Introduction
Worldwide, fish consumption is increasing at a faster rate than fish production from both capture and aquaculture fisheries (FAO, 2022; Wake & Geleto, 2019). This is due to the rapid population growth, changing lifestyles due to urbanization, and improved income status of the people. Additionally, the known health benefits of fish consumption as a major protein source have enhanced the fish intake (Anderson et al., 2017). Likewise, in Uganda, fish is a stable source of protein especially from the three commercially important species, that is Nile perch (Lates niloticus, Linnaeus, 1758), Nile tilapia (Oreochromis niloticus, Linnaeus, 1758), and Silver fish (Rastrineobola argentea) (Cowx & Ogutu‐Owhayo, 2019). During the late 1950’s and early 1960’s, Nile perch and Nile tilapia were introduced to Lake Victoria and Lake Kyoga (Ogutu-Ohwayo, 1990). This was done to boost the fisheries resources that were majorly composed of tilapiines and haplochromines (Ogutu-Ohwayo, 2004). The boost resulted in their catches dominating the international, regional and national markets (Witte et al., 2009).
Previous studies indicated that Nile perch and Nile tilapia are prolific breeders, thus provided with conducive environmental parameters, they are capable of producing enough fish for domestic and commercial purposes (Cornelissen et al., 2015; Hughes, 1992). Nile perch and Nile tilapia are known to breed during the rainy season and in the inshore shallow waters with less currents (Ojwang et al., 2014). When it rains, the warmer water, which is less dense, comes up on the lake surface while the cooler rain water goes to the lower layers. Continuous warmer temperature (temp) mixed with rainfall, trigger breeding in fish in the natural environment (Njiru et al., 2006). Additionally, during the rainy season, more food is available and as such, the fish reduce their movements and inhabit particular sites depending on their characteristics (Goudswaard et al., 2011). In particular sites on the lake, are the fish breeding habitats, that provide favorable biotic and abiotic conditions for fish to spawn (Rosenfeld & Hatfield, 2006). Identifying and protecting fish breeding habitats from the impacts of anthropogenic activities is a practical step towards sustainable fisheries management (Chen et al., 2020).
Fish breeding habitats have been used on Lake Victoria and Lake Albert, as an effective fisheries management tool for biodiversity conservation, natural recovery of fish stocks and reverse habitat degradation (Nkalubo et al., 2018; Ojwang et al., 2014). Protection of fish breeding habitats is comparatively cheaper than restocking or conventional lake patrols to enforce compliance (Aura et al., 2018). Additionally, the degrading activities affect the fish spawning and nursery habitats, shoreline vegetation and water quality (Chen et al., 2020). Unfortunately, the fish breeding habitats on Lake Kyoga are not well known making the management of the fishery in a sustainable way complex. Therefore, the main objective of this study was to identify and characterize the breeding habitats for Nile perch and Nile tilapia so that they can be protected from anthropogenic activities.
Materials and Methods
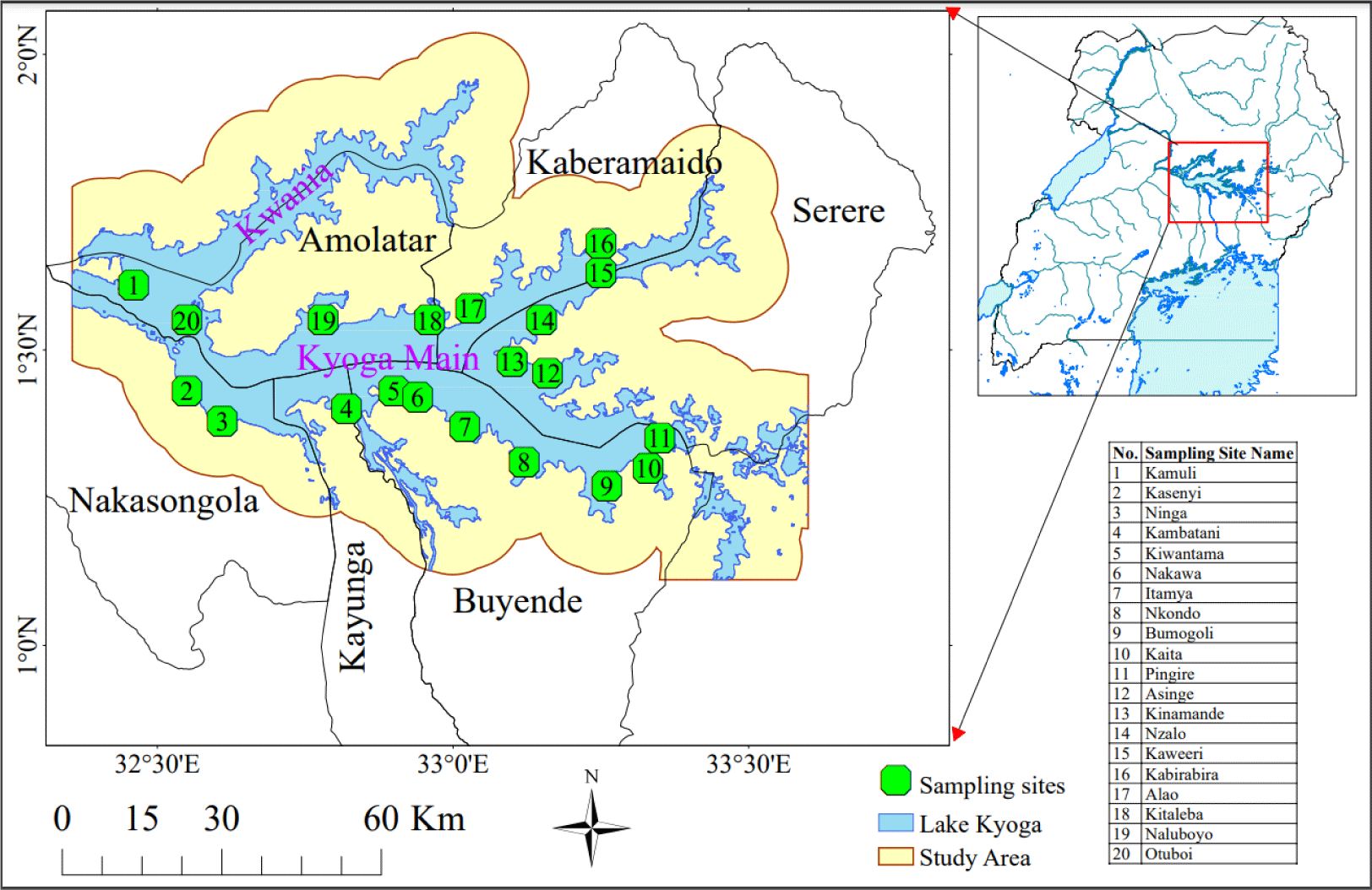
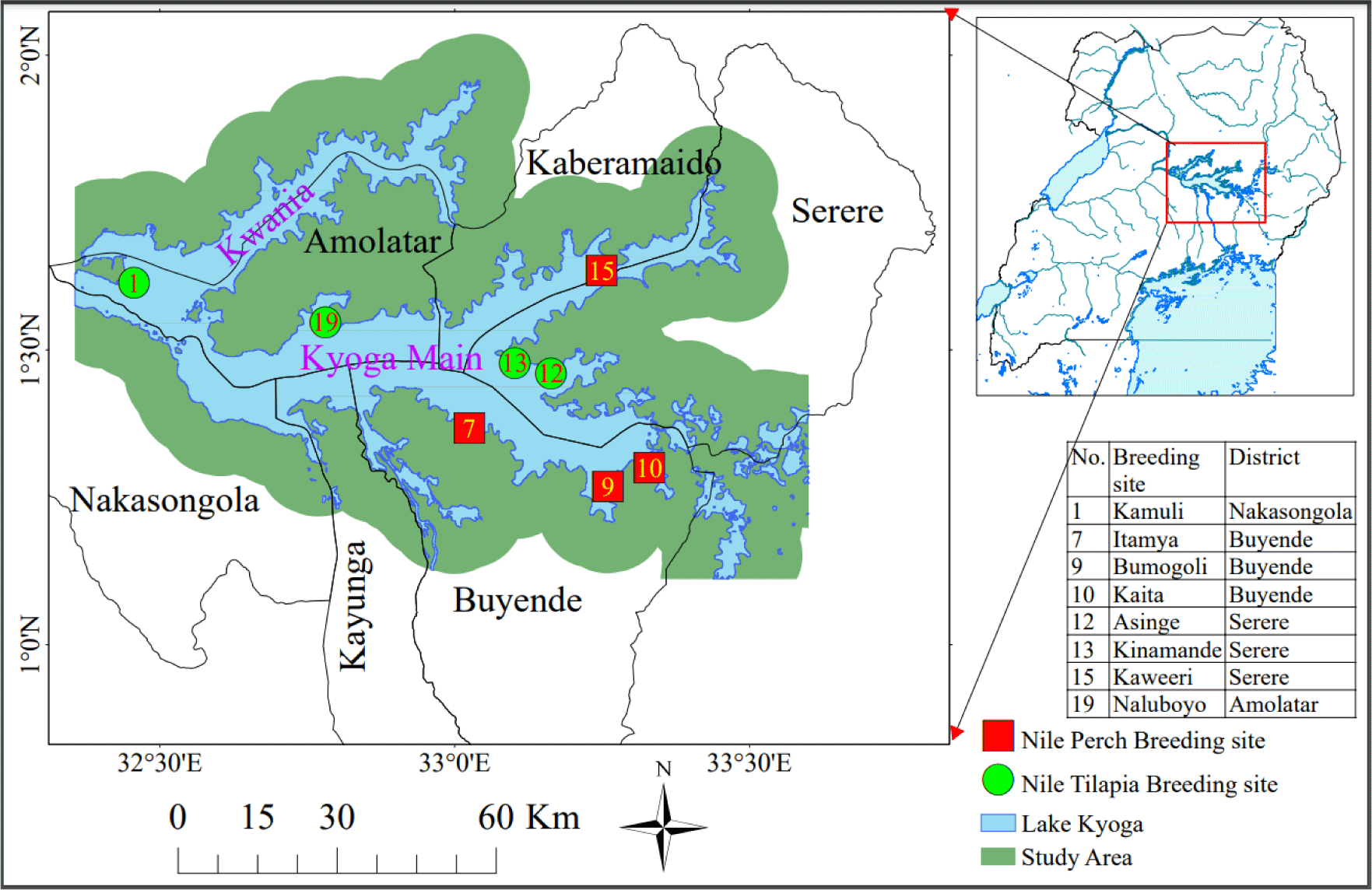
Lake Kyoga is located in central Uganda, between longitudes 32°05′ and 33°35′ E and latitudes 1°05′ and 1°55′ N. Its location is part of the central African plateau at an altitude of 1,100 meters above mean sea level. Lake Kyoga is connected with Lake Victoria in the south and Lake Albert in the North-West via the Victoria Nile and Albert Nile respectively. It is composed of two arms, Kwania and Kyoga main (Fig. 1), and over 30 satellite lakes connected to the main lake by the wetlands (Green, 2009), that form bays along the lake shorelines. The lake is shallow with an open water area of about 2,600 km2 and a mean depth of 2–4 m (Andama et al., 2017). The Lake is invaded by the water weeds mainly water hyacinth—Eichhornia crassipes and the Kariba weed—Salvinia molesta (Obubu et al., 2021a). It is surrounded by dense swamps covered with papyrus—Cyperus spp., water lilies—Nymphaeaceae spp., and hippo grass—Vossia spp. and has numerous aquatic fauna including fish. Lake Koga has a unique ecosystem where the cichlids are densely populated in the satellite lakes whereas Nile perch exists in the Kwania and Kyoga main arms (Mbabazi et al., 2004).
A reconnaissance survey was carried out to identify the potential fish breeding and nursery habitats, which was immediately followed by sampling from the identified sites. Selection of the potential sites was based on the environment preferences for the two fish species (Aura et al., 2018; Cornelissen et al., 2015) and indigenous knowledge from fishermen. Using a handheld (Garmin) Global Positioning System, geo-referenced coordinates indicating the sampling sites were captured and the generated data were incorporated into GIS using ArcGIS 10.2. The sites identified were delineated as shapefiles spatially using World Geographic System 84 UTM Zone 36N projection. Samples of water, fish, lake bottom and habitat vegetation were collected from 20 sites (Fig. 1) using two boats consecutively by the research crew. Previous studies on breeding seasons of Nile perch and Nile tilapia on Lake Victoria indicated that they breed during the rainy seasons when the water temp increases and more food is available (Njiru et al., 2006; Nkalubo et al., 2018; Ochumba & Many Ala, 1992). Thus, sampling was done in April of 2021 and 2022, which is part of the longer rainy season of March, April, and May with warmer temp ranges (Obubu et al., 2021a).
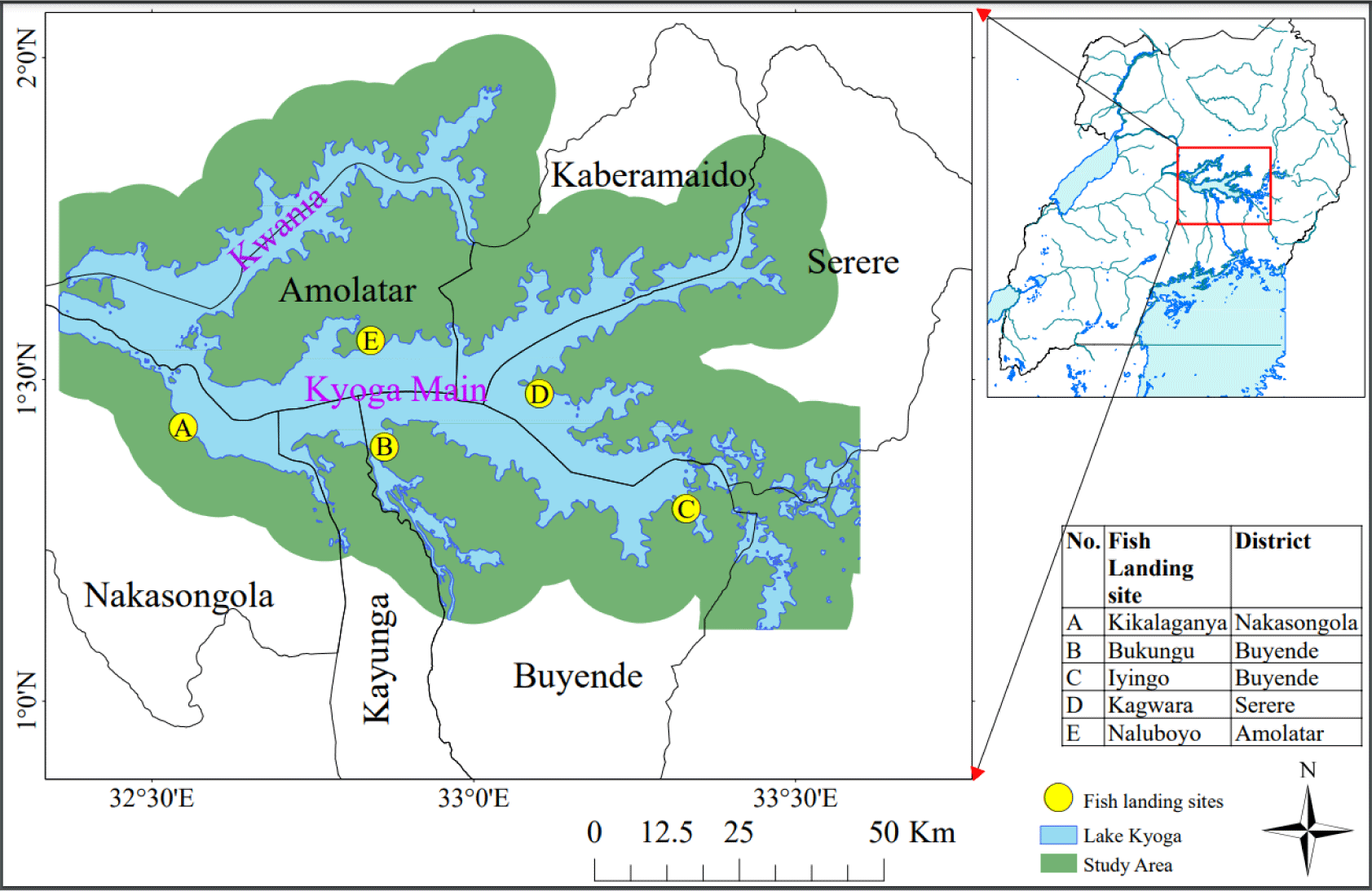
Key informant interviews were conducted with active fishermen with at least 10-years of experience in fishing on the lake. Through interactions with the landing site management committees, three experienced fishermen were identified and out of the three fishermen, one of them was part of the research crew, during data and sample collection. The interviewed fishermen were selected from fish landing sites that included Kikalaganya from Nakasongola District, Bukungu and Iyingo from Buyende District, Kagwara from Serere District, and Naluboyo from Amolatar District (Fig. 2). These sites were chosen because they handle fish destined for factory processing and as such, their management systems are well streamlined. At each of the five fish landing sites, three fishermen were interviewed making a total of 15 people. Data were analyzed using thematic content analysis. Key informant interviews were carried out to validate the primary data gathered from field observations and analysis of water, fish and vegetation samples.
Water samples were collected before setting the fishing nets to avoid sampling mixed water. It was done at a depth of 0.5 m due to the shallow nature of the lake. The field parameters were temp, dissolved oxygen (DO), electrical conductivity (EC), potential hydrogen (pH), Secchi depth, and turbidity (turb), while the laboratory parameters were chlorophyll-a (Chl-a) and total suspended solids (TSS). Measurement of the field parameters was done using a multi-probe (CTD-90) at geo-referenced points in their standard units (temp in °C, DO in mg/L, EC in μS/cm and turb in NTU). A Secchi disc was used to determine the Secchi depth in meters (m). Calibration of the instruments was done in the laboratory before measurements in the field according to the instructions on the multi-probe manuals. Distilled water was used to rinse the apparatus after use in each site to avoid sample mixing. Procedures for sampling and analyses were from the American Public Health Association (APHA) standard methods (Bridgewater et al., 2012).
Water samples for Chl-a (µg/L) analysis were collected from the lake and were filtered using a hand pump in the field. The algae (Chl-a) was concentrated onto a 47 mm diameter glass microfiber Whatman filter paper. Filtration was done until the green color was observed on the filter paper, in which the paper was folded using a pair of forceps and wrapped in an aluminum foil to avoid light degradation of the samples. The foil was labelled, put in a glass bottle and stored in an ice-cool box. The samples were transported to the National Water Quality Reference Laboratory—Entebbe, for analysis. The algal pigments were extracted from the filter residue using absolute ethanol in the laboratory. A spectrophotometer was used to measure the absorbances, which were converted into concentrations while a gravimetric method using a virgin GF/B filter paper was used to determine TSS (mg/L). Principal component analysis (PCA) with viramax rotation and descriptive statistics were used to analyze the recorded data.
The lake bottom was sampled using Ekman grab sampler, where the sediment was placed in a tray for determination of the type. Bivariate correlation was used to show how the lake bottom impacts fish breeding.
Boat seine and cast nets were used to collect the fish samples. At each site, the boat seine was hauled three times while cast net, nine times per sampling time. The 20 sites were sampled two times in April 2021 and 2022, making a total of four fishing operations per site. The harvested young fish were immediately put back to the lake without causing them damage. The captured Nile perch and Nile tilapia were sorted, counted and dissected to assess the maturity stages of the gonads using the conventional category for maturity stages (Ogutu-Ohwayo, 1988). Stages I–III were considered as immature while stages IV–VI were taken as mature levels. Fish dissection and gonads observation was done on the boat and the data were analyzed using one-way analysis of variance (ANOVA) and Bivariate correlation.
Habitat vegetation—the macrophytes, were collected during water and fish sampling. Identification of the species names of the habitat vegetation cover types was done both onsite and in the Botany laboratory of Makerere University. Observations of the current status of the lake shoreline were done and the activities taking place were recorded. Bivariate correlation and descriptive statistics were used to analyze the collected data.
Results
Respondents comprising of 20% were not sure whether the two species share the same breeding sites, while 33% said yes that they share and 47% said, the two fish species don’t share breeding sites. Of the 15 respondents, 60% agreed that the breeding sites require protection from human activities in order to increase production, 20% were not sure and 20% said no, the sites do not require protection. The fishermen went ahead to describe the breeding site indicators, the activities carried along the shoreline and suggested protection measures (Table 1).
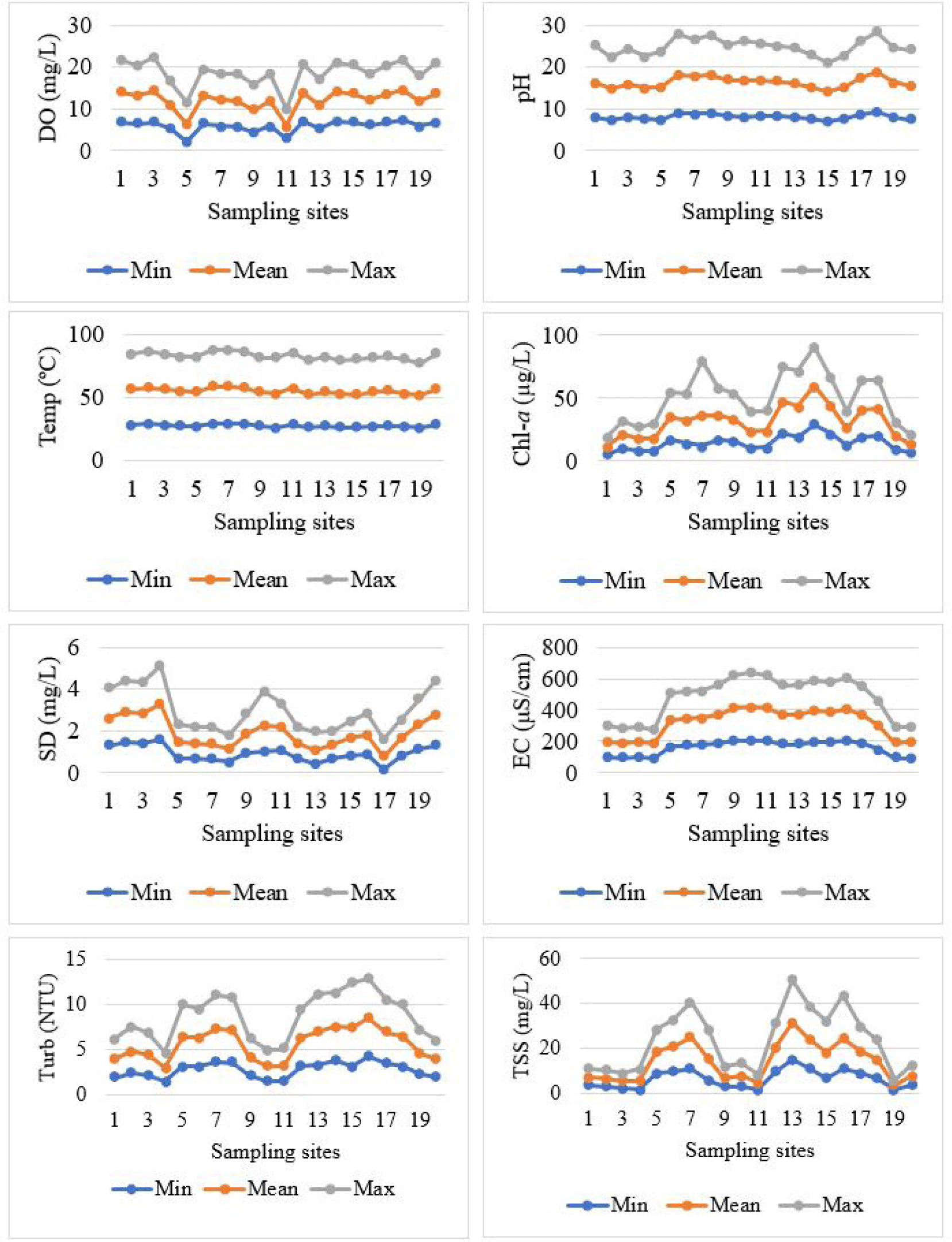
The minimum, mean and maximum values of the water quality parameters were presented as line graphs (Fig. 3). Temperature range for all the sites, was between 25.9°C to 29.4°C. Secchi depth values ranged from 0.2 to 1.9 m, while DO were between 2.2 to 8.2 mg/L. The EC was between 91 to 216 μS/cm and the pH ranged between 7.0 to 9.6. The turb and the TSS ranged between 1.4 to 3.8 NTU and 2.0 to 16.0 mg/L, respectively. The overall mean values of the water quality parameters indicated that the lake water quality is within the recommended levels (Table 2) for fish to breed. The data set was further analyzed using one-way ANOVA and the results for the water quality parameters indicated significant differences among the sites (Table 3).
* Data from Ahmed et al. (2019).
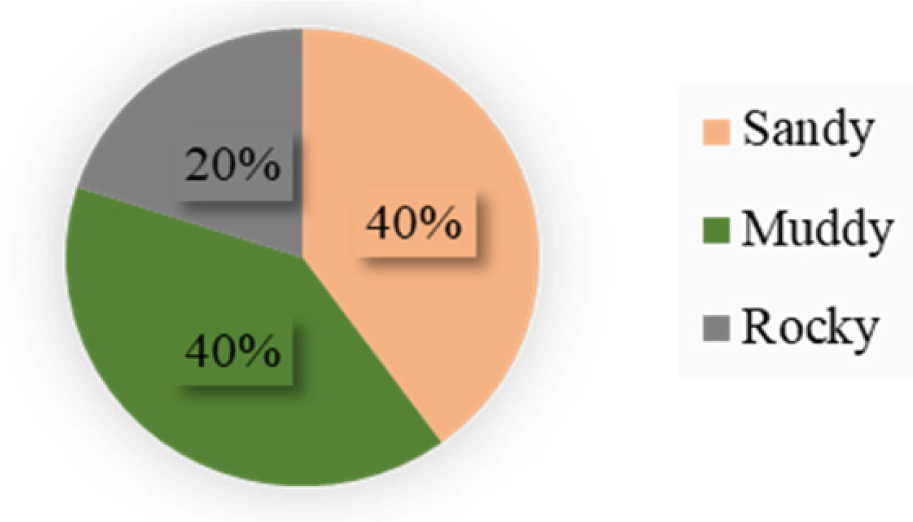
Results indicated three types of lake bottom types (Table 4). The rocky bottom type covered only 20% within the sampled sites. Sandy and muddy bottom types were equally spread at 40% each (Fig. 4). Bivariate correlation analysis (Table 5) of the bottom types indicated a strong negative correlation with Nile perch.
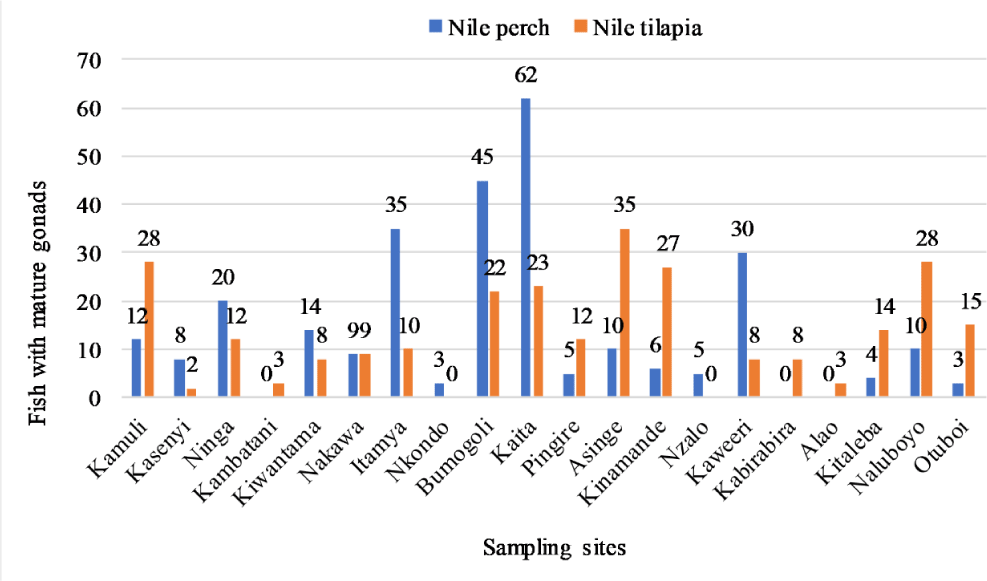
The data set was analyzed and presented using a bar graph and Kaita, Bumogoli, Itamya and Kaweeri had more Nile perch with mature gonads than other sites. Asinge, Kamuli, Kinamande and Naluboyo fish breeding sites had more Nile tilapia with mature gonads than other sites (Fig. 5). Analysis of the fish gonads data with the one-way ANOVA indicated no significant difference with in the sites. Bivariate correlation analysis (Table 5) of the fish gonads indicated a strong negative correlation with Nile perch while Nile tilapia had a positive correlation with the vegetation types. Out of the 20 potential fish breeding sites sampled, only four sites were more suitable for Nile perch breeding and also four for Nile tilapia (Fig. 6).
From observations of the major physical attributes surrounding the lake, the shoreline of Lake Kyoga is generally still covered by aquatic vegetation. Different habitat vegetation covers were observed ranging from those rooted in the sediment, the freely floating and the submerged macrophytes. Bivariate correlation analysis of the vegetation types (Table 5) indicated a strong negative correlation with Nile perch while Nile tilapia had a positive correlation.
Running the PCA of all the parameters considered for characterization of breeding sites for Nile perch on Kyoga main, water quality, fish gonads and habitat vegetation components cumulatively contribute 82.5% (Table 6). The components were categorized into 4 factors, with components in factor 1 category having the highest eigen valves and components in factor 4 having the least eigen values.
Discussion
Analysis of the fish, water quality, vegetation and lake bottom types data collected from the 20 sampling sites showed that 8 sites exhibited more favorable conditions for Nile perch and Nile tilapia breeding. The four sites favoring Nile perch breeding were Itamya, Bumogoli, Kaita, Kaweeri while for Nile tilapia were Kamuli, Asinge, Kinamande and Naluboyo (Fig. 6). The sites favoring Nile perch were associated with rocky bottom type while Nile tilapia were with sandy bottom type (Table 4). From the interview with the fishermen, they described Nile tilapia to breed in sites with sandy bottoms locally known as “Kumukyanga”. Nile tilapia has been studied to prefer sandy bottom sites because during the breeding season, the male builds a nest in the sand, then entices the ripe female tilapia into the nest to lay the eggs. Thereafter, the male fertilizes the eggs and the female keeps them in the buccal cavity for hatching (Goudswaard et al., 2011; Ojwang et al., 2014; Rana, 1988). The sand is easy to remove while digging the nest, thus less energy consuming when compared to rocky or muddy bottom.
The bottom types showed a negative correlation with Nile perch (Table 5) because the lake bottom has a connection with the quality of water. Taking Pingire site as an example, because of its muddy nature, the DO and Chl-a levels were very low (Fig. 3). This means, the sun rays don’t enter to lower areas of the water for the plants to make enough oxygen yet Nile perch is very sensitive to low oxygen levels. Previous studies indicated that Nile perch preferred sites with vegetated and rocky bottoms and this could be due such areas having well DO levels (Kahwa, 2013; Manyala et al., 2005; Muyodi et al., 2011). Chapman et al. (2002) found out that Nile perch can be found in muddy areas as long as DO levels are enough for fish to survive. According to Ahmed et al. (2019), the recommended DO levels for fish to breed and survive should be not less than 5 mg/L (Table 2).
There was a negative correlation of Nile perch and vegetation cover (Table 5). Pistia spp. (water lettuce) and Salvinia spp. (Kariba weed) showed a negative correlation (–0.632). Presence of this kind of vegetation types on the lake affects the water quality making the site unsuitable for fish breeding. These vegetation types cover the water surface blocking the light rays from penetrating the water. Kariba weed has been reported to thrive in shallow areas with less turbulences, which are also suitable breeding and nursery grounds for many fish species (Asmare, 2017; Aura et al., 2018). Its ability to cover the water surface, hence preventing light penetration and oxygen circulation, and acting as a habitat to predators of the fish eggs or the fish fry, which affects the productivity of the lake (Cohen, 1994; Obubu et al., 2021b). The weed is spreading to various corners of the lake due to the moving suds on the lake that move along with it, requiring action to impede its spread.
The water quality parameters (Table 3) indicated significant differences with in the sites at 0.05 confidence interval. The quality of the water has a great impact on determining a suitable site for fish breeding (LVFO, 2018). The mean values of the water quality parameters were within the recommended levels (Table 2). This could be due to the wetlands surrounding Lake Kyoga, which act as buffer zones (Were et al., 2019), thus maintaining good water quality for fish breeding and growth. The wetland is generally not degraded and the bays within the study area are not yet exploited by human activities. For example, Kamuli site, located at a small Kamuli island, had no settlements by the time this research was conducted, hence no land degradation observed in the area. Kaweeri is also a small island with a few settlements practicing small-scale agriculture for their survival, which did not affect the suitability of the site as a fish breeding habitat. According to LVFO (2018), the bays are associated with calm warmer waters and abundant food, which are very vital for nursery grounds. Such sites are sheltered with macrophytes providing the juveniles with protection from predators (Nagelkerken et al., 2000). In the study by Aura et al. (2018), to map the fish breeding and nursery grounds on Lake Victoria, it was also found that juvenile and mature fish existed in sheltered bays compared to offshore areas, an indicator that these are preferred sites as breeding and nursery grounds.
Water quality parameters, fish gonads and vegetation types accounted for 82.5% of the factors to considered while characterizing a site as a fish breeding habitat as indicated by the PCA of the data (Table 6). Secchi depth and turb indicate how clear the water is while high Chl-a levels indicate high productivity of the lake and saturation of DO. For example, Kaita site had low turb or high-water transparency (Fig. 3 and Table 3) and this could be attributed to the nature of the shoreline being rocky with little human activities taking place around the site. Kiwantama site had low DO levels (Fig. 3 and Table 3) and this could be linked to the agricultural activities in the area that are extended through the buffer zone to the lake. When it rains, the lake water becomes very turbid due to the eroded soils. The physico-chemical properties of the water indicate the health status of aquatic ecosystems and the survival of the organisms therein (LVFO, 2018; Obubu et al., 2021b). For the case of fish, DO is required for respiration, which supports growth and survival, pH influences the physiological functions while EC predicts gonadal maturation. Furthermore, low turb facilitates fish to swim up and downstream the water column thus influencing their search for food, hence influencing their abundance, distribution, and diversity. The water transparency or Secchi depth affects the visual capacity of fish and the photosynthetic activities of the plant organisms (Suzuki & Pompeu, 2016).
In summary, Nile perch and Nile tilapia breed in separate habitats and breed during the rainy seasons. Nile perch breeds in rocky areas with well DO levels while Nile tilapia breeds in sandy habitats. Water quality and vegetation cover contribute greatly in determining the suitability of an area as a breeding site thus activities that degrade the water quality and vegetation cover within the breeding sites should be monitored and controlled. Increased production of these two fish species will highly boost the living standards of the fishing communities and enhance government revenue collections. Nile perch and Nile tilapia are very important sources of protein, thus protecting their breeding sites from human activities like fishing in such sites, is an achievable approach through co-management. Therefore, the 8 characterized sites are recommended for demarcation and protection as fish breeding habitats by the resource managers and fishing communities, and should be submitted for gazettement by policymakers for sustainable management of the fishery.