Introduction
The challenge of food spoilage and rancidity was less significant then than now as food products these days are often stowed for days or months before their distribution and consumption. Food spoilage is often attributed to the undesirable activities of microorganisms while food rancidity is often caused by autoxidation of lipids-related compounds which are initiated by free radicals. Common practices in controlling microbial contamination and oxidative deterioration in the food industry involve the incorporation of food additives and preservatives. However, rising concerns of side effects and carcinogenicity of synthetic food additives prompt for prospection of naturally derived bioactive with significant antioxidants and antimicrobials activities. Such natural bioactive, preferably from continuous dietary items, have shown to be promising in improving the shelf life and quality of various food products (Chan et al., 2012a; Chan et al., 2014b).
Jellyfish belong to the phylum Cnidaria and edible ones are from the class Scyphozoa (jellyfish or true medusa) where most, if not all, falls into the order Rhizostomeae. Edible jellyfish are commonly available in the market as either the bell or oral arms, where a bell includes the exumbrella, mesoglea, subumbrella, stomach, and gonads of the jellyfish, whereas the oral arms include the manubrium and oral arms, following the common processing practice of fishermen. Jellyfishes are unique as compared to other organisms as they possess the ability to retain high water content. They are a popular delicacy among locals and overseas consumers with increasing demands from markets in China, Japan and Korea. Jellyfish have been an important fishery commodity in many countries in Southeast Asia especially in Vietnam, Malaysia, Thailand, Indonesia, and the Philippines (Nishikawa et al., 2008). One strong promoting factor of jellyfish consumption involves their traditional use as part of the Chinese Materia Medica in the relief of swelling, hypertension, asthma, and bronchitis. Cnidarians have been reported to contain high content of isoprenoid, acetogenin, terpenoid, and peptide, with jellyfish having high amounts of protein and peptides (Harper, 2001). Khong et al. (2016) reported collagen as the major protein found in edible jellyfishes. The river jellyfish, Acromitus hardenbergi is abundantly caught from the Perak River, Malaysia. Year-round availability of A. hardenbergi in the Perak River is unique worldwide, as commonly, jellyfish fisheries are characterized by a short, restricted fishing season that lasts from two to four months (Yusoff et al., 2010). In a recent ethnopharmacological survey performed in Bagan Datoh, Malaysia, local fishermen have been attributing the cure of joint problems and slowing of aging as well as fatigue to the consumption of this jellyfish (unpublished results).
The jellyfish fisheries are highly neglected in both research studies and its socio-economic development even though this industry provides a very important source of revenue. The fishery industry of A. hardenbergi in Malaysia has been carried on for more than three generations with very limited information available. In addition, their contributions towards pro-health elements are scarcely reported. Therefore, this study aimed to fractionate the bioactive in the bell and oral arms of the jellyfish sequentially using solvents of ascending polarity and to investigate the antioxidant and antimicrobial activities of the fractions obtained. This provides significant insights to prospective bioactive leads with valuable potentials to beneficial outcomes in supplementing desirable nutraceutical benefits.
Materials and Methods
All chemicals used were of analytical grade. Petroleum ether (PE), dichloromethane (DCM), chloroform (CHCl3), methanol (MeOH), dimethyl sulphoxide (DMSO), sodium bicarbonate, Folin-Ciocalteu’s phenol reagent, Mueller-Hinton agar and Mueller-Hinton broth were obtained from Merck KGaA, Darmstadt, Germany. Linoleic acid, gallic acid, β-carotene (Type I synthetic, 95%), 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox), 2,2’-azino-bis(3-ethylbenzthiazoline-6-sulphonic acid) (ABTS), potassium persulphate, 2,2-diphenyl-1-picrylhydrazyl (DPPH), 5,5-dimethyl N-oxide pyroline (DMPO), hydrogen peroxide (H2O2) and ethylenediaminetetra acetic acid (EDTA) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Tween 20 and 0.5 McFarland Standard were obtained from Thermo Fisher Scientific (Waltham, MA, USA). All aqueous solutions were prepared using Milli-Q water (Millipore, Molsheim, France).
The river jellyfish, Acromitus hardenbergi was collected from the Perak River at Bagan Datoh, Perak, Malaysia. A. hardenbergi was identified similarly as described before (Khong et al., 2018), where the species is easily distinguished by the absence of terminal appendage on the oral arms of the organism. The jellyfish bell (JB) and oral arms (JOA) were instantaneously separated typical to the conventional processing of jellyfish by local fishermen, stored in 4°C and transported immediately back to the laboratory. JB and JOA were cleansed thoroughly with distilled water to remove exogenous matters as well as mineral salts prior to lyophilization.
Schematic representation of the sequential extraction procedure is depicted in Fig. 1. Briefly, freeze-dried JB and JOA powders (30 mesh sieved; finale moisture content (%): JB: 12.17 ± 0.59; JOA: 15.93 ± 0.61) were extracted sequentially in an increasing polarity gradient by using PE, DCM, CHCl3, MeOH, and water (H2O). Samples were homogenized (Ultra-turax T25 basic, IKA®-WERKE GmbH & Co. KG, Staufen, Germany) with the solvent (1:10 w/v) for 15 min at 9,500 rpm. Subsequently, the mixture was filtered through Whatman No. 1 filter paper and the residue was re-extracted twice using the same procedure before drying and proceeding to the next solvent. Solvents were removed from the resulting fractions under reduced pressure or lyophilization (Rotavapor R210, Buchi, Postfach, Flawil, Switzerland; Virtis Benchtop K Freeze Dryer, SP Industries, Warminster, PA, USA). The yield of each fraction was measured, and the fractions were stored at –80°C till further analysis.
Radical scavenging activities of jellyfish fractions against various radicals was determined by using the electron spin resonance (ESR) spin-trapping technique (JEOL, JEOL-JES-FA100, Tokyo, Japan) with the conditions as follows: sweeping field, 336.45 ± 5 mT; microwave power, 8 mw; mod width, 0.1 mT; sweep time, 2 min; time constant, 0.1 s and amplitude, 160. The ESR measurement was conducted 1–2 min after preparing each reaction mixture at room temperature. The radicals used (DPPH, ABTS·+, and ·OH) were prepared as follow:
DPPH radical was generated according to the method of Chan et al. (2012c). Radical scavenging activities of the fractions were determined by reacting 50 µL of individual sample with 195 µL of DPPH methanolic solution (0.2 mM) in a 96-well plate. Homogenized mixtures were allowed to stand in the dark for 1 h and spectrometry of radical-sample mixture was measured using ESR. Trolox was used as the standard.
ABTS radical cation was produced by reacting 50 mL of 7 mM ABTS stock solution with 50 mL of 2.45 mM potassium persulfate for 24 h in dark at room temperature as described previously (Chan et al., 2012c). The ABTS·+ scavenging activity was measured by reacting 950 µL of ABTS·+ solution (adjusted to 0.70 ± 0.02 at 734 nm with ethanol) with 50 µL sample and allowed to react in the dark at room temperature for 10 min. Spectrometry of radical-sample mixture was measured using ESR. Trolox was used as standard.
Hydroxyl radical (·OH) was generated through Fenton reaction according to Chan et al. (2014a), with DMPO as the trapping agent. The reaction mixture contained 40 μL of DMPO (400 mM), 37.5 μL of FeSO4 (0.4 mM), 112.5 μL of EDTA (0.1 mM), 60 μL of sample or blank and 150 μL of H2O2 (2.0 mM). ·OH scavenging activity of all jellyfish fractions were determined by using ESR. DMSO was used as standard.
The BCB inhibitory activity of test fractions was evaluated by the model system described by Chan et al. (2012c). In brief, 3 mL of β-carotene solution (1 mg β-carotene/ 10 mL CHCl3) was added to 120 mg of linoleic acid and 1,200 mg of Tween 20 followed by drying under vacuum. After that, 100 mL of distilled water was added to the dried mixture to form β-carotene-linoleic acid emulsion. Next, 1.5 mL aliquots of emulsion were transferred into different test tubes containing 20 µL of samples followed by incubation at 50°C for 1 h. Absorbance of all samples at 470 nm were taken before (t = 0) and after 60 min of incubation. Trolox was used as the standard.
The reducing potentials of various jellyfish fractions were assessed using Folin-Ciocalteu reagent following the method of Chan et al. (2014a). Briefly, 2.5 mL of 10% Folin-Ciocalteu phenol reagent were dispensed into 500 µL of sample followed by 2.0 mL of 7.5% Na2CO3 prior to incubation at 40°C for an hour. The absorbance of samples was measured using a spectrophotometer (Pharmaspec UV-1700, Shimadzu, Kyoto, Japan) at 765 nm. Results were expressed as gallic acid equivalents (GAE) in milligrams per gram of fraction.
Escherichia coli ATCC 25922, Salmonella typhimurium ATCC 14028, Staphylococcus aureus ATCC 25923, Enterococcus faecalis ATCC 19433, Bacillus cereus ATCC 13061, Bacillus subtilis ATCC 6633, Saccharomyces cerevisiae ATCC 2601 and Candida albicans ATCC 10231 were purchased from American Type Culture Collections (ATCC) and maintained in the microbial culture collections of the Department of Microbiology, Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia. All bacteria were cultured and maintained on Mueller-Hinton agar and broth (pH 7.2 ± 0.2).
A standardized Kirby-Bauer disc agar diffusion procedure was performed to determine susceptibility of microorganisms towards jellyfish extracts (CLSI, 2006). Inoculum of microorganism were prepared from exponential phase old broth cultures. The absorbance was read at 550 nm and adjusted with the same sterile medium used to grow the bacterium to match 0.5 McFarland standard solution. Mueller Hinton Agar plates were lawned with bacterial suspension evenly. Sterilized filter paper disc (5 mm diameter) was each saturated with 30 μL of with respective fractions in the concentration of 1.0 mg/ mL and allowed to dry. Discs were aseptically placed on agar surface at a well-spaced interval ensuring a firm contact with agar surface. Antibacterial activity of jellyfish fractions was assessed by measuring the diameter (mm) of growth inhibitory zone upon 24 h incubation at 37°C, while antifungal activity was assessed similarly upon 48 h incubation at 25°C. The assay was repeated thrice for each fractions.
A spectrum 100 FTIR spectrometer accessorized with an ATR (PerkinElmer, Waltham, MA, USA) was used for FTIR data collection. Using a Micro Chattaway spatula, each fraction was placed directly to the universal ATR polarization accessory at controlled room temperature. All samples were analyzed over that scanning range of 4,000 to 280 cm–1 at a resolution of 4 cm–1. To verify possible noises, background spectrum was collected and compared to the previous one. FTIR spectra of all fractions was recorded as absorbance values at each data point in triplicate, following which principal component analysis (PCA) was performed to uniquely reduce the dimension of large datasets into principal components using Minitab 17 statistical software (Minitab, State College, PA, USA).
Values are given as the means ± SD of three replications. Where appropriate, data were analysed using GraphPad Prism Version 5.0 (GraphPad Software, San Diego, CA, USA). One-way ANOVA accompanied with least significant difference (LSD) tests by using SPSS Version 17.0 (SPSS, Chicago, IL, USA) to identify the significant difference between treatments (p < 0.05).
Results and Discussion
The yield of both JB and JOA fractions were found to be similar i.e. decreasing in the order of MeOH > H2O > CHCl3 > DCM > PE (p < 0.05) (Table 1). Methanol fraction yielded the highest as compared to the other four fractions for both JB (44%) and JOA (38%) (p < 0.05). Sequential gradient fractionation was performed so as the bioactive compounds/ metabolites fractionated would be distributed according to their polarity. Commonly, PE tends to extract metabolites of low polarity such as oil (hydrocarbon and fatty acids) and lipophilic compounds (acetogenins and terpenes) whereas DCM extract yields compound of medium polarity such as peptides and depsipeptides. On the other hand, substances such as saponins, polyhydroxysecosteroids, alkaloids and amino acid derivatives can be found in polar extracts as extracted by CHCl3, MeOH, and H2O (Ebada et al., 2008). The meagre yield in the PE fraction was paralleled to our previous finding of limited lipids in the jellyfish (Khong et al., 2016). Besides, we also found that jellyfishes contain high amount of protein and carbohydrate (Khong et al., 2016). The high yield of the methanol fraction might indicate high content of glycoproteins and carbohydrates that are commonly separated from food matrices using alcohols. Methanol and ethanol are commonly used to estimate and extract total proteins and carbohydrates.
The radicals scavenging activity of different fractions of Acromitus hardenbergi was evaluated using DPPH·, ABTS·+, and ·OH scavenging assays measured using ESR spectrometry (Fig. 2). Generally, DCM and H2O fractions from both JB and JOA of the jellyfish exhibited the highest DPPH· scavenging activity (p < 0.05). DPPH· scavenging activity of JB fractions followed the order: DCM ≥ H2O > CHCl3 > PE > MeOH whereas the activity of JOA fractions followed the order: DCM ≥ CHCl3 ≥ H2O > PE ≥ MeOH (p < 0.05). A conclusive finding could be observed in ABTS·+ and ·OH scavenging assay whereby H2O fractions from both JB and JOA of A. hardenbergi displayed significantly higher (p < 0.05) radical scavenging activity than any other fractions; and were found to have scavenged the radicals almost completely. ABTS·+ and ·OH scavenging effect of JB fractions followed the order: H2O > DCM > CHCl3 > PE > MeOH whereas the activity of JOA fractions followed the order: H2O > PE > CHCl3 > DCM > MeOH (p < 0.05). ABTS·+ scavenging assay has been reported to be more efficient in evaluating the antioxidant activity of a sample than DPPH· radical scavenging assay as it is sensitive and produces more consistent results over a wide range of pH and media (Chan et al., 2012b; Chan et al., 2012c; Lemańska et al., 2001). For all three assays, the lowest radical scavenging activity was observed in the MeOH fraction of JB and JOA (p < 0.05).
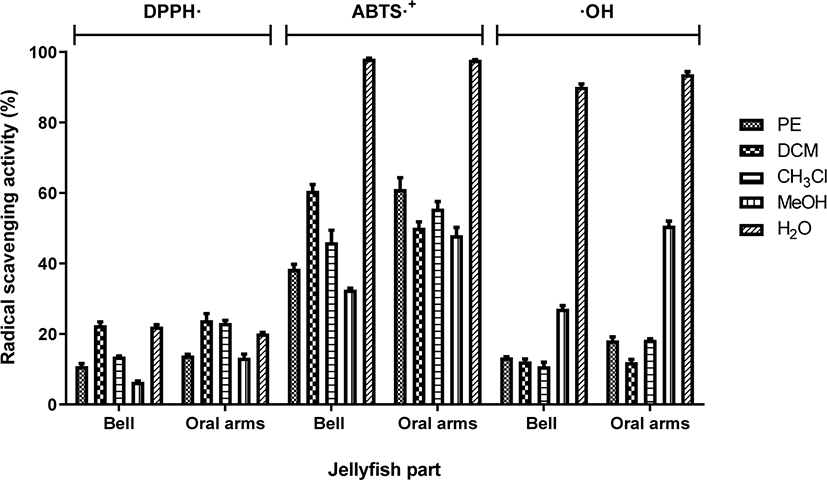
The study concurs with the findings of Morais et al. (2009) where it was reported that the jellyfish, Catostylus tagi exhibited low reducing power and moderate DPPH· scavenging activities. The same study also found that the jellyfish scavenge hydroxyl radical (·OH) better than ascorbic acid. A few reports showed positive correlations between the antioxidant activities exhibited by jellyfish to their protein content (Yu et al., 2006; Zhuang et al., 2010). Thus, it is postulated that the radical scavenging activity from jellyfish were contributed by the content of –SH and antioxidant enzymes such as superoxide dismutase, catalase and peroxidise. This study also detected exceptionally good ABTS·+ and ·OH radical scavenging activity (< 97% scavenging activity) in the H2O fraction of jellyfish whereby polar compounds such as amino acids and proteins are usually extracted. Having strong hydrophilic antixodant is very advantageous as water extraction is a cheap, safe, and green. This may also justify to some anti-aging and anti-arthritic claims of this jellyfish by the locals as the jellyfish is most commonly boiled in soup or eaten raw as means of its cooking preparation in regular households.
Although the yield of DCM fraction was found to be less than 2% (w/w), the fraction also showed relatively high antioxidant potential. The radical scavenging activity of DCM fraction in JOA was significantly higher than that of PE, CH2Cl and MeOH fraction (p < 0.05) whilst in JB, DCM fraction was significantly higher than PE and MeOH fraction (p < 0.05). The opposite could be seen in the MeOH fraction where although it was the fraction with the highest yield, it gave the lowest free radical scavenging activity among other four fractions (p < 0.05).
In general, A. hardenbergi performed modestly in the inhibition of linoleic acid peroxidation as compared to their radical scavenging effect (Fig. 3). The ability of fractions from JB to inhibit linoleic acid oxidation followed the sequence of CHCl3 > PE > DCM ≥ H2O > MeOH (p < 0.05), while JOA fractions followed the sequence: PE > DCM > CHCl3 > H2O > MeOH. (p < 0.05). PE or hexane and CHCl3 are common solvents used routinely for the extraction of total crude lipid, fats, oils and possibly sterols such as tocopherol, carotenoids and Co-Q10 which were lipophilic antioxidants. The findings aligned with that of Yu et al. (2006) who reported that jellyfish extracts with appreciable radical scavenging activity hardly exhibited any inhibition of β-carotene bleaching. Moreover, extraction temperature may play an important role in even effective antioxidant extraction from food matrices. Some studies observed that hot water extract produced stronger antioxidant capacity than cold water extracts (Chen & Yen, 2007). Additionally, MeOH fractions from both JB and JOA exhibited pro-oxidative activity despite presence of radical scavenging activity discussed earlier in DPPH·, ABTS·+, and ·OH scavenging assays. Khan et al. (2012) proposed that prooxidant actions are often important indicators of anticancer and apoptosis properties which might occur to some well-known antioxidative ingredients. As dried A. hardenbergi are composed mainly of minerals (ash) and proteins (Khong et al., 2016), thus transition ions (such as ferrous ion) and prooxidative enzymes such as lipoxygenase) might be the major contributors to the pro-oxidative activity. These mineral ions and proteins tightly bound to each another is usually unable to react with tissue components thus may have been liberated or broken down during the fractionation procedure. A study by Cherrak et al. (2016) revealed that antioxidants have both prooxidant and antioxidant activity depending on the nature,concentration and interactions of the antioxidants and metal ions.

The mean values of total reducing capacity in JB fractions varied from 0.01 to 0.24 mg GAE/ g of fraction whereas in JOA ranged from 0.09 to 0.32 mg GAE/ g of fraction, depending on extraction solvents (Table 2).
PE and DCM fractions of both JB and JOA showed significantly higher (p < 0.05) total reducing capacity than the rest of the fractions while CHCl3 fraction was the lowest. Till date, there is a lack of information on the total phenolic compounds of jellyfish fractions. Feeding properties may play an important role in the levels of phytochemicals and reductants in animal tissues. It is commonly observed that total reducing capacity or total phenolic content peaked in digestive tract tissues of animals or insects as these organisms may have derived reductants (phenols) from plants they consumed or acid hydrolysis may release bound phenolic compounds (Chan et al., 2012b; Chan et al., 2012c; Suh et al., 2010). Leone et al. (2019) also found that antioxidant and total phenolic content of jellyfishes varied under different thermal processing.
The antimicrobial activity of different fractions extracted from JB and JOA at 1.0 mg/ mL is presented as diameter (mm) of inhibitory zones in Table 3. Overall, the lipophilic extracts of both JB and JOA especially PE fraction, was found to exhibit high antimicrobial activity. Lipophilic fraction of JB was found to be inhibitive to E. coli and E. faecalis whereas JOA to E. coli, B. cereus, B. subtilis, E. faecalis, S. aureus, S. cerevisiae, and C. albicans. Clearly, the antimicrobial property of the lipophilic fractions of JOA were found to inhibit the growth of more pathogens compared to JB; and only JOA was found to be antifungal. The findings in this study aligned to that of Suganthi & Bragadeeswaran (2012) where extract from the tentacles of the jellyfish Chrysaora quinquecirrha were also found to inhibit the growth of bacterial and fungal pathogens and the authors attributed these properties to the venom of the animal. Previously, aurelin isolated from the invasive moon jellyfish, Aurelia aurita was also reported to be active against E. coli (Ovchinnikova et al., 2006). Then again, Morales-Landa et al. (2007) found no activity in the cnidarian extracts (including 2 jellyfishes, Cassiopea xamachana and Carybdea marsupialis) against Gram positive and Gram-negative bacteria tested at a concentration of 0.01 mg/ mL.
In this study, the most potent antimicrobial potentials of the river jellyfish were found in the PE fraction from JOA. The PE fraction of JOA is found to be particularly effective in inhibiting the growth of Gram-positive bacterias tested i.e., B. cereus, B. subtilis, E. faecalis, and S. aureus. The first report of antibacterial and fungistatic properties from jellyfish was that of Betz (1988) in the study of the cannonball jellyfish, Stomolophus meleagris where the active is found to be a squalene containing, neutral terpenoid fraction. Naturally, chemical defenses are available in every living organism especially marine organism that exist in an environment populated with diverse microbial ecology. Lipidic compounds such as brominated C16, C18, and C20 acetylenic acid have been reported to inhibit bacteria involved in food spoilage (Dembitsky & Srebnik, 2002). Jellyfish have been described to be rich in polyunsaturated fatty acids (PUFA) particularly alpha-linolenic acid, eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA) and they also have been reported to possess rare fatty acids such as trans-6-hexadecenoic acid and 7-methyl-7-hexadecenoic acid as well as the unusual very long chain PUFA, tetracosahexaenoic acid (THA, 24:6n-3) and the rarely reported tetracosapentaenoic acid (24:5n-6) (Nichols et al., 2003). Eventually, antimicrobial actives from A. hardenbergi in this study were found to be lipophilic in nature and it is highly suspicious that this characteristic is for adapted for their role in chemical defense in aquatic, highly diluted habitats. On the other hand, disc saturated with H2O fractions from both JB and JOA were observed not only inactive in inhibiting test microorganisms but encouraged the growth of bacteria. Previously, Condon et al. (2011) reported that the organic matters of jellyfish are likely to contain a combination of polysaccharides and other C-rich compounds that are highly bioavailable to heterotrophic bacteria. The possible presence of antibacterial and antifungal agents within the lipophilic fractions of jellyfish would be the subject of further study to fill in the demand for efficient and safe preservatives in the food industry.
Table 4 provide a description of all the characteristic bands present in the PE, DCM, CHCl3, MeOH, and H2O fractions of JB and JOA, along with the related vibrations of particular functional groups. From the FTIR spectra in PE fraction, both JB and JOA indicated strong signal for the presence of carboxyl group (-COOH) and -CH bond at 1,300–1,000 cm–1 and 3,000–2,850 cm–1 respectively. It was observed hydroxyl group (-OH) and amide bond (-NH) were absent. This is because PE is hydrophobic thus does not solubilize hydrophilic functional groups. Interestingly, at peak position 722.82 cm–1 of PE of JOA, overlapping of CH2 rocking out of plane vibration of cis-disubstituted oleofins at 721 cm–1 were detected. This is a characteristic of long chain fatty acids (Mahesar et al., 2011). Moreover, the detection of C-X chlorine, bromine (785–540 cm–1) in PE also goes very well with the previous assumption that brominated lipidic compounds may be contributing to the antimicrobial properties of the lipophilic jellyfish fractions in this study alike that performed Dembitsky & Srebnik (2002). As for DCM fraction of both JB and JOA, polar lipids were dissolved in this fraction as functional group -COOH and -CH were found to be present. Aside from this, the ketose group (-C=O), N-H bend, and methyl group (-CH3) were detected indicating a group of alkaloids, known to have these functional groups were found present. Whereas in the CHL fraction, the functional groups responsible for alkaloids were found to be present in both JB and JOA. The detection of infrared spectra at 1,680–1,630 cm–1 suggested that MeOH was able to extract polysaccharides from jellyfish - seen with the presence of the ketose group (-C=O) and hydroxyl group (-OH) in both JB and JOA. This supports our previous supposition that the H2O fractions contains sugars that support microbe growth. The presence of the -OH and -COOH group may indicate presence of aglycones, a non-sugar compound, while the -S=O functional group confirms the presence of amino acids within this fraction. The H2O fraction was the most polar fraction among all and is predicted to solubilize proteins. This is further evidenced in the FTIR spectra which indicates signals at the -CH2, -NH2 (amide bond) and -S=O bond at both JB and JOA and bell. The S=O bond is an indication of disulphide bonds within amino acids especially cysteine and methionine (Karimi et al., 2016). There were some noise in the H2O fraction of bell which may largely be due to water vapour peaks that causes the interference. During this case, it is advised to ensure that samples are completely free of water vapour to minimize unwanted interference.
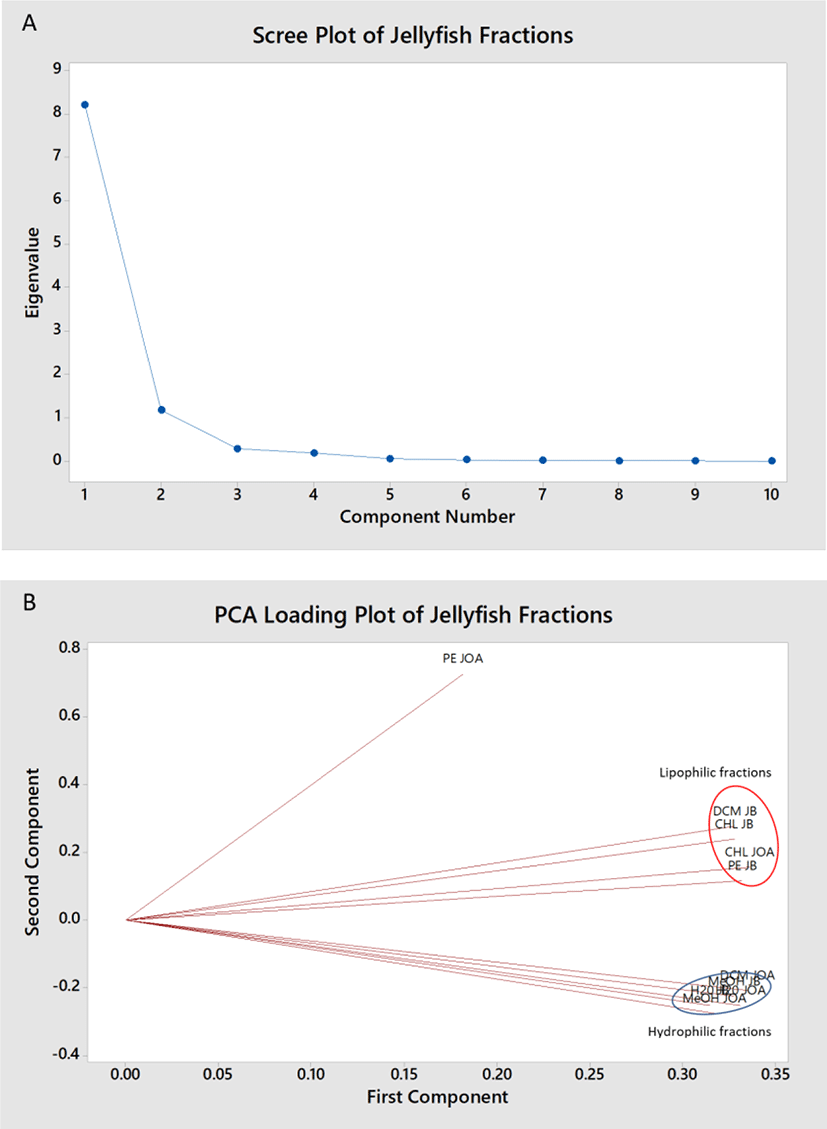
Next, PCA was used to compress and process the entirety of the FTIR data from the spectral range of 280–4,000 cm−1 (Fig. 4). The first principal component accounts for 82.1% of the total variance where all variables (all fractions from JB and JOA) was found to correlate positively with the first principal component in the range of 0.300 except PE fraction of JOA (0.182). Therefore, data from this first principal component would equally explains 82.1% of the variation in the data (Fig. 4A). Fig. 4B showed two distinct groups i.e. PE, DCM, and CHCl3 fractions in one and H2O and MeOH fractions in another. Comparing this to the antioxidant and antimicrobial activities aforementioned, it is rather interesting to note that this two groups also signifies the groups exhibiting high antioxidant activities (H2O fractions) and high antimicrobial activities (PE and DCM fractions). PE fraction from JOA was found to deviate from both groups, but relatively contains functional groups closer to the lipophilic fractions than the hydrophilic fractions. Clear enough, the PE fraction from JOA is the only fraction found to exhibit antifungal property and was capable of inhibiting the growth of all four Gram-positive bacteria tested besides highly inhibitive to E. coli. This deviation may also explains the effect it brings to the subsequent extract i.e. DCM than follows during the sequential solvent extraction process. Lipophilic fractions (PE, DCM, and CHL) collectively shows vibrations corresponding to the -CH3 and -COOH groups involving peaks within the ranges of C-H alkanes stretch (3,000–2,850 cm–1), C=O aldehyde (1,740–1,720 cm–1), CH3 bend (1,450–1,375 cm–1), C-O alcohols or esters (1,300–1,000 cm–1), C-N amines (1,350–1,000 cm–1), and interestingly C-H aromatics (900–690 cm–1) and C-X chlorine, bromine (785–40 cm–1). This is in line with Matwijczuk et al. (2019) that concluded that infrared spectra located within the ranges of 1,720–1,500 cm−1, ~1,426 cm−1, 1,369 and 1,237 cm−1 are good markers which are useful in the monitoring of the physicochemical changes in oils that commonly related to the intensification of bands reflecting the vibrations of C–C, C=C, and C=O groups. In contrast, hydrophilic fractions (MeOH and H2O) shares vibration corresponding to the -OH,-CH2, -NH2 (amide bond) and -S=O groups which are indicative of proteins and peptides. Notably, the -S=O group within the MeOH and H2O fractions could potentially be key that influences the antioxidant-prooxidant activity of the fractions. Karimi et al. (2016) demonstrated that some protein disulfides exist in strained conformations that are abundant in proteins with catalytic disulfides or those that have allosteric actions and the reactivity of protein disulfides is dramatically affected by local structure and conformation. This explains that although the jellyfish MeOH and H2O have similar sets of functional groups, MeOH fractions was found to prooxidised linoleic acid while H2O fractions protects them from oxidation, significantly (p < 0.05).
The biochemistry of macromolecules is determined by their functional groups. Presence and arrangement of each functional group in a molecule changes the properties and its functionality. For example, carbohydrates consist of carbon, hydrogen, and oxygen each when arranged differently produces glucose or fructose (sweeter) by a mere difference of position of carbonyl (-C=O) group at end and middle respectively. Another example is the biochemistry of lipids. Lipids are made of fatty acids, long chains carbon and hydrogen with a carboxyl (-COOH) attached to an end. Interestingly, presence of double bonds within the long chain determines the unsaturation of the lipid molecule. Finally, proteins contain a distinct functional group that sets it apart from the lipids and polysaccharides, that is, presence of the amine group (-NH2). With modern spectroscopy and the instrument FTIR in particular, functional group belonging to different classes of macromolecules can be distinguished. Infrared spectra are commonly utilised to hint on the type of molecules present in an unknown sample. Peak positions are used as an indicative of a molecular structure much like a fingerprinting technique where not only are the spectra generated fast, easy and inexpensive, it is sensitive, needing relatively small amounts of samples and producing reproducible results (Franca & Oliveira, 2011; Kumar et al., 2019). In other words, infrared spectroscopy is an excellent chemical analysis technique and is the perfect tool for analysing unknown compounds.
Conclusion
Antioxidants and antimicrobials from Acromitus hardenbergi bell (JB) and oral arms (JOA) were distributed into fractions according to their polarity using different solvents. Of the five fractions obtained, H2O fraction showed significant level of DPPH·, ABTS·+, and ·OH scavenging activity. CH3Cl fraction from JB and PE fraction from JOA exhibited the highest antioxidant activity in BCB assay. PE fraction were found to exhibit antimicrobial effects towards Gram-negative and Gram-positive bacteria, highlighting its potential in the improvement of food safety. Notably, the PE fraction from JOA inhibited E. coli, B. cereus, B. subtilis, S. aureus, E. faecalis, and the two yeasts, S. cerevisiae, and C. albicans. ATR-FTIR spectra of JOA and JB clearly discreted all fractions into two groups i.e. PE, DCM and CH3Cl into one and MeOH and H2O the other. Spectra found in the PE and DCM fractions indicated presence of lipids, CH3Cl alkaloids, MeOH polysaccharides and H2O fraction proteins and peptides. Overall, this study demonstrates the polar nature of antioxidants and apolar nature of antimicrobials and lipid protectant in JB and JOA of A. hardenbergi. Jellyfish powder could be a potential food ingredient and nutraceutical for the formulation of medicated diets and functional food for the enhancement of human well-being.








