Introduction
Ultraviolet (UV) B exposure can help the body synthesize vitamin D, which is significant for bone and calcium homeostasis (Bouillon, 2017). However, as a high-energy component, overexposure to UVB leads to various skin damage, including sunburn, photoaging, photocarcinogenesis, and other disorders (Matsumura & Ananthaswamy, 2004). The risk of skin damage is increasing due to the increasing amount of UVB reaching the Earth’s surface (Cavinato & Jansen-Dürr, 2017; Schuch et al., 2017). Consequently, researchers have shown increased interest in resources that can safeguard the skin from photodamage caused by UVB radiation.
Sargassum horneri, belonging to the northwestern Pacific Ocean, is a brown seaweed that rich in various bioactive components such as sulfated polysaccharides, phlorotannins, fucoxanthin, sterols, and terpenoids (Kirindage et al., 2022; Li et al., 2020; Shao et al., 2014). It leads to various health benefits, and show great potential applications in medicine, food, and cosmetic. Recent research indicated that some derivative compounds of S. horneri exhibited strong photoprotective effects. Wang et al. (2021) reported that (-)-loliode isolated from S. horneri significantly decreased oxidative damage and inhibited apoptosis in UVB-irradiated human keratinocytes and zebrafish. It is reported that sargachromenol obtained from S. horneri attenuated cellular oxidative stress in human dermal fibroblasts (HDF) irritated by UVA through activator protein-1 pathway (Kim et al., 2012). Fernando et al. (2020) prepared a fucoidan fraction with low molecular weight from S. horneri and found that it could reduce UVB-triggered human keratinocytes damage.
In addition, many studies have found that some phytosterols are highly permeable to the skin and can protect the skin from UV radiation. For example, Hwang et al. (2014) found that fucosterol obtained from the brown seaweed Hizikia fusiformis reduced UVB-induced expression of matrix metalloproteinase (MMP)-1, interleukin (IL)-6, p-c-Jun, and p-c-Fos, and increased the production of pre-collagen type I and transforming growth factor-β1 to protect the skin from photodamage. We also obtained a sterol compound, 3-hydroxy-5,6-epoxy-β-ketone (HEBI), from S. horneri and found that HEBI possessed a strong anti-inflammatory activity in our previous study (Asanka Sanjeewa et al., 2021; Kim et al., 2021). In this study, we aim to assess the photoprotective effect of HEBI so as to further develop the application of HEBI in functional foods, pharmaceuticals and cosmetics.
Materials and Methods
S. horneri was collected in June 2020 from the coastal area of Jeju Island, Korea. After washing and drying, S. horneri was stored at 4°C. The enzyme-linked immunosorbent assay (ELISA) kits (including tumor necrosis factor [TNF]-α, IL-6, IL-1β and MMPs), 1,3-bis(diphenylphosphino)propane (DPPP), diaminofluorophore-FM diacetate (DAF-FM-DA), 2,7-dichlorofluorescein diacetate (DCFH-DA), and methylthiazolyldiphenyl-tetrazolium bromide (MTT) were purchased from Sigma-Aldrich (St. Louis, MO, USA). All other drugs utilized in this investigation met analytical grade standards.
The preparation procedure for HEBI was described in our earlier study (Kim et al., 2021). Briefly, an 80% methanol extract of S. horneri was fractionated sequentially with hexane, chloroform, and ethyl acetate. Then, the chloroform fraction was separated via high-performance centrifugal partition chromatography (HPCPC) (the solvent system consisting of n-hexane/ethyl acetate/methanol/distilled water (5:5:5:5:5, v/v)), and further purified via prep-high performance liquid chromatography (HPLC) using a semi-preparative C18 column (YMC-Pack ODS-A, YMC, Kyoto, Japan; 5 μm, 10 × 250 mm) with a gradient elution of acetonitrile and distilled water. After freeze-drying, HEBI was obtained with a purity of over 99%. HEBI were dissolved in dimethyl sulfoxide and diluted using phosphate buffered saline for the cell and zebrafish experiments.
The HaCaT cells (ATCC® PCS-200-001™, ATCC, Manassas, VA, USA) and HDF cells (ATCC®PCS201012™, ATCC) were exposed to HEBI (6.25–100 μg/mL) and cultured at 37°C for 24 h (Wang et al., 2018a). The assessment of HEBI cytotoxicity on cells were performed by detecting cell viability through MTT assay.
Cells were pretreated with HEBI and then exposed to UVB (Wang et al., 2021). The UVB irradiation dose was determined based on our previous research to be 30 mJ/cm² for HaCaT cells and 50 mJ/cm² for HDF cells (Wang et al., 2018b). The reactive oxygen species (ROS) levels were determined by DCFH-DA, and absorbance values were measured at 516 nm emission wavelength at 488 nm excitation wavelength. The cell viability was measured by MTT, and absorbance values were determined at 490 nm. The formation of apoptotic bodies in HaCaT cells was measured by Hoechst. ELISA kits were used to determine the collagen content, pro-inflammatory cytokines, and MMPs production in HDF cells.
Adult zebrafish were induced by light to spawn naturally and then the embryos were collected. Hatched zebrafish larvae at 2 days post-fertilization were randomized into groups of 10 each. The larvae were pretreated with HEBI (6.25–25 μg/mL) for 1 h, following by UVB exposure for 6 h (Wang et al., 2020). To assess ROS levels, nitric oxide (NO) levels, cell death, and lipid peroxidation, larvae were stained by DCFH-DA (20 μg/mL for 1 h), DAF-FM-DA (10 μM for 3 h), acridine orange (10 μg/mL for 0.5 h), and DPPP (3 μM for 1 h), respectively (Kawashima et al., 2018). The experiments were approved by the Animal Care and Use Committee of the Ocean University of China (Approval No. SPXY2025040801).
Results
In living cells, MTT can be reduced to blue-violet crystals by the succinate dehydrogenase of the mitochondria, which indirectly reflects the cell viability by absorbance measurements (Stockert et al., 2018). Therefore, the cytotoxicity of HEBI was assessed through MTT assay. The data demonstrated that the cell viability exceeded 90% at HEBI concentrations between 6.25 and 25 μg/mL, suggesting that there is no significant cytotoxicity towards HaCaT cells (Fig. 1B). However, cell viability was significantly impacted at treatment concentrations of 50 and 100 μg/mL, decreasing to 79.52% and 72.07%, respectively. Consequently, HEBI at concentrations of 6.25, 12.5, and 25 μg/mL were selected for subsequent experiments.
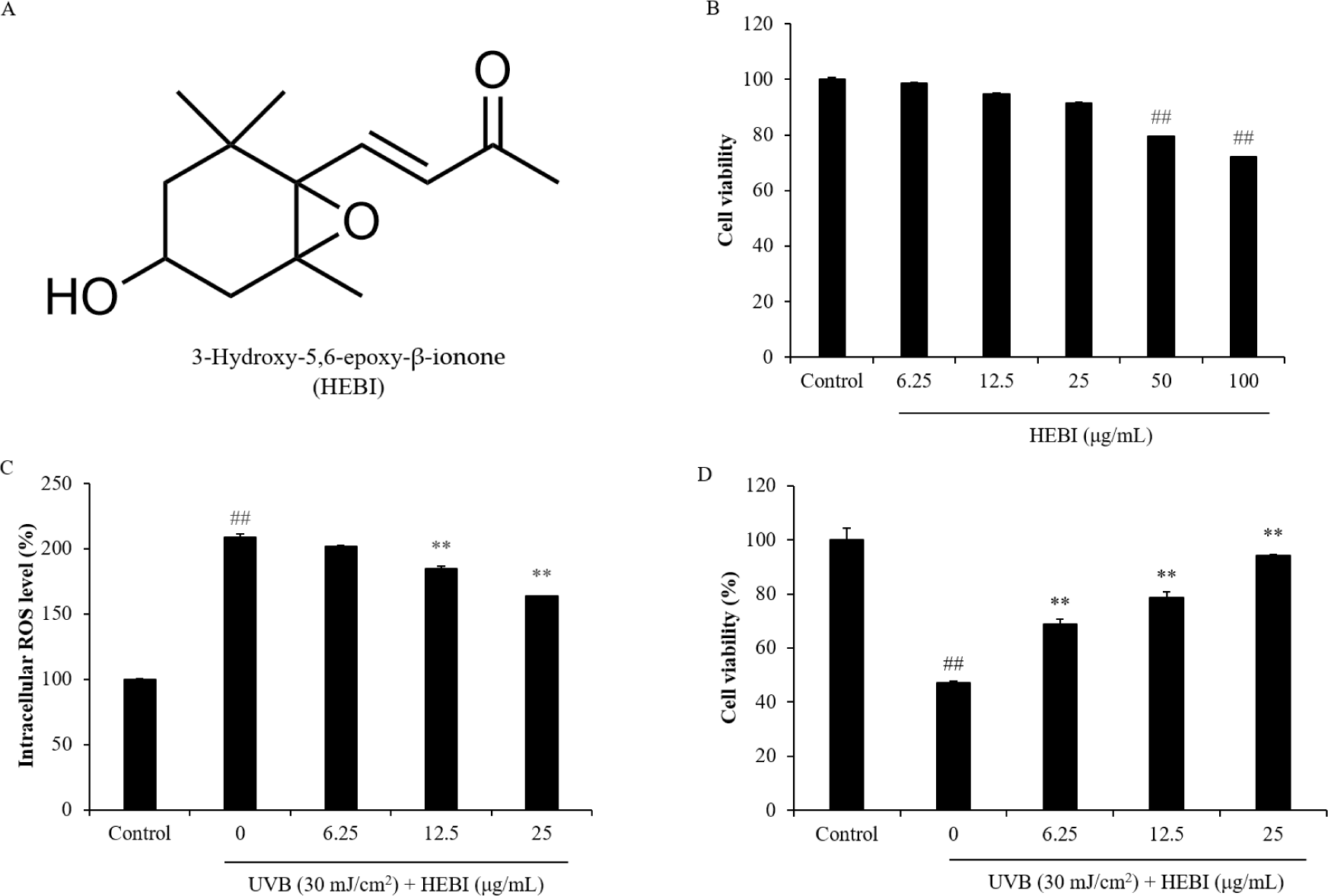
HaCaT cells pretreated with HEBI were irradiated with 30 mJ/cm2 UVB, and then the photoprotective effect of HEBI was discovered by assessing cell viability, ROS levels and apoptosis. The results revealed that there was a 208.77% increased of intracellular ROS level and a 47.13% decrease of cell viability on HaCaT cells irradiated by UVB comparing to the controls (Fig. 1C and Fig. 1D). HEBI treatment significantly suppressed ROS production and enhanced the cell viability dose-dependently. When treated with 25 µg/mL HEBI, the ROS level in cells was downregulated to 163.63% and the cell viability was upregulated to 94.21%.
The dye Hoechst penetrates the cell membrane and releases blue fluorescence after embedding in the DNA (Bucevičius et al., 2018). The nuclei of apoptotic cells fluoresce more brightly and chromatin condensation in the nucleus. Fig. 2 shows that UVB stimulated the formation of apoptotic bodies in HaCaT cells, whereas HEBI reversed this trend. At a HEBI concentration of 25 μg/mL, it can be seen that there is a reduction in bright blue fluorescence and an even distribution of chromatin, close to that of the control group.

The results demonstrated that HEBI can effectively alleviate UVB-induced damage in HaCaT cells.
The results of the toxicity experiments of HEBI on HDF cells are presented in Fig. 3A. The high concentration of HEBI showed significant toxicity to HDF cells. The cell viability decreased to 42.41% at the concentration of 100 μg/mL. Fortunately, 6.25–25 µg/mL HEBI had no significant toxicity on HDF cells, which were chosen for further exploration.
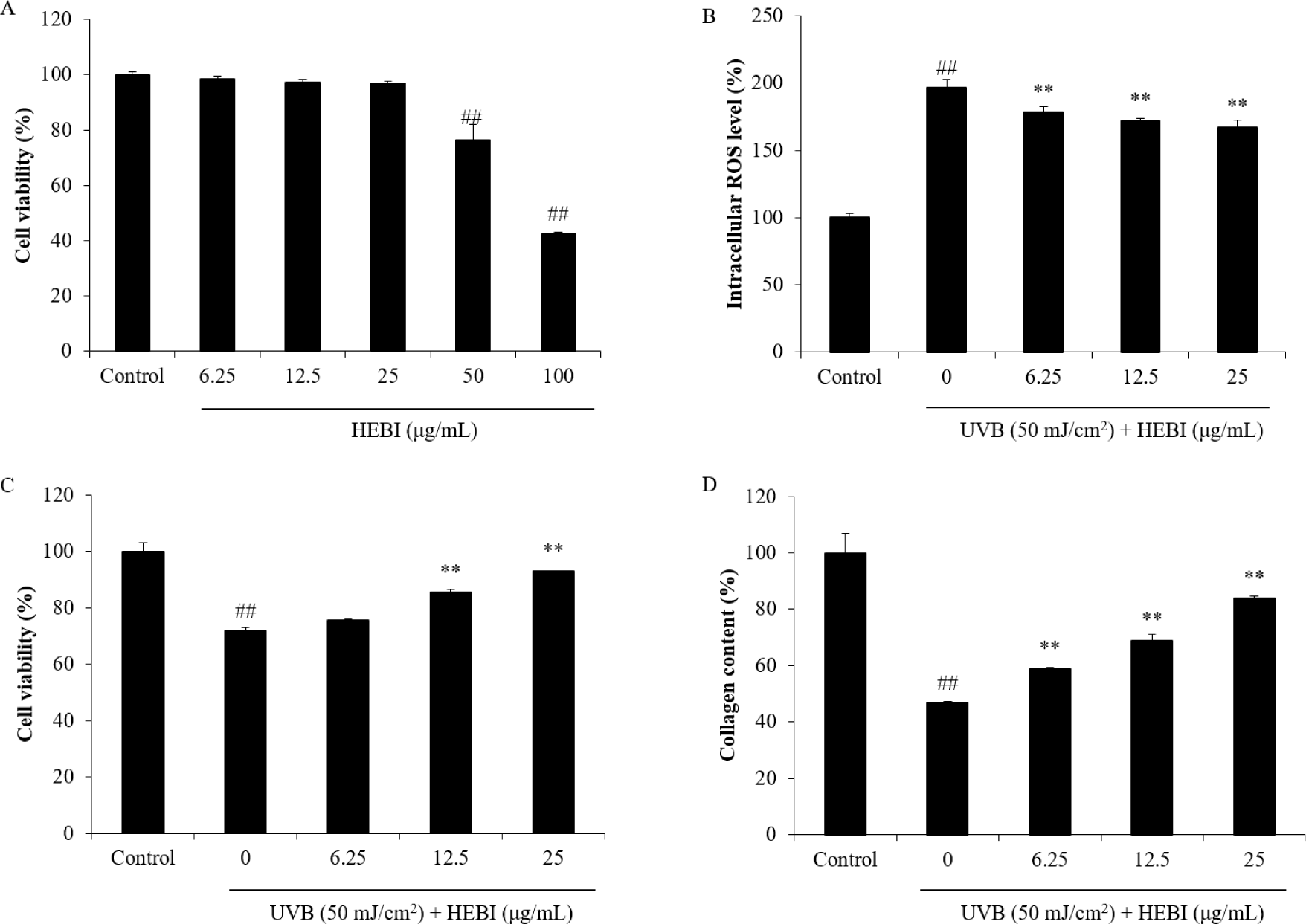
The assessment of ROS levels in HDF cells was conducted using the DCFH-DA assay. The result demonstrated that HEBI treatment at 25 μg/mL concentration suppressed the increase in intracellular ROS levels from 196.73% to 167.72% after UVB irradiation (Fig. 3B). In addition, MTT analysis showed that the cell viability of HEBI-pretreated HDF cells was significantly improved comparing to the UVB irradiation only group (Fig. 3C). Moreover, Fig. 3D shows that collagen content in HDF cells irradiated with UVB was dramatically decreased to 46.80% comparing to the controls. 6.25–25 μg/mL HEBI treatment significantly increased the collagen level to 58.90%, 68.97% and 83.93%, respectively.
As Fig. 4 shows, the UVB irradiation upregulated the level of collagen-degrading MMPs, among which MMP-1 showed the largest increase of 294.60% compared to controls. However, the expression of MMPs was all significantly inhibited by HEBI in HDF cells induced by UVB. Furthermore, UVB promoted TNFα, IL-1β, and IL-6 production, which were suppressed by HEBI dose-dependently (Fig. 5).
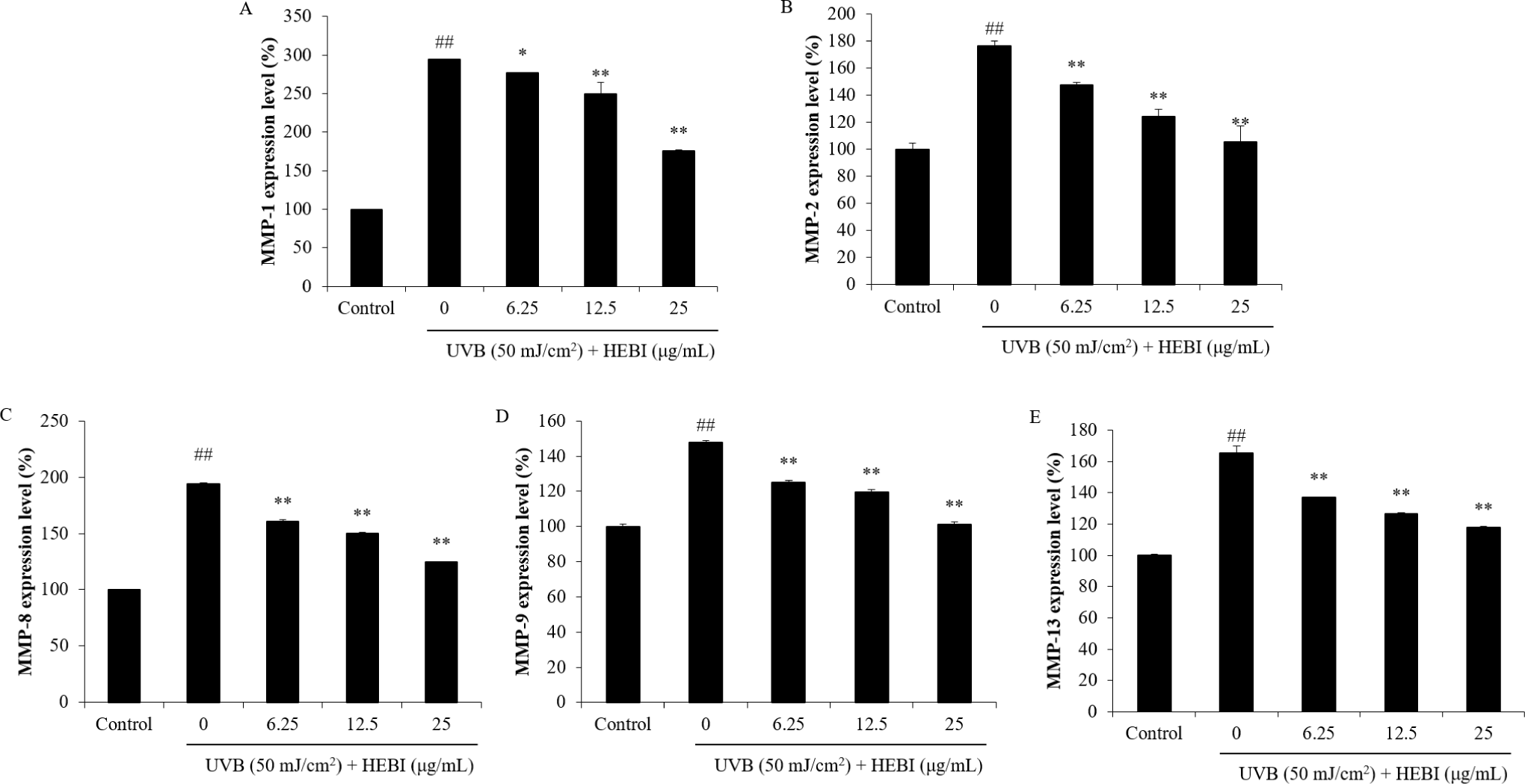
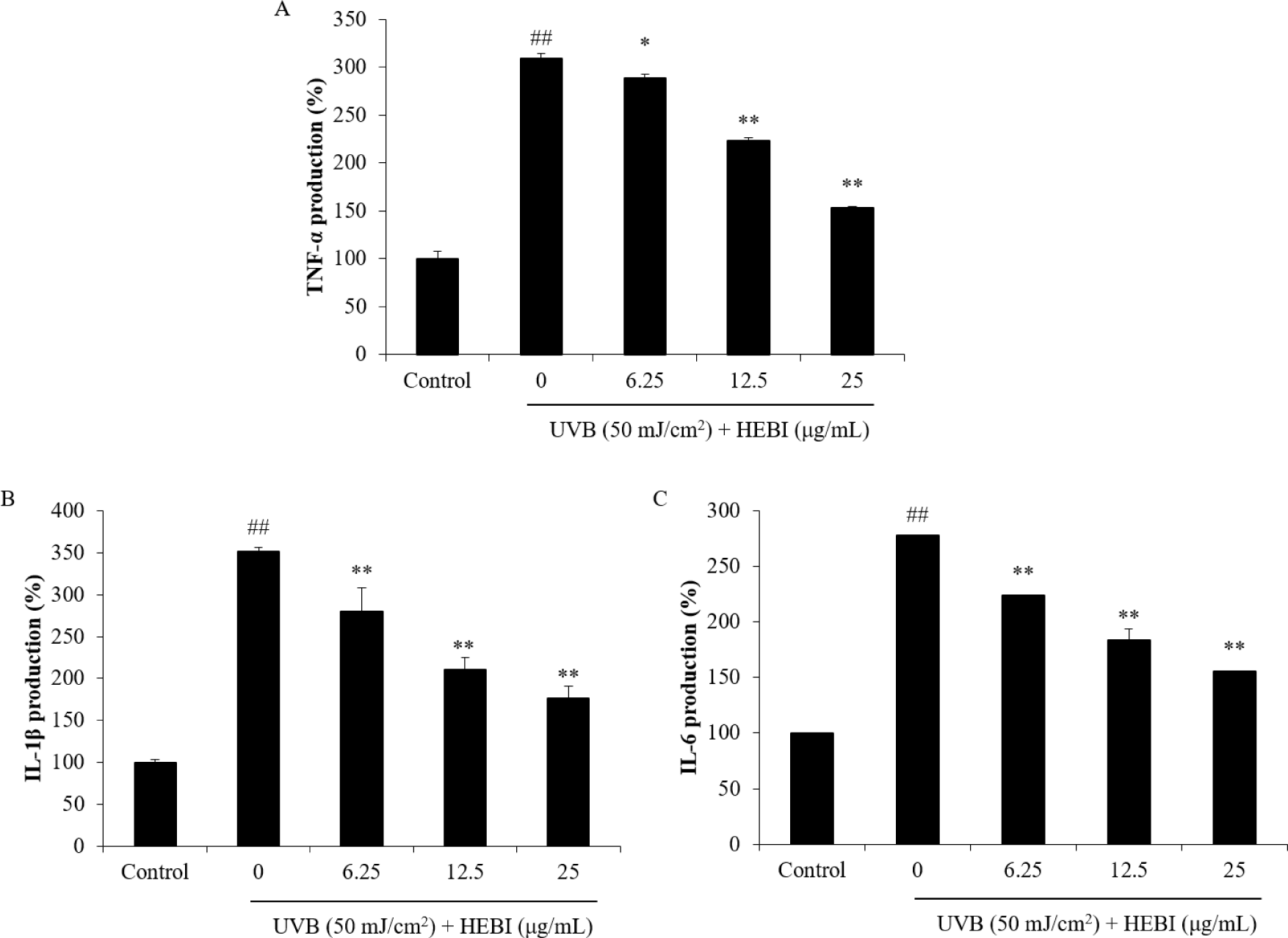
The results suggested that HEBI possessed a strong photoprotective effect on HDF cells.
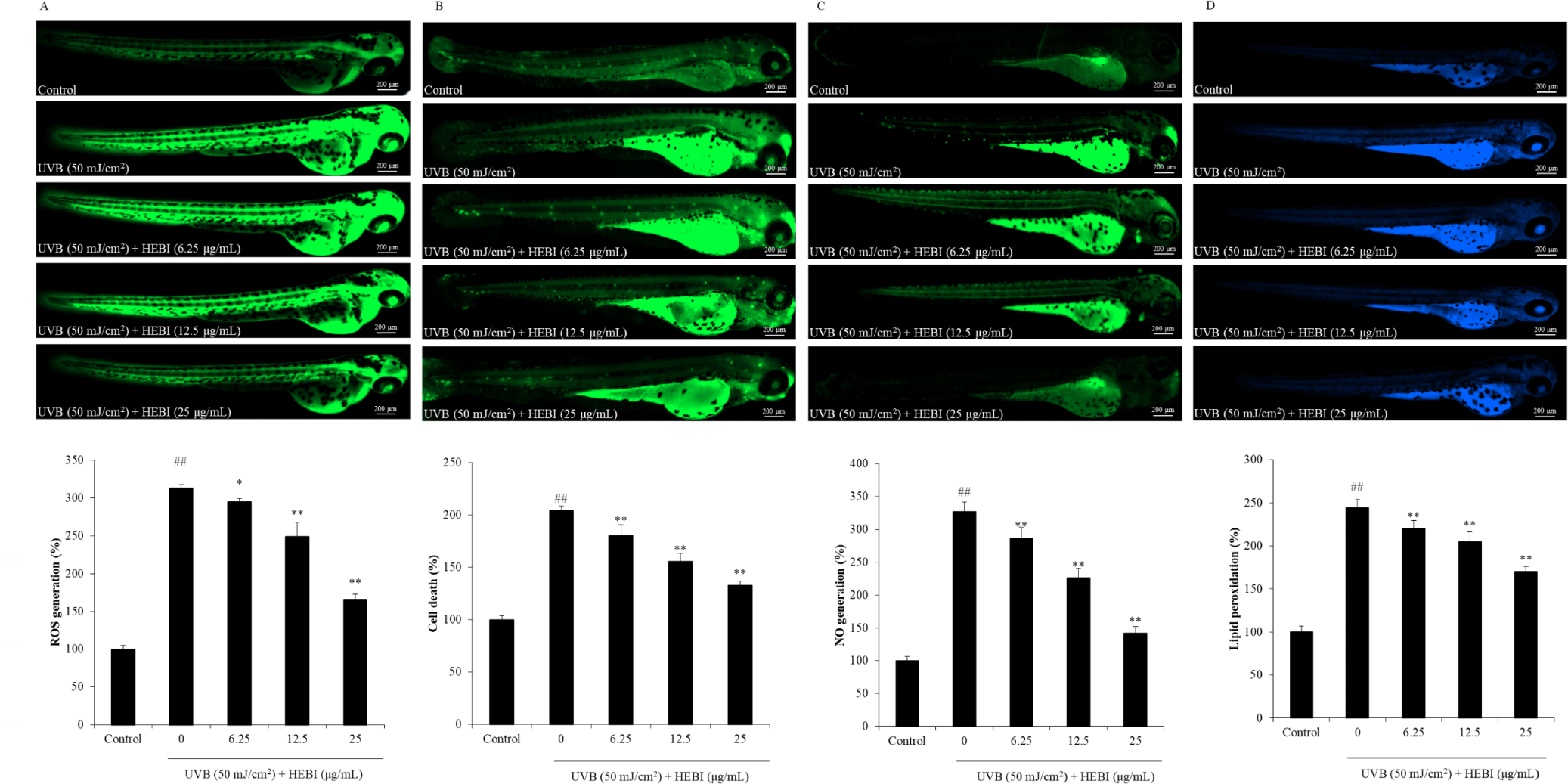
To verify the photoprotective effect of HEBI, further experiments were performed with zebrafish. The ROS production in zebrafish exposed to UVB was 3 times higher than in unirradiated zebrafish (Fig. 6A). However, HEBI effectively decreased the ROS levels to 166.04%. As Fig. 6B shows, HEBI dos-dependently and significantly inhibited cell death of zebrafish irradiated with UVB. In addition, UVB significantly promoted the production of NO in zebrafish, while HEBI effectively suppressed the NO generation in a dose-dependent manner (Fig. 6C). HEBI at 25 µg/ml concentration reduced NO production from 326.86% to 141.40%. Moreover, HEBI also inhibited lipid peroxidation in zebrafish irradiated with UVB (Fig. 6D). The results above indicated that HEBI can attenuate the UVB-induced damage in zebrafish.
Discussion
UV radiation is a significant environmental element that contributes to skin cancer as well as photoaging (Kawashima et al., 2018). UVB is a wavelength band of UV radiation and when it irradiates the skin, the majority is absorbed by the epidermis and a minority portion penetrates into dermis (Fitsiou et al., 2021). After being absorbed by the epidermis, UVB induces intracellular redox processes to produce ROS (Schuch et al., 2017). Excess ROS will cause oxidative stress, resulting in cellular damage, leading to low cell viability and even apoptosis (Kawashima et al., 2018; Schuch et al., 2017). In addition, UVB photons can be directly absorbed by DNA, leading to DNA damage, which also triggers a number of cellular responses, including cell cycle arrest, DNA repair, and apoptosis (Schuch et al., 2017). As immortalized human keratinocytes, HaCaT cells are frequently utilized in research focusing on the human epidermis. In this study, we examined the photoprotective efficiency of HEBI using the HaCaT cell model. Our findings indicated that HEBI increased cell viability, reduced the level of ROS, and inhibited apoptosis in HaCaT cells induced by UVB. Therefore, we suggested that HEBI may protect the epidermis from UVB damage by enhancing cell viability through scavenging intracellular ROS and inhibiting apoptosis. Interestingly, the inhibitory effect of 25 μg/mL HEBI on the level of ROS production and apoptosis showed that HEBI was more effective in protecting cells from damage by inhibiting apoptosis. It should also be noted that HEBI has not yet been verified to have a protective effect against other types of epidermal cell damage caused by UV radiation, such as DNA damage and mitochondrial damage, and further research is needed.
Fibroblasts, in charge of synthesizing elements of the extracellular matrix (ECM), are the predominant cell type in the dermis (Fitsiou et al., 2021). UVB not only triggers the generation of intracellular ROS but also prompts fibroblasts to modify the ECM and influence the behavior of adjacent cells, leading to photodamage (Fitsiou et al., 2021). For example, UVB accelerates the breakdown of dermal ECM components, such as glycoproteins, elastin, and collagen. Moreover, exposure to UVB significantly elevates the production and release of MMPs, contributing to the degradation of ECM proteins (Salminen et al., 2022). UVB also promoted the release of cytokines (such as IL-1β, IL-6 and TNF-α) which can also promote the expression of MMPs and may cause inflammatory responses (Hasegawa et al., 2016). In a reported study, HEBI was suggested to possess a strong anti-inflammatory effect that could suppress NO generation and pro-inflammatory cytokine production in particulate matter-stimulated mouse lung macrophage (Asanka Sanjeewa et al., 2021). HDF cells are an important cell type present in the human dermis. We examined the protective efficiency of HEBI on HDF cells against damage induced by UVB in this study. The results showed that in UVB-exposed HDF cells, HEBI showed ROS inhibiting and cell viability improving effects. In addition, HEBI could effectively improve collagen synthesis while reduce MMPs and pro-inflammatory cytokines secretion. Therefore, we suggested that HEBI may protect the dermis from UVB damage by increasing collagen content through attenuating oxidative stress, reducing MMPs as well as pro-inflammatory cytokines expression.
Taken together, HEBI isolated S. horneri showed strong photoprotective effects on both epidermal and dermal cells. In a previous report, clerosterol, a sterol obtained from a green seaweed Codium fragile, improved the cell viability of HaCaT cells irritated by UVB from 60.5% to about 80% at a concentration of 3 μg/mL (Lee et al., 2013). Meanwhile, fucosterol, prepared from a brown seaweed H. fusiformis, reduced MMP-1 production by 46% and IL-6 secretion by 64% in UVB-irradiated NHDF cells at 10 μg/mL (Hwang et al., 2014). Compared with the present results, the photoprotective effect of HEBI on HaCaT cells was lower than that of clerosterol, but stronger than that of fucosterol on HDF cells. However, due to the different treatments of the cells in different studies, variables need to be controlled for a more in-depth comparison with these sterols. Additionally, in contrast to our previously reported compound (-)-loliode, which was also isolated from S. horneri, they had similar ROS scavenging abilities in UVB-induced HaCaT cells and HDF cells (Wang et al., 2021). However, 25 μg/mL HEBI had a stronger effect on cell viability enhancement in HaCaT cells irritated by UVB than (-)-loliode (around 80%).
In vitro cell modeling is efficient and results are intuitive. However, it does not involve the absorption and metabolism of substances and has certain limitations. As a consequence, animal experiments in vivo are necessary. Comparing with other vertebrates, zebrafish have significant advantages in terms of body size, feeding conditions, and early morphological observations. Zebrafish have become commonly used in pharmacological, toxicological, and biological studies in recent decades. UV-exposed zebrafish has proven effective for examining the photoprotective effects of agents (Wang et al., 2021). In this research, we discovered that HEBI could attenuate the generation of ROS and NO, as well as the lipid peroxidation and cell death triggered by UVB in zebrafish. Therefore, it can be concluded that HEBI is protective against UV-induced depletion injury both in vitro and in vivo.
Conclusion
The photoprotective effect of HEBI obtained from S. horneri was evaluated in the present research. The results showed that in HaCaT cells, HEBI attenuated UVB-induced damage by improving cell viability via scavenging intracellular ROS and inhibiting apoptosis. In UVB-mediated HDF cells, HEBI alleviated oxidative stress, reduced the expression of MMPs to increase collagen content, and decreased the expression of pro-inflammatory cytokines. In addition, HEBI mitigated UVB-induced ROS, NO production, lipid peroxidation and cell death in zebrafish. Thus, HEBI displayed strong photoprotective effects, indicating its potential application in cosmetics and pharmaceuticals.








