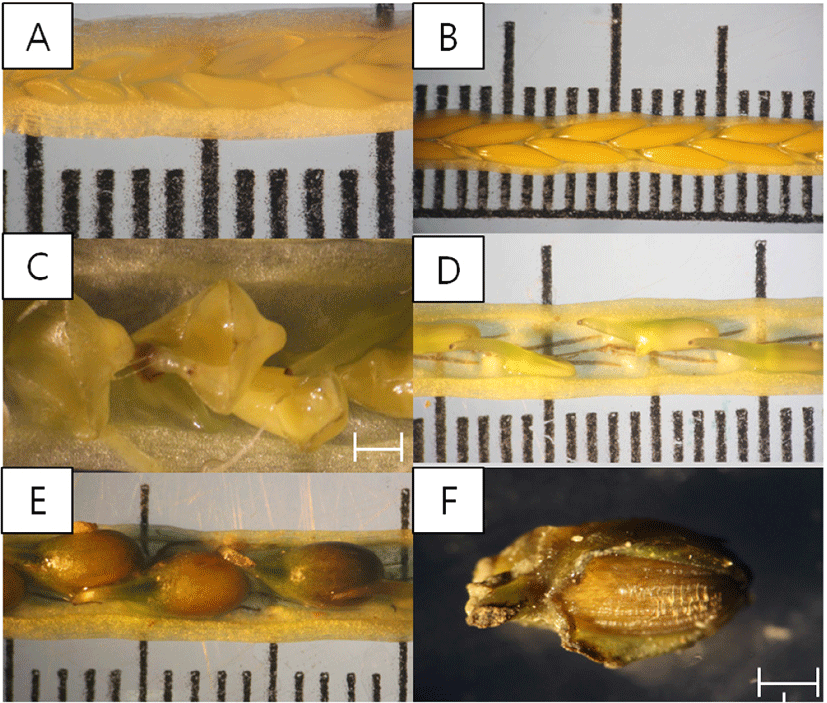
The development of inflorescence on Z. caespitosa was classified according to the modified division method by Yoon (2019), which was adapted from Alexandre et al., 2005. Maturity development of spathe on genus Zostera was divided into six stages, with zero as undefined flowers inside the immature inflorescence; I, flowers aligned on a single stem and sheath closed; II, mature females erected outside sheath for fertilization; III, filiform pollen released by mature males (anther dehiscing); IV, fertilized females, thecae empty (if still present); and V, presence of fruits (Va, small embryos inside the female flowers; Vb, fully developed fruit; Vc, creviced fruit skin [ready for seed release]; Vd, released seed [only fruit skin remaining, without seed]). The length and width of the spathe and spadix, weight of spathe and fruit, and number of anthers, pistils and fruits per spadix were measured during the flowering period.
Statistical analyses were performed using the SPSS Windows program (Release 20.0, SPSS, Chicago, IL, USA ). Monthly variation of morphological features and characteristics of inflorescence were analyzed by one-way analysis of variance (ANOVA). When a significant difference was observed, the means were analyzed by Duncan’s multiple range test to establish significant differences among them (p < 0.05). The relationship between water temperature and morphological features was estimated using curve estimation, and the significances of these regressions were tested using ANOVA with the significance level set at 0.05. All values are reported as mean 95% confidence interval ± 95% confidence interval.
Results
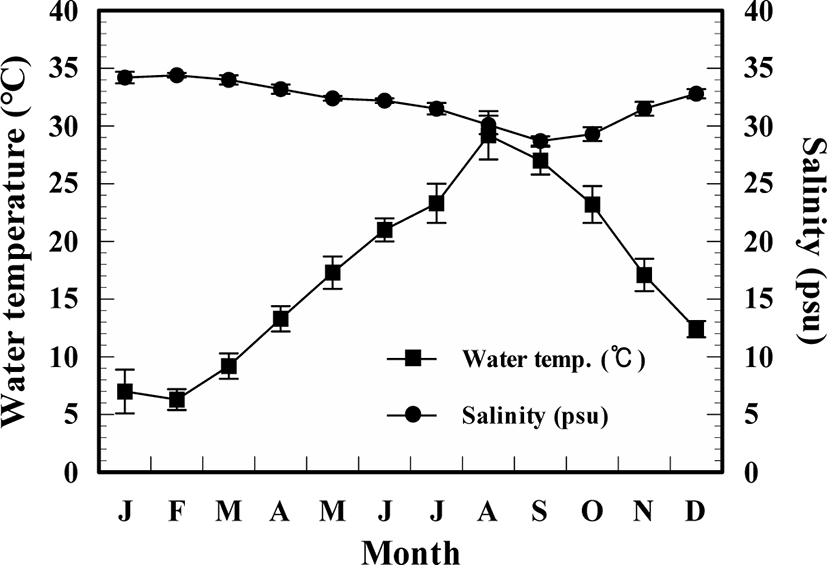
There were clear seasonal variations in water temperature in the study area, but not salinity (Fig. 2). The annual mean values of temperature and salinity were 17.2 ± 7.1°C and 34.3 ± 0.2 psu, respectively, and they had the highest value each in August (29.2 ± 2.1°C) and February (34.3 ± 0.2 psu) and lowest value each in February (6.3 ± 0.9°C) and September (28.7 ± 0.4 psu).
Above ground, Z. caespitosa showed clear seasonal variations in leaf and sheath length and leaf width, (p < 0.001) and displayed a significant positive linear relationship (R2 = 0.15, 0.54, 0.21, p < 0.001 for each leaf and sheath length and leaf width). Leaf length increased in spring (March) to early summer (June) and dramatically decreased in midsummer (July) and then slowly decreased. The highest value of leaf was 141.4 ± 3.2 cm in June and lowest value was 55.9 ± 2.8 cm in November. Sheath length also increased from spring to midsummer and decreased in late summer in August. The highest sheath value was recorded in July (16.3 ± 0.9 cm) and the lowest value in January (8.3 ± 1.8 cm). The widest value of leaves was recorded in May at 5.0 ± 1.1 mm and the narrowest in winter (January) at 4.0 ± 0.7 mm. Leaves grew gradually wider in spring, were the widest in spring and slowly became narrower in fall (Fig. 3).
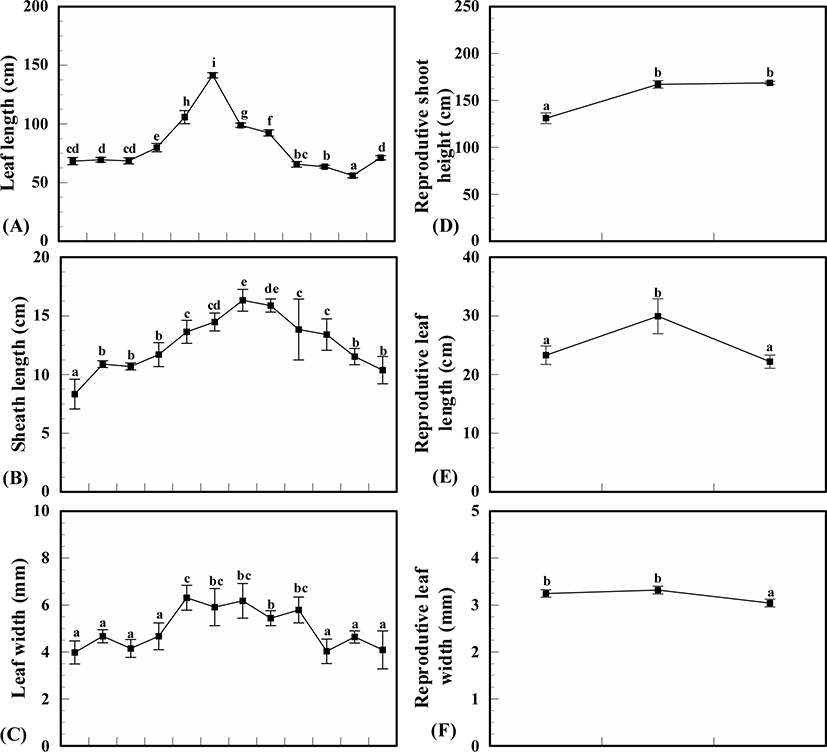
The reproductive shoots of Z. caespitosa occurred from April (13.3 ± 1.1°C) to June (21.0 ± 1.0°C) and disappeared after June. Shoot height, leaf length and width of reproductive shoots showed monthly variations (p < 0.001). The average height of reproductive shoots was recorded as 155.6 ± 18.6 cm, with the longest in June (168.7 ± 2.4 cm) and shortest in April (131.0 ± 8.1 cm). The leaf length and width of reproductive shoots had the highest values in May, 29.7 ± 4.2 cm and 3.3 mm, respectively, and lowest values in June, 22.0 ± 1.6 cm and 3.0 ± 0.2 mm, respectively (Fig. 3).
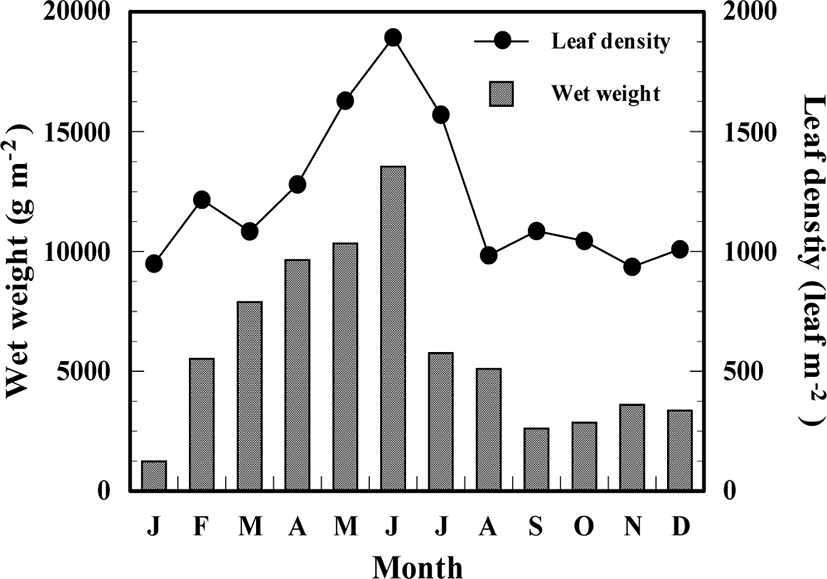
Shoot density and biomass of Z. caespitosa showed seasonal variations, with an increasing tendency during spring and early summer, and dramatically decreasing in midsummer. The number of leaves ranged from 936 to 1,893.0 leaf m−2 and had the highest value in June and lowest value in November. Biomass ranged from 1,224.0 to 13,541.6 g w.w.m−2 and recorded the maximum value in June and minimum value in January (Fig. 4).
Inflorescence of Z. caespitosa exhibited various maturity stages from I to Va, with erected pistils (Fig. 5B), anther dehiscing (Fig. 5C) and small embryos (Fig. 5D) in April. In May, fully matured fruits were observed (stage Vb, Fig. 5E). In stage Vc, cracks in fruit skin, and in stage Vd, traces of released seeds were observed in July (Fig. 5F). There was no significant difference in the length and width of spathe and spadix (p > 0.05), but there was a significant difference in the weight of spadix and fruits and the number of fruits (p < 0.001) between months in the flowering period. Spathe was longest and widest in May and April, at 6.9 ± 0.9 cm 5.7 ± 0.6 mm, and shortest and narrowest in July, at 6.3 ± 0.6 cm and 5.3 ± 0.3 mm. Spadix was longest and widest in April, at 5.6 ± 0.7 cm, 4.1 ± 0.4 mm, and shortest and narrowest in July, at 5.4 ± 0.7 cm, 3.8 ± 0.2 mm. The weight of spathe in July was almost 3- to 4-fold higher than in April. The number of pistils and anthers per spadix were recorded as 10.0 ± 2.6 and 18.5 ± 4.7 in April. Pistils and anthers were not observed after April. The highest number of fruits was seen in May (9.6 ± 1.3) and the lowest in June (5.2 ± 1.3). Fruit wet weight was almost two to three times higher in July than in April (Table 1).
Discussion
Morphological features, density and biomass of seagrass usually exhibit seasonal variations, and these values were reported as fluctuating trends that increased during spring and summer and decreased in fall and winter (Dunton, 1994; Lee & Dunton, 1997; Zhang et al., 2016). But in this study, the density, biomass and growth of Z. caespitosa increased in spring and early summer and decreased in midsummer, and these results are similar to those of Lee et al. (2005c).
Water temperature has been considered a major factor in controlling growth, and this factor can limit the growth and germination of seagrass during high temperature periods (Biebl & McRoy, 1971; Lee et al., 2007; Morita et al., 2010; Phillips et al., 1983). Seagrasses may show maximum growth with optimal water temperature, and photosynthetic rates decreased when over 30°C (Marsh et al., 1986). Höffle et al. (2011) reported that the mortality rate of Z. marina cultured at 27°C was higher than that cultured at 18°C and 21°C. In the study site, the water temperature almost reached 30°C in August and at that time, leaf length, leaf density and biomass already decreased, so the decreasing tendency in midsummer coincided with the season of flowering shoot senescence.
The growth, morphological features, density and biomass of seagrass showed different values with environmental factors such as temperature, salinity, photon radiation, sediment and latitude (Bradley & Stolt, 2006; Bulthuis, 1987; Livingston et al., 1998; Lee et al., 2005a; Phillips et al., 1983). Lee et al. (2005c), who conducted a studied on Geoje Island off the southern coast of Korea, reported that the blade length and width of Z. caespitosa ranged from 23.9 to 100.2 cm and 4.4 to 7.6 mm, respectively. This indicated that Z. caespitosa distributed in our study area may be longer and narrower than that near Geoje Island. In our study, the shoot growth rate was optimal rate during the rising water temperature period from March (13°C) to June (21°C). This is similar to the study by Lee et al. (2005c).
It has been reported that there is no considerable variation in the number of shoots on Z. caespitosa (Lee et al., 2005c) but in this study, there was a clear seasonal variation in the number of leaves on Z. caespitosa (p < 0.001). This difference suggests that the number of leaves in turf fluctuates more than the number of shoots.
The relationship between water temperature and morphological features was significantly different (p < 0.001) but exhibited a poor relationship. This result may be related to the growth of Z. caespitosa showing a positive correlation in the rising water temperature period (spring, early summer) but show a negative correlation in the summer high temperature period.
Several studies have said that temperature is the most important factor that controls and regulates seagrass reproduction (Buia & Mazzella, 1991; McMillan, 1976; Phillips et al., 1983). Alexandre et al. (2006) reported that the pollination process (Stages I–IV) required about 10 days for Z. noltii. In our study, various stages (I–Va) of inflorescence of Z. caespitosa were observed in April; therefore, the first occurrence of reproductive shoots may occur at the end of March (10°C) and the time of appearance was similar to a study by Lee et al. (2005c). The flowering period was affected not only by water temperature, but also latitude and genetic variation (Phillips et al., 1983). Lee et al. (2005b) in a study in Ulpo, Korea reported that the reproductive shoots of Z. caespitosa observed from February (10°C) to May (16°C), in the study area had a longer period of occurrence.
In our study, the weight of spathe showed clear seasonal variations and a tendency to increase; these variations may be affected by the fluctuation of fruit weight. The number of fruits decreased by 54% in June compared to May, and this reduction may be related to the release in seeds by maturation.
In conclusion, there were clear seasonal variations of the morphological features of vegetative shoots and reproductive shoots, density and biomass of Z. caespitosa, and it had an optimal growth rate of 9°C–21°C from spring to early summer. Reproductive shoots may occur at 10°C and seeds may mature and release above 20°C.