Introduction
In 1984, the term “functional food” first appeared in Japan. The government of Japan introduced a new food group to their market, Food for Specific Health Uses, as “food containing an ingredient with functions for health and officially approved to claim their physiological effects on the human body” and produced a devoted statutory framework (Stanton et al., 2005). According to the general definition, functional foods can improve the overall conditions of the body (pre and probiotics), able to reduce the risk of noncommunicable diseases (diabetes), and could even be used to treat diseases (Cencic & Chingwaru, 2010). Typically, food marketed as functional is usually enriched with, technologically developed ingredients with some specific health benefits to its consumer other than its nutritional value. Even though “functional foods” have already been defined several times, there has not yet been a unified definition for functional foods (Alongi & Anese, 2021; Siró et al., 2008). Taken together, generally, functional food can be defined as “natural or processed food products that contain identified or unidentified bioactive compounds; which, in defined, effective, and non-toxic amounts, provide a clinically proven and reported health benefit for the prevention, management, or treat to chronic diseases”.
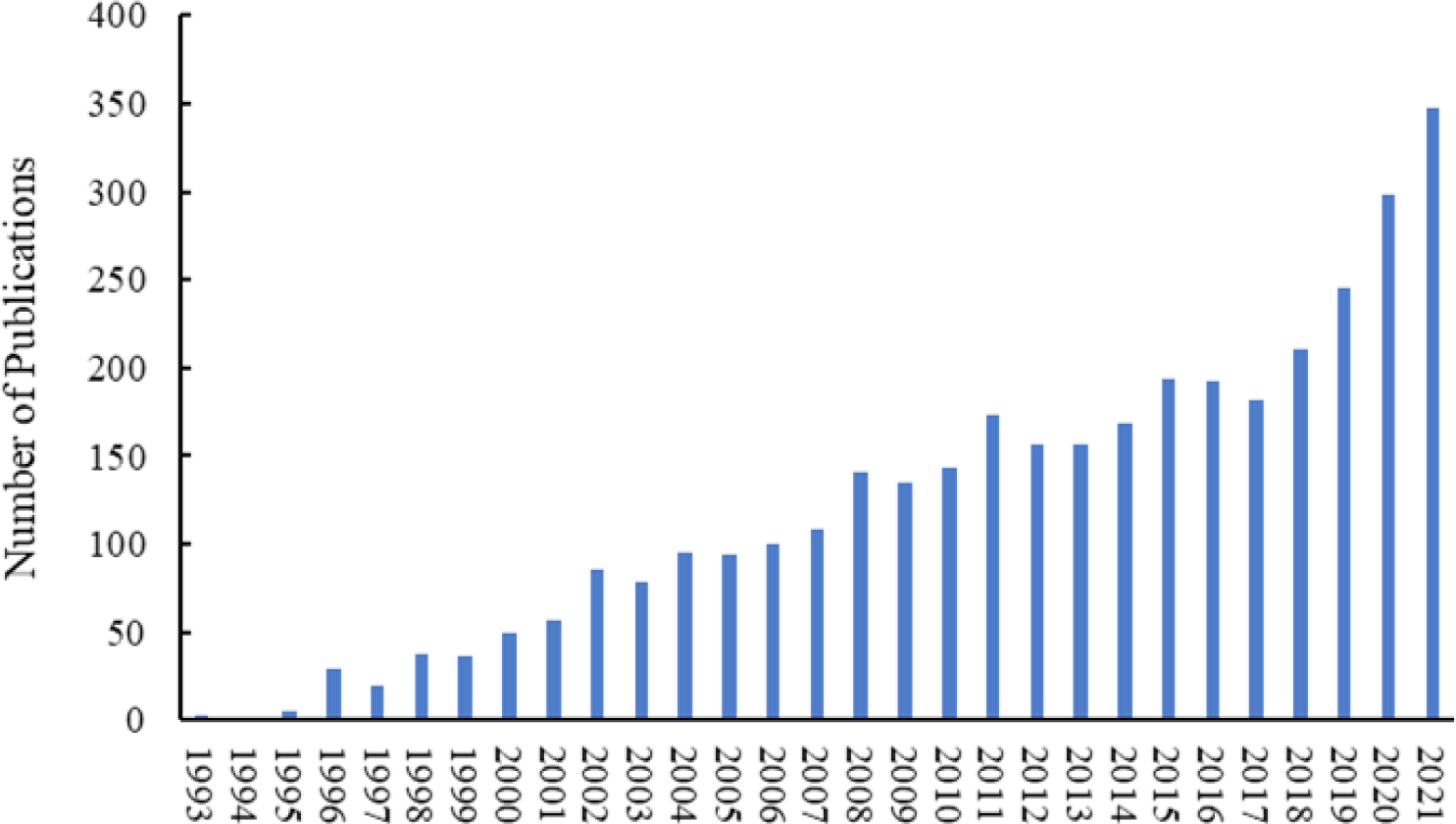
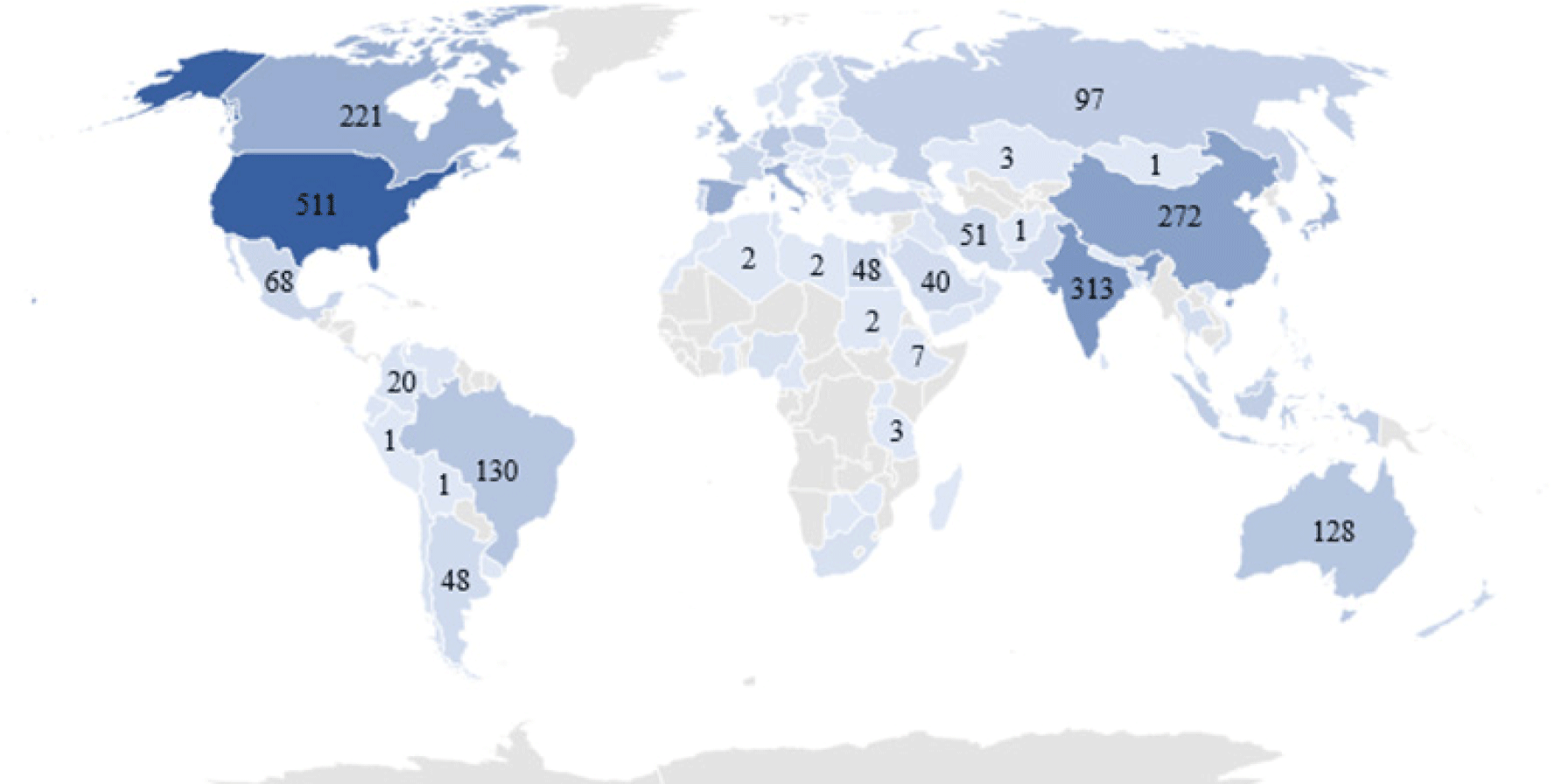
As shown in Fig. 1 research interest towards functional foods is increasing and from 1984 to 2021 more than 3,500 articles were recorded in the SCOPUS database just with the title “functional food”. In addition, out of 3,500 documents around 450 articles are reported about functional food applications or the potential of marine bioresources to develop functional foods. Besides that, more than 30% of the total published articles were published by the United States, China, and India (Fig. 2). These findings demonstrate the future potential for the functional food industry and the potential markets for sea cucumber.
Sea cucumbers are a group of ancient marine invertebrates of the class Holothuroidea and phylum Echinodermata and are widely distributed all around the world (Zhao, 2015). Sea cucumbers live on ocean floors and play a significant role in marine ecosystems. It is believed that sea cucumbers get their foods from eating marine sediments and filtering seawater, which provides a unique, fertile environment for marine microbes such as bacteria and fungi to thrive (Xia et al., 2022). Sea cucumbers also known called holothurians or holothuroids, have traditionally been classified through their morphological phenotype, particularly the analysis of their anatomy, including papillae, tentacles, and ossicles (Mohsen & Yang, 2021; Zhao, 2015). According to the literature, there are around 1,500 species of sea cucumbers reported and which are separated into 3 subclasses (Apodacea, Aspidochirotacea, and Dendrochirotacea). Moreover, these ancient marine invertebrates are separated into six orders as Aspidochirotida, Apodida, Elasipodida, Dactylochirotida, Dendrochirotida, and Molpadida, Dactylochirotida (Bordbar et al., 2011).
According to the statistics, sea cucumber fisheries worldwide have expanded annually in terms of value and catch. However, the harvest of sea cucumbers not directly comes from the wild harvest, considerable amount of harvest coming from aquaculture. Mostly these harvested sea cucumbers are mostly used in the food industry and folk medicine (Toral-Granda et al., 2008). Specifically, these sea cucumber-based food items are popular in the countries such as Korea, China, Hong Kong, Japan, Indonesia, and Malaysia (Dai et al., 2021; Eriksson & Clarke, 2015; Pangestuti & Arifin, 2018). According to the literature, Japanese sea cucumber (Stichopus japonicus) has been produced in Japan and China since, the 8th and 16th, centuries respectively (Scanes, 2018).
According to previous studies, sea cucumbers have negligible amounts of fat and cholesterol, and good source of essential nutrients (Hossain et al., 2020; Janakiram et al., 2015). Specifically, sea cucumbers are rich in proteins and lipids (collagen, omega-3, and -6 fatty acids) vitamins (including Vit A, riboflavin, thiamine, niacin), and minerals (calcium, magnesium, iron, and zinc) (Hossain et al., 2020). Besides the nutritional value, sea cucumber species such as Actinopyga mauritiana,Thelenota anax, Stichopus hermanni, Thelenota ananas, and Holothuria fuccogilva are the most highly-valued sea cucumber species around the globe and are potential marine organisms to mine bioactive secondary metabolites with functional properties (Pangestuti & Arifin, 2018). Furthermore, the compounds isolated from sea cucumbers (saponins, glycosaminoglycans, chondroitin sulfate, sulfated polysaccharides, phenolics, peptides, lectins, cerebrosides, sterols) were found to possess many bioactive properties, such as anticoagulant, antimicrobial, antioxidant, anti-inflammatory, anticancer, antiangiogenic, and antihypertensive properties that are ideal properties to develop functional foods (Aydın et al., 2011; Dai et al., 2021; Hossain et al., 2020; Kim & Himaya, 2012; Li et al., 2008; Song et al., 2013).
As far as we know, previously no comprehensive review article as such has ever been published to address the research gap between sea cucumbers to develop functional foods and their bioactive properties. The purpose of this review is to assemble an exhaustive report that covers the components and the biological properties of sea cucumbers, as one of the potential sources of functional foods.
Cancer is a group of diseases in which cells divide continuously and excessively (Matthews et al., 2022). According to the literature, cancer can evolve mostly from the mutations that take place in somatic cells, and some mutations are inherited, emerge randomly, or develop due to the effects of mutagens (Nussinov et al., 2021). Mutations in the oncogenes with the dominant gain of function over tumor suppressor genes and malfunction of tumor suppressor genes are known as the key cause of cancer pathogenesis (Hanahan & Weinberg, 2000). Annually, millions of new cancer patients are identified and the annual health reports related to cancers indicate the sharp growth of cancer patients and cancer-related death rates (14.1 million new cancer patients in 2012 and 19.3 million new cancer patients in 2020) (Ferlay et al., 2015; Ferlay et al., 2021). According to Ferlay et al. (2021) new cancer incidence and mortality rates for 2020 were estimated for 38 cancer sites and 185 countries or territories worldwide. There were an estimated around 19.3 million new cases and almost 10.0 million life losses from cancer worldwide in 2020 alone. The most commonly diagnosed cancers worldwide were female breast cancer (2.26 million), lung cancer (2.21 million), and prostate cancer (1.41 million). Besides that, in 2020 the most common causes of cancer death were lung cancers (1.79 million), liver cancers (0.83 million), and stomach-based cancers (0.769 million) (Ferlay et al., 2021). According to the previous research studies, initiation of apoptosis, inhibiting the colony formation ability of cancer cells, inhibiting the angiogenesis potential of cancer cells, activating inflammatory responses in cancer cells and enhancing anticancer efficacy of chemotherapeutic agents are some possible approaches to inhibit/avoid cancer cell pathogenesis (Sanjeewa et al., 2017). The foods that naturally carry the aforementioned properties undoubtedly can be considered as functional foods to avoid cancer cell pathogenesis in the human body. Many studies highlighted the anticancer properties of sea cucumbers and the potential of sea cucumber derivied bioactive metabolites to use in different industries such as functional foods and nutraceuticals. In this section, the authors are mainly focused to collect recent literature reported about anticancer properties of sea cucumbers.
Saponins are bioactive secondary metabolites reported animals, such as sea cucumbers, and terrestrial plants, such as ginseng (Yi, 2021). The saponins of sea cucumbers are reported to play a vital role in their chemical defense against predators and also saponins regulate the reproduction of sea cucumbers. Besides these activities, saponins in sea cucumbers are found to possess anticancer activity against different cancer cell lines (Dai et al., 2020). According to Dai et al. (2020) saponins in sea cucumbers including Apostichopus japonicus are able to stimulate apoptosis in cancer cell lines such as B16F10, HL-60, Hep3B, and MCF-7. According to the author sea cucumbers, extracts with high saponin contents were able to decrease the growth of the aforementioned cancer cells, via downregulating B-cell lymphoma (Bcl) extra-large and upregulating caspase-3 and 9, Bcl-2-associated X protein, polymerase cleavage, and apoptosis.
A triterpene saponin (also known as stichoposide D) isolated from the Stichopus chloronotus was also found to possess strong anticancer properties against NTERA-2 (half-maximal inhibitory concentration [IC50] 0.26 ± 0.02 µM). According to the authors stichoposide D (Fig. 3) treated NTERA-2 cells had upregulated expression of caspase-3 compared to the control treatment (Cuc et al., 2020). Furthermore, their cell cycle analysis confirmed that stichoposide D was able to arrest cells at the sub-G1 phase (15%) and S-phase for DNA synthesis in NTERA-2 cells. Mohomed et al. (2021) also reported the anticancer effects of saponin separated from Holothuria arenicola against Ehrlich ascites cancer cells in swiss albino mice. According to the authors, saponin treatment had a significant inhibitory effect on Ehrlich ascites carcinoma cells and improved activities in the antioxidant system, liver function, and kidney functions in saponin treated mice group. Nursid et al. (2019) also observed the anticancer properties of saponin-rich fractions extracted from different sea cucumbers collected from Tilamuta village, Boalemo district, Gorontalo-Indonesia. According to the authors Holothuria atra and Bohadschia marmorata had strong cytotoxicities against human breast ductal carcinoma cell lines with IC50 values of 23.0 and 28.1 ug/mL, respectively (Nursid et al., 2019). Other than Nursid et al. (2019), Hawas et al. (2021) also report the anticancer properties of saponins isolated from H. atra. According to the authors crude ethanol extract of H. atra also found to possess weak anticancer activities against Ehrlich ascites carcinoma cell lines. In addition, the authors also reported the structure of the saponins four identified during the study (24-dehydroechinoside B, echinoside B 12-O-methyl ether; echinoside B, and holothurin B).
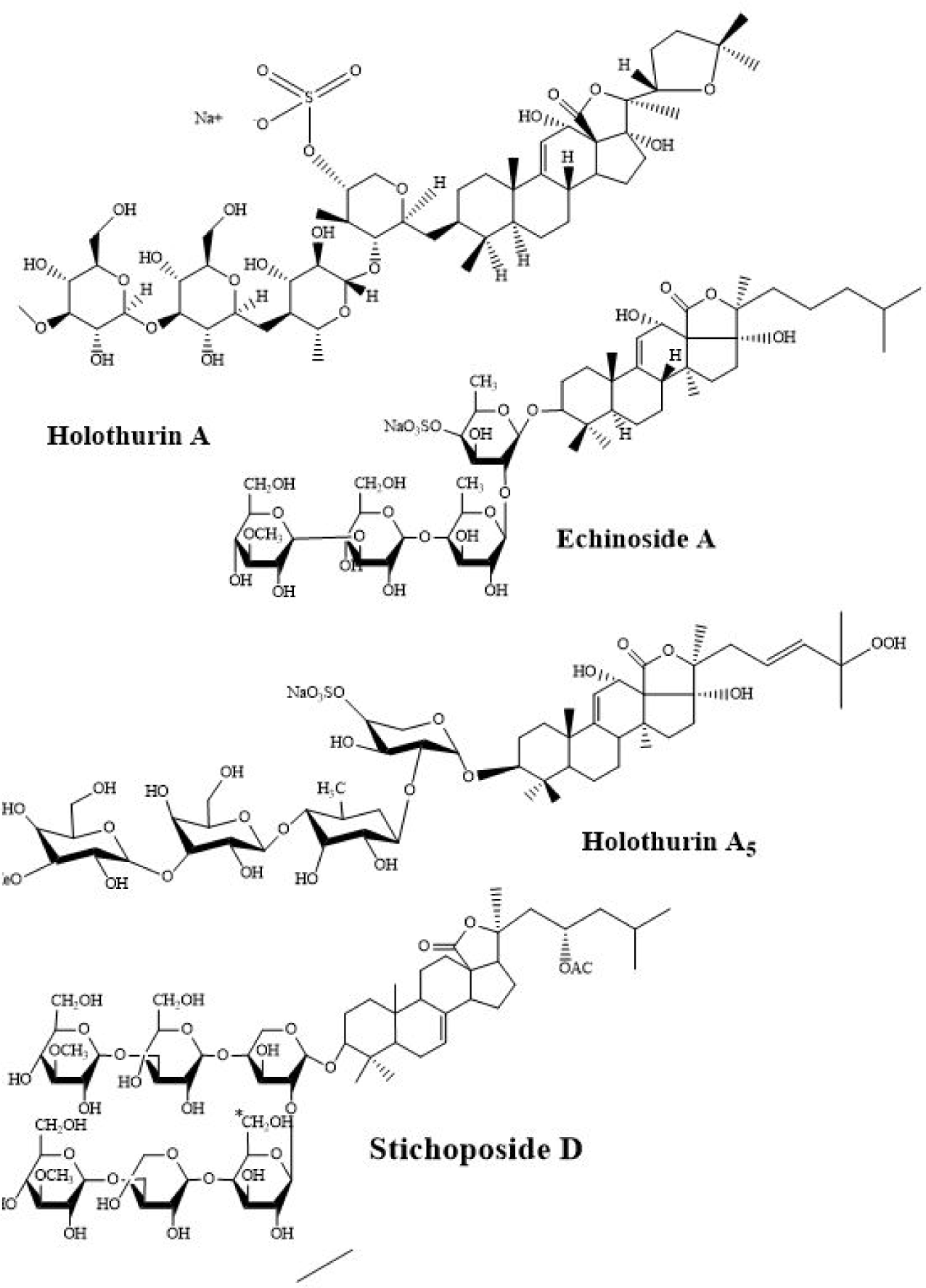
Besides the saponins, proteins separated from sea cucumbers are also found to possess promising anticancer properties. Specifically, several studies highlighted the anticancer potential of sea cucumber protein extracts. Recently, Ru et al. (2022) reported the anticancer activities of protein isolated from sea cucumbers. According to the authors, protein isolated from Holothuria leucospilota was found to possess a cytotoxic effect against tested cancer cells including A549, HepG2, and Panc02 (Ru et al., 2022).
In addition to the interesting research studies discussed the above anticancer properties of the coelomic fluid collected from Holothuria tubulosa against MDA/MB-231 breast cancer cells were reported by Luparello et al. (2019). According to the authors, H. tubulosa coelomic extract was found to restrict activities of MDA/MB-231 via cell cycle arrest, altering mitochondrial transmembrane potential, and stimulating cellular autophagy. Furthermore, H. tubulosa coelomic extract was found to restrict activities of beclin-1, and total LC3 and LC3-II under the tested conditions (Luparello et al., 2019). In addition, aqueous extracts prepared from Actinopyga lecanora, H. atra, and Stichopus vastus were also found to possess promising anticancer properties. According to the authors, all three species induced apoptosis in T-47D breast cancer cells with the IC50 values of 3.25 µg/mL (S. vastus), 6.25 µg/mL and (A. lecanora), 7.5 µg/mL (H. atra) (Santhanam et al., 2022). Anticancer properties of methanol extract of H. leucospilota against breast carcinoma cell line (SKBR3) were reported by Khaledi et al. (2022) in a recent study. According to the authors, H. leucospilota treatment induced the apoptosis of the SK-BR-3 cells, via upregulating DNA fragmentation, and arrested G2/M phase. In addition, treatment of the methanolic extract to cancer cells are caused to inhibit the expression of Bcl-2 protein, which is a well-known antiapoptosis protein. In addition, the authors reported the upregulated expression of caspase-3, -7, and Bax, in SKBR3 cells after early exposure to the preparations. Yasman et al. (2020) also confirmed the anticancer properties of 24-dehidroechinoside and holothurin A using in silico research model. According to the authors, holothurin-A blocked the activities of the breast cancer cells via down-regulating progesterone receptor, fibroblast growth factor receptor 1, estrogen receptor alpha, vascular endothelial growth factor receptor 2, and insulin-like growth factor 1 receptor. Anticancer properties of the peptides identified from Cucumaria frondose were also confirmed by Wargasetia et al. (2021) using in silico model. According to the authors, two peptides (WPPNYQW and YDWRF) identified from sea cucumbers were found to inhibit activities of AKT1, EGFR, P13K, and CDK4 in the tested cancer cells (Table 1). Other than the saponins and peptides, triterpene glycosides are also considered important secondary metabolites of sea cucumbers. Triterpene glycosides isolated from sea cucumbers are reported promising bioactivities against different cancer cell lines. A recent study carried out with triterpene glycosides isolated from Psolus chitonoides were also found to possess anticancer activities against several cancer cell lines such as colorectal adenocarcinoma DLD-1 and adenocarcinoma HeLa cells (Silchenko et al., 2021). According to the authors, ED50 of isolated triterpene glycosides had similar effects compared to the positive control (cisplatin) used in their study. Hoang et al. (2020) also reported the anticancer activities of eight compounds isolated from Holothuria edulis. According to the authors, among the isolated compounds holothurin A5 (triterpene glycosides; also known as saponins) was found to possess cytotoxic activity against five human cancer cells including epidermoid carcinoma cancer cells (KB), hepatoma cancer cells, prostate cancer cells, MCF7 breast cancer cells, and SK-Mel2 melanoma cancer cells under the tested conditions (Hoang et al., 2020).
| Name | Major bioactive compound | Cell line/animal model | Mechanisms | Reference |
|---|---|---|---|---|
| Actinopyga lecanora | Saponins Triterpene glycosides |
T-47D breast cancer cells | Apoptotic induction | Santhanam et al., 2022 |
| Apostichopus japonicus | Saponins | HL-60, B16F10, MCF-7, and Hep3B | Apoptosis | Dai et al., 2020 |
| Bohadschia marmorata | Saponins | Human breast ductal carcinoma cell | Apoptotic induction | Nursid et al., 2019 |
| Cucumaria frondosa | Isolated peptides | Breast cancer cells | Inhibit activities of EGFR, P13 K, AKT1, and CDK4 | Wargasetia et al., 2021 |
| Holothuria arenicola | Saponins | Swiss albino female mice (ascites carcinoma tumor model) | Suppress tumor growth | Mohamed et al., 2021 |
| Holothuria atra | Saponins Triterpene glycosides |
Human breast ductal carcinoma cell, Ehrlich ascites carcinoma cell lines, T-47D breast cancer cells | Cytotoxicity and apoptotic induction | Hawas et al., 2021; Nursid et al., 2019; Santhanam et al., 2022 |
| Holothuria edulis | Holothurin A5 | Hepatoma cancer, epidermoid carcinoma, LNCaP prostate cancer cells, MCF7 breast cancer cells, and SK-Mel2 cancer cells | Cytotoxicity | Hoang et al., 2020 |
| Holothuria leucospilota | Protein | HepG2, A549, Panc02 | Inhibition of cancer cell immigration | Ru et al., 2022 |
| H. leucospilota | Methanolic extract | SK-BR-3 cells | Apoptosis | Khaledi et al., 2022 |
| Holothuria tubulosa | Coelomic fluid | MDA-MB231 breast cancer cells | Impairment of cell cycle progression | Luparello et al., 2019 |
| Isolated compound ***Name of sea cucumber not specified |
Holothurin A | Breast cancer cells | Inhibit ER-α, FGFR1, VEGFR2, and PRIGFR1 receptors in breast cancer cells | Yasman et al., 2020 |
| Psolus chitonoides | Triterpene glycosides | HeLa and colorectal adenocarcinoma DLD-1 | Cytotoxicity | Silchenko et al., 2021 |
| Stichopus chloronotus | Stichoposide D | NTERA-2 cells | Apoptotic induction | Cuc et al., 2020 |
| Stichopus vastus | Saponins triterpene glycosides | T-47D breast cancer cells | Apoptotic induction | Santhanam et al., 2022 |
Taken together these results are suggesting the anticancer potential of sea cucumbers under in vivo and in vitro conditions. The compounds available in sea cucumbers such as saponins, and proteins are promising agents to be included in functional foods.
Worldwide, thromboembolic diseases cause a substantial number of deaths and disabilities (Li et al., 2021). In addition, pandemics like COVID-19 outbreaks also highlighted the importance of the development of effective anticoagulative agents to treat COVID-19. According to previous studies, uncontrolled blood clotting and increased micro-thrombus formation have been identified as one main cause of organ dysfunction and cause for death in infected patients (Gozzo et al., 2020). Thus, anticoagulants play important role in people who at a high risk of getting clots, and minimize their possibility of developing serious health issues like strokes and heart attacks. Therefore, foods with anticoagulative properties can also be considered as functional foods and those can effectively reduce risk of the thromboembolic diseases. Several metabolites such as fucoidans isolated from sea cumbers were found to possess promising anticoagulant properties (He et al., 2020; Li et al., 2021; Mansour et al., 2019; Zheng et al., 2019).
The value of sea cucumbers specifically dried products of sea cucumber (bêche-de-mer) depends on several factors such as weight, color, length, smell, etc. (Wen et al., 2010). Underutilized sea cucumber species such as Acaudina leucoprocta have low demand compared to commercial sea cucumber (Apostichopus japonicas) as they have less market demand due to the fishy smell. However, He et al. (2020) report the anticoagulant properties of simple sulfated fucan (→ 3)-α-L-Fucp-(1→) separated from A. leucoprocta, collected from the East China Sea. According to the authors, isolated fucan was found to possess selective anti-thrombin activity through heparin cofactor II regulation, with a mechanism of action different from that of heparin (He et al., 2020). In addition, Zheng et al. (2019) also reported the anticoagulant properties of two sulfated polysaccharides isolated from Pattalus mollis. According to the results, both sulfated polysaccharides demonstrated robust activated partial thromboplastin time (APTT) prolonging intrinsic factor IXa–VIIIa complex (Xase) inhibition activities (Zheng et al., 2019). Intrinsic Xase (Xase), is the last and rate-limiting enzyme complex in the intrinsic coagulation pathway and is also considered an ideal target for antithrombotic treatment (Sun et al., 2020). Therefore, studies focused on anticoagulation results mainly evaluate the Xase activities to determine the anticoagulation properties of the testing compounds. In addition to Zheng et al. (2019) observations, Ma et al. (2021) also reported the anticoagulant properties of five fucan sulfates isolated from P. mollis. According to the authors, fucan sulfates isolated from P. mollis had strong and dose-dependent anticoagulant activities under the tested conditions. In another study, fucan sulfate isolated from H. floridana was also found to possess strong APTT-prolonging activity (Ning et al., 2022). Interestingly in this study authors pointed out a positive relationship between molecular weight and APTT-prolonging activity. High molecular weight fucan sulfates were found to possess strong anticoagulant properties (443.4 kDa) and low molecular weight fucans were found to possess low anticoagulant properties (11.5 kDa). However, high levels of bioaccessibility and bioavailability of bioactive ingredients in functional foods are the key requirements to develop functional foods (Rein et al., 2013). Therefore, even though the sulfated fucans have high bioactivities with high molecular weights they might not consider for functional food development as they have low bioaccessibility and bioavailability. Thus, people who trying to develop functional foods must want to consider this factor too.
Other than the P. mollis; sulfated polysaccharides isolated from Holothuria fuscopunctata. also found to possess intrinsic factor Xase inhibition activities (Gao et al., 2020). Mansour et al. (2019) also report the anticoagulant properties of fucoidan (1,376 kDa) separated from H. polii. In this study, the authors determined the thrombin generation using a calibrated automated thrombography. According to the results, the isolated polysaccharide was found to inhibit thrombin generation under tested conditions with a dose-dependent manner (5 μg/mL) (Mansour et al., 2019). In addition, Li et al. (2020a) also report the anticoagulant properties of a fucoidan (103.1 kDa) identified from H. polii using a Balb/c mice model. According to the authors, fucoidan reduces platelet aggregation caused by cyclophosphamide (cyclophosphamide is a chemotherapy drug with a broad spectrum of antitumor activity).
In addition to the fucoidans, low molecular weight fucosylated chondroitin sulfate (5.3 kDa) isolated from Ludwigothurea grisea was also found to possess promising ant icoagulation properties via inhibiting the Xase complex formation (Yin et al., 2022). Other than the above study fucosylated chondroitin sulfate isolated from Paracaudina chilensis and H. hilla are also reported to have anticoagulant properties when they were subjected to test for anticoagulant properties using clotting time assay. The clotting time assay is connected with the ability of these fucosylated chondroitin sulfates to induce the inhibition of factor Xa and thrombin in the presence of anti-thrombin IIIaw well as with the direct inhibition of thrombin in the absence of any cofactors (Ustyuzhanina et al., 2020). Guan et al. (2019) reported anticoagulation properties of fucosylated glycosaminoglycan isolated from A. japonicus. A. japonicus is extensively distributed on the northern coast of China. Other than China A. japonicus is also widely distributed in Japan and is one of the most popular and economically important sea cucumber species around the world due to its high value as a marine tonic. The results of Guan et al. (2019) revealed that A. japonicus has intrinsic factor Xase inhibition without showing any side effects such as FXII activation and platelet aggregation. These results scientific evidence for the traditional belief about A. japonicus medicinal values. Taken together, A. japonicus-based functional foods might have good demand in the market. Stichopus monotuberculatus is another well-known tropical sea cucumber used in folk medicine and tonic food with the traditional belief of its bioactive properties. However, in a recent study Yuan et al. (2022) scientific evidence for its anticoagulant activities using fucosylated glycosaminoglycan. According to the authors, fucosylated glycosaminoglycan had the potential to selectively inhibit intrinsic tenase complex without showing any effects on anti-factor IIa, Xa, and VIIa activity under the tested conditions (Yuan et al., 2022).
Taken together these results are suggesting that the consumption of sea cucumbers have the potential to reduce the risk of thromboembolic diseases. Therefore, sea cucumber-based functional food industry can get additional attention from potential consumers when marketing over other functional foods. Table 2 summarizes the recent anticoagulant properties reported from sea cucumbers.
| Name | Major bioactive compound | Method used | Mechanisms | Reference |
|---|---|---|---|---|
| Acaudina leucoprocta | (→ 3)-α-L-Fucp-(1→) | APTT, PT, and TT activities were determined using a coagulometer | Selective AT activity through HCII | He et al., 2020 |
| Apostichopus japonicus | Fucosylated glycosaminoglycan | Coagulometer | Inhibitory effect on Xase complex formation | Guan et al., 2019 |
| Cucumaria syracusana | Fucosylated chondroitin sulfate | APTT, TT, TGT and anti-FIIa/AT and anti-FIIa/ HCII assays | Mediation of HCII | Chahed et al., 2020 |
| Holothuria fuscopunctata | Fucosylated glycosaminoglycan | APTT assay, The potency for anti-iXase | APTT-prolonging activity | Zheng et al., 2022 |
| H. fuscopunctata | Sulfated polysaccharides | Calibrated automated thrombography | Xase inhibition | Gao et al., 2020 |
| Holothuria hilla | Fucosylated chondroitin sulfate | Clotting time assay | Inhibition of thrombin and factor Xa in the presence of ATIII | Ustyuzhanina et al., 2020 |
| Holothuria polii | Fucoidans (1,376 kDa and 103.1 kDa) | Calibrated automated thrombography, BALB/C mice model | Inhibited thrombin generation, reduces platelet aggregation | Li et al., 2020a; Mansour et al., 2019 |
| Holothuria floridana | -[l-Fuc2S4S-α(1,3)-l-Fuc-α(1,3)-Fuc-α(1,3)-l-Fuc2S-α(1,3)-l-Fuc2S-]n-. (443.4 kDa) | Plasma clotting assay | APTT-prolonging activity | Ning et al., 2022 |
| Ludwigothurea grisea | Fucosylated chondroitin sulfate | Commercially available heparin anti-IIa kit and heparin anti-FXa kit | Inhibitory effect on Xase complex formation | Yin et al., 2022 |
| Paracaudina chilensis | Fucosylated chondroitin sulfate | Clotting time assay | Inhibit thrombin and factor Xa in the presence of ATIII | Ustyuzhanina et al., 2020 |
| Pattalus mollis | Sulfated polysaccharides | PTT, PT, and TT reagents and standard human plasma on a coagulometer | APTT prolonging and intrinsic factor Xase inhibition | Ma et al., 2021; Zheng et al., 2019 |
| Stichopus monotuberculatus | Fucosylated glycosaminoglycan | Commercially available detection kits | Selectively inhibit intrinsic tenase complex | Yuan et al., 2022 |
Inflammation is part of our defense mechanism (Murakami et al., 2013). It is the process by which the immune system identifies and eliminates hazardous and foreign stimuli and starts the healing process (Fernando et al., 2017). Inflammation is divided into two forms as acute inflammation and chronic inflammation. Acute inflammatory responses are initiated with tissue damage due to microbial invasion, trauma, or poisonous substances that can trigger induced acute inflammation. Symptoms of acute inflammation usually appear within a few hours, and they last for a few days, such as cellulitis or acute pneumonia (Hirano, 2020). In contrast to acute inflammation, chronic inflammation is referred to as slow, long-term inflammation, which can be lasting for prolonged periods (a few months to several years). Usually, the severity and effects of chronic inflammatory responses vary with the cause of the injury and the capacity of the immune system to repair and overcome the damage (Lawrence & Gilroy, 2007; Pahwa et al., 2021). Furthermore, chronic inflammatory diseases have been identified as one of the major root causes of death in the world (Pahwa et al., 2021). Therefore, functional foods that are claimed to have anti-inflammatory properties have a good demand in the market. Recently, a number of studies are reported about the anti-inflammatory properties of sea cucumbers (Table 3). This section mainly discusses recent anti-inflammatory properties reported from sea cucumbers.
| Name | Major bioactive compound | Method used | Mechanisms | Reference |
|---|---|---|---|---|
| Apostichopus japonicus | Protein hydrolysates | Zebrafish model | Inhibition of inflammation-associated leukocyte recruitment | Zhang et al., 2021 |
| Cucumaria frondose | Sterol sulfate | RT-qPCR | Pro-inflammatory cytokine inhibition | Zhang et al., 2020 |
| Holothuria arguinensis | Crude extracts | COX-2 inhibitory assay | COX-2 inhibition | Carletti et al., 2022 |
| Holothuria leucospilota | Methanol extract | RAT | Hind paw edema | Moradi et al., 2020 |
| H. leucospilota | Methanol extract | BLAB/C mice model | COX-2 inhibition | Sasongko et al., 2020 |
| Holothuria scabra | Crude ethyl acetate extract | Cell culture | NF-κB and JNK inhibition | Pranweerapaiboon et al., 2020 |
| Isostichopus badionotus | Glycosaminoglycan-rich fraction | Male CD1 and C57Bl/6 | Down-regulate TPA-induced inflammatory responses | Olivera-Castillo et al., 2020 |
| Stichopus japonicus | Major yolk protein | C57BL/6J male mice | Prevents tissue damage, diminished occult diarrhea and bleeding | Feng et al., 2022 |
Holothuria scabra, has long been used as a medicinal ingredient in traditional therapies to treat various diseases due to its disease healing properties. Pranweerapaiboon et al. (2020) report the anti-inflammatory properties of an ethylacetate fraction of H. scabra, using lipopolysaccharide-exposed RAW 264.7 macrophages. According to the results, H. scabra treatment downregulated the well-known pro-inflammatory cytokines such as tumor necrosis factor (TNF)-α and interleukin (IL)-1β, inflammatory mediators (nitric oxide [NO]/inducible NO synthase and prostaglandin E2), and inflammatory signal cascades such as nuclear factor kappa-light-chain-enhancer of activated B cells and JNK (Pranweerapaiboon et al., 2020). In a recent study, Zhang et al. (2020) reported the potent anti-inflammatory properties of sterol sulfate extracted from the body wall of dietary sea cucumber (C. frondose). According to the author’s high-fat-high-fructose diet significantly upregulated the mRNA expression of proinflammatory factors and toll-like receptor 4, and downregulated IL-10 gene expression in male C57BL/6J mice. However, according to the authors sterol sulfate administrated mice had decreased expression levels of TNF-α, monocyte chemoattractant protein 1, and IL-6 (Zhang et al., 2020). In addition, cyclooxygenase-2 (COX2) inhibition activities of creams containing Anguilla bicolor (Eel) and sea cucumber H. leucospilota extract on croton oil-induced inflammation in BALB/C mice were also reported by Sasongko et al. (2020). According to the authors, H. leucospilota treatment was found to decrease inflammatory cell number, epidermal thickness, and decreasing number of cells expressing COX2 in cotton oil-induced BALB/C mice compared to the control (Sasongko et al., 2020). In addition to the BALB/C mice model Moradi et al. (2020) attempted to evaluate the anti-inflammatory mechanisms of methanolic extract of Persian Gulf sea cucumber (H. leucospilota) on rats. Similar to Sasongko et al. (2020) observation H. leucospilota treated group had significantly low Hind paw edema compared to the carrageenan-induced rat group (Moradi et al., 2020). In addition, Zhang et al. (2021) also report the anti-inflammatory properties A. japonicus hydrolysates against CuSO4-induced neuromast damage in a zebrafish model. According to the authors A. japonicus protein hydrolysates blocked (< 10 kDa fraction) the leukocyte migration to the site of injury in the zebrafish model compared to the untreated control (Zhang et al., 2021). Similarly, a glycosaminoglycan-rich fraction prepared from Isostichopus badionotus was also found to possess potent anti-inflammatory properties under in vitro and in vivo conditions. To evaluate anti-inflammatory activities the authors used 12-O-tetradecanoylphorbol-13-acetate (TPA) mouse ear inflammation model. TPA typically upregulates the inflammation via regulating inflammatory cell infiltration, and this model is commonly used to determine the anti-inflammatory potency of samples. According to the results, co-treatment of the mouse ear with fucosylated chondroitin sulfate was found to down-regulate TPA-induced inflammatory responses in male CD1 and C57Bl/6 mice models (Olivera-Castillo et al., 2020). Taken together these results suggest that sea cucumbers have strong anti-inflammatory properties that can be used as a functional food for the prevention of inflammation-associated disorders. In another study, Feng et al. (2022) attempted to evaluate the acute colitis inhibitory properties of major yolk protein from sea cucumber (S. japonicus) using dextran sulfate sodium-induced colitis mice model (C57BL/6J male mice). In this study, surprisingly authors observed S. japonicus major yolk protein has the potential to improve the health of colitis in dextran sulfate sodium-induced induced colitis mice. According to the results, major yolk protein prevents tissue damage, diminished occult diarrhea and bleeding, promotes anti-inflammatory cytokines, and increase the contents of total short-chain fatty acids (Feng et al., 2022).
Based on the above in vivo and in vitro studies it is clear that regular consumption of sea cumbers have the potential to improve the immune system to fight against inflammatory diseases.
Under stress conditions, the human body generates reactive oxygen species (ROS), and these ROS responsible for initiating various disorders in the body such as inflammatory diseases, cancer, aging, cardiovascular diseases, brain dysfunction, and cataracts (Hossain et al., 2022a; Verma & Mishra, 2014). Antioxidants have the potential to prevent free radical formation and therefore slow down the oxidative stress in the cells and exert a protective role against ROS-mediated disorders. It is thought that dietary antioxidants can help prevent oxidative stress-induced diseases. The use of natural antioxidants in food products has gained tremendous attention in recent years due to studies indicating that synthetic antioxidants may cause adverse effects (Lourenço et al., 2019).
Consequently, sea cucumbers are one of the most significant marine organisms for their bioactive compounds that are antioxidant in nature (Table 4). Guo et al. (2020) evaluated the antioxidant effect of protein hydrolyzate separated from sea cucumber in a parquet-induced animal model by using nematodes (Caenorhabditis elegans). Protein hydrolyzate of sea cucumber upregulated the superoxide dismutase and catalase activities and reduced the malondialdehyde contents. Moreover, it suppressed the ROS accumulation and scavenged 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radicals. This resulted in a higher survival rate and reduced production of reactive ROS promising antioxidant potential. In the same year Li et al. (2020b) studied the anti-oxidant activities of a novel fucoidan extracted from the sea cucumber S. chloronotus. This novel fucoidan significantly reduced lipid peroxidation when treated in RAW 264.7 cells, and attenuated NO and TNF production. Using microwave-assisted enzymatic hydrolysis instead of conventional enzymatic hydrolysis may be a promising approach to preparing antioxidant peptides from the collagen of sea cucumber Acaudina molpadioides. The peptide extracted using Neutracec showed high DPPH scavenging activity. Polysaccharides obtained from sea cucumber gonads through ultrasound-assisted enzymatic extraction techniques were found to demonstrate higher anti-oxidant potential (Wang et al., 2022a). Using the ultrasound-assisted enzymatic techniques led to sea cucumber gonad polysaccharide chains being degraded and more sulfate groups being exposed. According to the authors’ findings, increasing concentrations (0.25 to 4.00 mg/mL) of polysaccharides obtained from sea cucumber gonads showed higher DPPH and Hydroxyl radical (OH) scavenging ability and ferric reducing/antioxidant power. According to Hossain et al. (2022b), sea cucumber Cucumaria frondosa processing discards rich in phenolic compounds due to high-pressure processing. Authors revealed that phenolic compounds inhibited cupric ion-induced human low-density lipoprotein peroxidation, exhibited tyrosinase inhibitory activity, and suppressed advanced glycation end products produced through non-enzymatic Maillard reaction. In 2020, Reza and Zahra reported that the hydrolyzed protein (molecular weight of < 30-KDa) of sea cucumber H. leucospilota extracted using Alcalase and Flavourzyme enzymes exert anti-oxidant potential (Safari & Yaghoubzadeh, 2020). According to the authors, Flavourzyme enzymes (concentrations of 5 and 2 mg/mL) exerted a higher activity in the DPPH-free radical control than in the Aalcalase activity. According to Fan et al. (2023), a novel peptide isolated from sea cucumber showed excellent iron chelating activity at the concentration of 117.17 ± 2.62 mg/g. This peptide showed a loose and blocky structure with increased particle size. The authors revealed stronger radical scavenging properties and high-efficiency iron supplementation ability in peptides subjected to chelating.
| Name | Major bioactive compound | Mode of action | Bioactivity | Reference |
|---|---|---|---|---|
| Apostichopus japonicus | Protein hydrolyzate | Downregulate (reactive oxygen species) level in Caenorhabditis elegans under increased oxidative stress induced by paraqua | Antioxidant | Guo et al., 2020 |
| Cucumaria frondosa | Crude phenol fraction | Inhibited cupric ion-induced human low-density lipoprotein peroxidation | Antioxidant | Hossain et al., 2022b |
| Holothuria leucospilota | Hydrolyzed protein (molecular weight of < 30-KDa) | DPPH-free radical inhibition | Antioxidant | Safari & Yaghoubzadeh, 2020 |
| Sea cucumber name not mentioned by the authors | Crude polysaccharides | DPPH∙, ∙OH scavenging ability and ferric reducing/antioxidant power | Antioxidant | Wang et al., 2022a |
| Sea cucumber name not mentioned by the authors | Peptide | Iron chelating activity | Antioxidant | Fan et al., 2023 |
| Stichopus chloronotus | Fucoidan | This novel fucoidan significantly reduced lipid peroxidation | Antioxidant | Li et al., 2020b |
| Holothuria leucospilota | Diethyl ether extract of body wall | Via mitigating the replication potential of human immunodeficiency virus-1 | Antiviral | Bahroudi et al., 2018 |
| Isostichopus badionotus | Sulfated fucan and fucosylated chondroitin sulfate | Antiviral properties against adenovirus and human cytomegalo via blocking virusviral attachment and entry | Antiviral | Zoepfl et al., 2021 |
| Patallus mollis | Sulfated glycans | Antiviral effect on human rotavirus | Antiviral | Garcia-Candela et al., 2021 |
| Thelenota ananas | N-acetylgalactosamine, glucuronic acid, fucose, and ester sulfate in 1:1:1:3.7 | Bind with the S glycoprotein to prevent SARS-CoV-2 host cell entry | Antiviral | Song et al., 2020 |
| Holothuria atra | Coumaric acid, catechins, chlorogenic acid, rutin, pyrogallol, and ascorbic acid | Hepatoprotective activity against thioacetamide-induced liver fibrosis in rats | Hepatoprotective | Esmat et al., 2013 |
| H. atra | Phosphate buffer extract also | Hepatoprotective effect in 7,12-dimethylbenz[a]anthracene-induced hepatorenal diseases in rats | Hepatoprotective | Dakrory et al., 2015 |
| Holothuria scabra | Eicosapentaenoic acid-enriched ether-phospholipids | Prtect paracetamol-induced liver damage in mouse model | Hepatoprotective | Wang et al., 2022c |
| Cucumaria frondosa | Hydrolysates of gonads | Improving insulin resistance and lipid metabolism | Antidiabatic | Wang et al., 2022b |
| Holothuria leucospilota | Polysaccharide | Alleviate the symptoms of T2DM in Goto-Kakizaki rats | Antidiabatic | Zhao et al., 2020 |
| Holothuria nobilis | Hydrolysates | Protective effect in streptozotocin and high-fat-diet induced diabetic rats via activating the PI3K/Akt pathway | Antidiabatic | Wang et al., 2020 |
| Stichopus japonica | Proteins | Improve glucose uptake of 3 T3-L1 cells and high insulin-induced insulin-resistant Hep G2 cells | Antidiabatic | Gong et al., 2020 |
| Thelenota ananas, Cucumaria frondosa | Fucosylated chondroitin sulfate | Activate IRS/PI3K/AKT signaling and regulating GSK-3β gene expression in T2DM rats | Antidiabatic | Zhu et al., 2020 |
In order to fight against each other, humans and viruses adopt different strategies. The antiviral drug class consists of medicines that treat viral infections in particular. The development of antiviral drugs involves several steps, such as identifying targets and screening, generating leads, optimizing them, and registering them (Kausar et al., 2021). Several drug discovery scientists are interested in finding novel bioactive natural compounds with antiviral activity in natural resources. Sea cucumber is a promising natural candidate which exerts antiviral activities and is used in traditional medicine for different applications.
Experiments with some virus-infected cells have demonstrated that sea cucumbers possess promising antiviral properties. Previously, Garcia-Candela et al. (2021) reported that an aqueous extract of Patallus mollis sea cucumber exerts an antiviral effect on human rotavirus A by inhibiting 99% of the virus during two distinct phases of the virus: absorption and viral inactivation phase. Sulfated fucan and fucosylated chondroitin sulfate from the sea cucumber I. badionotus exhibited antiviral properties against adenovirus and human cytomegalovirus and it is accompanied by the sulfation content, negative charge density, and conformational shapes (Zoepfl et al., 2021). One recent virus responsible for the infection of millions of people in the world is COVID-19. The novel antiviral T. ananas, a sea cucumber contains N-acetylgalactosamine, glucuronic acid, fucose, and ester sulfate in 1:1:1:3.7 respectively responsible for the antiviral effect. Song et al. (2020) researched on sea cucumber sulfated polysaccharide and showed significant antiviral activities at concentrations of 3.90–500 μg/mL. Bahroudi et al. (2018) showed that crude extracts of sea cucumber H. leucospilota exert an anti-viral effect via mitigating the replication of human immunodeficiency virus-1. Diethyl ether extract of body wall with 2.79 thixotropic index displayed the highest antiviral activity.
Wang et al. (2022b) investigated eicosapentaenoic acid-enriched ether-phospholipids of sea cucumber H. scabra on hepatoprotective potential in alcohol-induced liver damage. The authors revealed that sea cucumber H. scabra exerts antioxidant properties at a dose of 500 mg/kg BW in a paracetamol-induced mouse model. Sticoposide has potent anticancer activity against HL-60 and subcutaneous tumor cells (CT-26) by inducing apoptosis through activation of both intrinsic and extrinsic signaling pathways indicating the benefits of sea cucumber has high cell regeneration ability. A group of researchers investigated the hepatoprotective activity against thioacetamide-induced liver fibrosis in rats (Esmat et al., 2013). The authors performed a high-performance liquid chromatography analysis for sea cucumber blend extracts revealed the presence of several phenolic components such as coumaric acid, catechins, chlorogenic acid, rutin, pyrogallol, and ascorbic acid. Sea cucumber H. atra phosphate buffer extract also exerted a hepatoprotective effect in 7,12-dimethylbenz[a]anthracene-induced hepatorenal diseases in rats (Dakrory et al., 2015). Sea cucumber H. atra phosphate buffer extract decreased levels of reduced glutathione-S-transferase, glutathione, catalase, and superoxide dismutase, in the liver tissue, and induced liver histopathological alterations. Similarly, mixed extracts of H. atra contained bioactive phenolic compounds with antioxidant activity and exerted potent hepatoprotective effects against thioacetamide-induced liver injury in rat mode (Dakrory et al., 2015).
Diabetes is one of the most common and chronic metabolic diseases, indicating an abnormally high level of blood glucose in the body (Kc et al., 2015). As a consequence of low blood insulin activity caused by insulin resistance or impaired insulin secretion in the pancreas, high blood glucose levels are associated with accelerated lipid peroxidation during the pathogenesis of diabetes and its complications. This leads to clinical symptoms of diabetes. Diabetes is still not effectively treated with any of the many drugs that are available for managing diabetes, providing cells with sufficient glucose supply, or preventing severe complications associated with hyperglycemia (Khotimchenko, 2018).
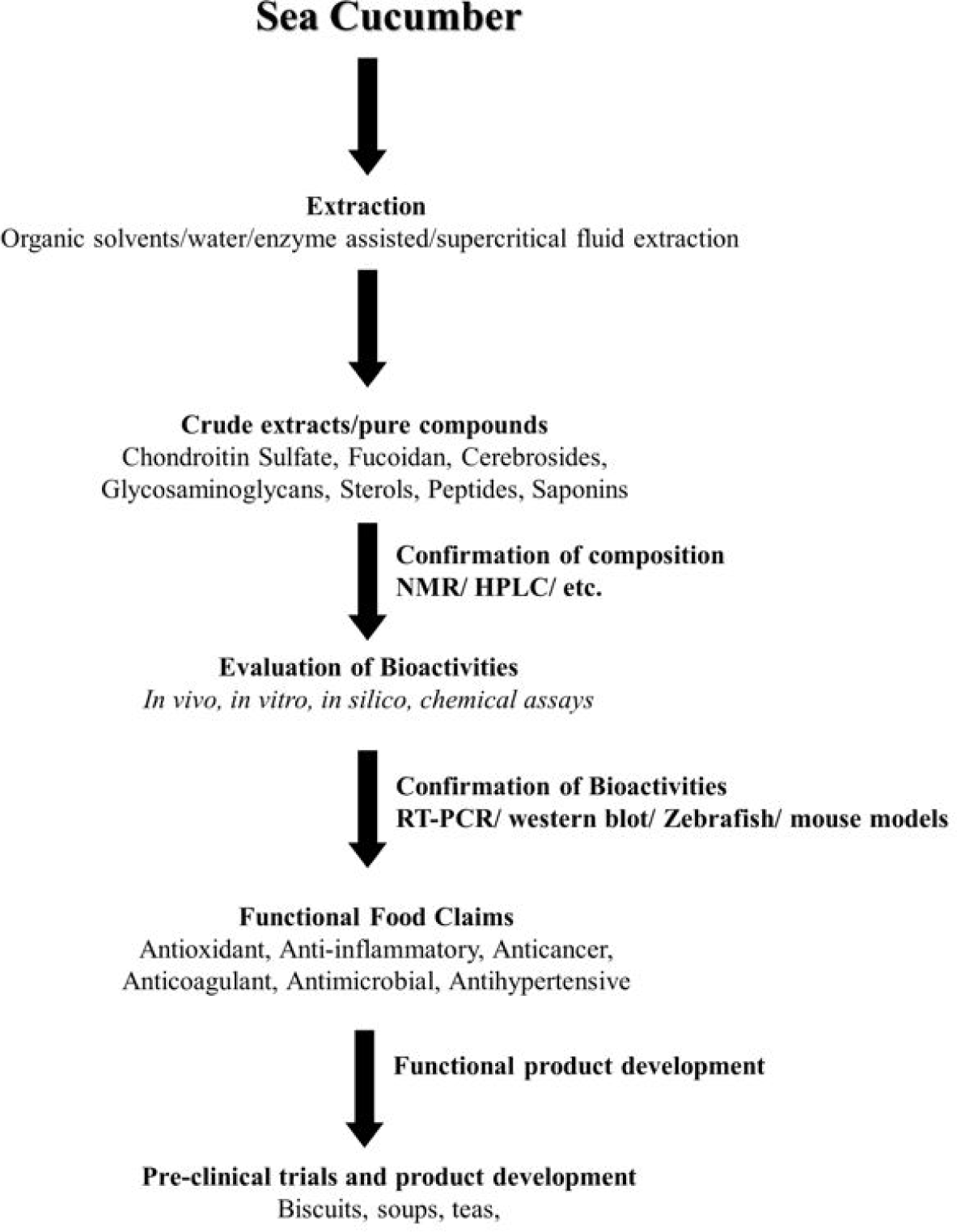
Polysaccharide from sea cucumber (H. leucospilota) is identified to improve the gut microbiome and alleviate the symptoms of type 2 diabetes mellitus (T2DM) in Goto-Kakizaki rats (Zhao et al., 2020). T2DM rats were given a polysaccharide fraction produced from low-edible sea cucumber, containing largely fucan and fucosylated chondroitin sulfate andnsea cucumber exerted hypoglycemic potential via activating IRS/PI3K/AKT signaling and regulating GSK-3β gene expression in T2DM rats (Zhu et al., 2020). Hydrolysates of gonads from sea cucumber (C. frondosa) exerted anti-diabetic effects by improving insulin resistance and lipid metabolism disorders (Wang et al., 2022c). Saponins isolated from sea cucumber have been identified to restore insulin action in the body by releasing insulin from pancreas and decrease the blood glucose levels (Tripathi & Srivastava, 2006). Further, saponins from different sea cucumber species have been identified to decrease the serum alpha amylase activity and enhance the synthesis of glycogen in the liver. This will repress gluconeogenesis by decrease the level of serum adiponectin (Tripathi & Srivastava, 2006). Gong et al. (2020) has investigated the hydrolysates of S. japonicus protein in the gastrointestinal digestion. Wang et al (2020) also identified that hydrolysate of sea cucumber exerts anti-diabetic potential in high-fat-diet induced diabetic rats. According to Wang et al. (2020), hydrolysate of sea cucumber has alleviated body weight loss, oral glucose tolerance and insulin resistance in diabetic rats. Anti-diabetic effect of hydrolysates of sea cucumber is revealed to be exerted via triggering PI3K/Akt signaling pathway (Wang et al., 2020). Peptides released from the digestion have been identified to increase the glucose uptake of 3 T3-L1 cells and high insulin-induced insulin-resistant Hep G2 cells (Gong et al., 2020). Therefore, different bioactive compounds isolated from sea cucumber species can be utilized for the better management of diabetes. Fig. 4 summerize the possible approaches to develop functional product from sea cucumbers to reduce risk of different diseases.
Conclusion
The functional food industry is found to be one of the most profitable and continuously growing segment in the global food industry. Furthermore, the increasing interest in the consumption of natural healthy foods instead of fast foods makes more market demand for functional foods. Therefore, foods rich in bioactive properties such as anti-inflammatory, antioxidant, antidiabetic, and anticancer are considered ideal foods to develop functional products. It is interesting to observe the prominent bioactive properties of compounds and crude extracts separated from different sea cucumber spp such as anticancer, anticoagulant, anti-inflammatory, antioxidant, hepatoprotective, etc. under in vivo and in vitro conditions. With these bioactive properties, sea cucumbers can play a pivotal role in the functional food industry. However, so far no studies summarize the potential of sea cucumbers to develop functional foods. Therefore, this review will be helpful to fill the knowledge gap between the functional food industry and the research community.








