Introduction
Fish canning is a well-established preservation technique that encompasses multiple stages, including sample preparation, pre-cooking, filling, exhausting, sealing, sterilization, cooling, and labeling (Mayasari, 2013). A significant by-product of this process is skipjack tuna (Katsuwonus pelamis) body juice, which constitutes the largest volume of waste generated. This by-product is particularly rich in valuable water-soluble proteins, accounting for approximately 4% of its composition, along with other bioactive compounds (Choi et al., 2012). It is recovering water that is discarded due to environmental pollution, and the cooking juice is separated from the solid debris, centrifuged to separate fish oil and liquid, and the liquid is evaporated to obtain body juice.
Fish protein hydrolysate (FPH) is derived from fish proteins through enzymatic hydrolysis, resulting in smaller peptides that typically contain 2–20 amino acids. This hydrolysis process reduces peptide size, rendering proteins more bioavailable and functionally versatile for both human and animal consumption. FPH exhibits diverse bioactive properties, including antioxidant, antimicrobial, antitumor, angiotensin I-converting enzyme (ACE) inhibitory, calcium-binding, and anticoagulant activities. These functional properties make FPH a highly valuable ingredient in nutraceutical and therapeutic applications. A wide range of proteolytic enzymes is employed for the enzymatic hydrolysis of fish proteins, including neutrase. Neutrase has broad specificity with a preferred cleaved N-terminus Leu and Phe (Chalamaiah et al., 2012; Idowu et al., 2021; Toldrá et al., 2018).
The production of bioactive FPHs is a valuable approach for generating high-value products, leveraging their natural abundance and cost-effective extraction processes. Low-molecular-weight (MW) FPHs exhibit superior biological activity compared to larger MW fractions, as smaller peptides are more efficiently absorbed in the gastrointestinal tract (Heffernan et al., 2021). Various techniques are available for the separation and purification of bioactive peptides. In this study, the ultrafiltration (UF) membrane, polyethersulfone, was used because the UF is particularly suitable for large-scale peptide separation because of its ability to allow low-MW solutes and solvents to permeate while retaining higher-MW compounds (Kasiwut et al., 2015).
Enzymatic production of protein hydrolysates can be achieved through two main approaches: conventional batch hydrolysis and continuous hydrolysis using an UF membrane reactor (Sitanggang et al., 2021a). However, the batch method presents several significant drawbacks, including high enzyme costs owing to single-use applications, low productivity, inconsistent product quality, elevated labor costs, and substantial energy consumption. These limitations highlight the need for more efficient alternatives. In contrast, enzymatic membrane reactors (EMRs) offer a more cost-effective and sustainable solution. EMRs enable the reutilization of enzymes, thereby reducing overall enzyme costs while simultaneously enhancing productivity and maintaining product consistency (Chiang et al., 1995; Sitanggang et al., 2016). Hence, this approach not only addresses the inefficiencies of the conventional batch system but also aligns with the growing demand for scalable and economically viable processes for the production of bioactive protein hydrolysates.
Bioactive peptides derived from FPH can be directly consumed as food or dietary supplements to enhance protein intake (Nurilmala et al., 2018). The composition and bioactivities of FPHs are influenced by several factors, including the type of enzyme used for hydrolysis, substrate, enzyme concentration, pH, and duration of hydrolysis (Idowu et al., 2021). Notably, several studies have demonstrated that peptides generated from tuna cooking juice, as well as those hydrolyzed by alcalase in a continuous EMR, represent promising sources of bioactive peptides capable of inhibiting ACE activity (Kasiwut et al., 2015, 2019).
Additionally, irradiation of tuna cooking juice has been reported to enhance antioxidant properties, as evidenced by an increase in ferric reducing antioxidant power (FRAP) from 1,470 μM to 1,930 μM and a reduction in IC50 values for superoxide anion scavenging from 3.91 μg/mL to 1.29 μg/mL at 50 kGy (Choi et al., 2012). Hydrolysis of tuna cooking juice with protease XXIII demonstrated the highest 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical scavenging activity, with a value of 82.19%. Seven peptides with MW ranging from 457 to 1,010 Da have been isolated, featuring sequences composed of up to eight amino acids, including Val, Ser, Pro, His, Ala, Asp, Lys, Glu, Gly, and Tyr (Jao & Ko, 2002)
Furthermore, hydrolysis of dark muscles from skipjack tuna using various proteases, such as pepsin, trypsin, neutrase, papain, and alcalase, revealed that alcalase and neutrase hydrolysates exhibited superior antioxidant capacities against DPPH radicals, with EC50 values of 1.08 ± 0.08% and 0.98 ± 0.07%, respectively (Chi et al., 2015). Although extensive research has been conducted on the hydrolysis of tuna cooking juice, studies focusing on the production of antioxidative peptides from skipjack tuna cooking juice using neutrase in EMR remain scarce.
In light of this gap, the present study aimed to produce antioxidative peptides from skipjack tuna-cooked juice using EMR technology. The initial objective was to identify the optimal substrate-to-enzyme and enzyme-to-substrate ratios in a batch reactor to maximize the antioxidative peptide activity. Subsequently, antioxidative peptides derived from skipjack tuna cooking juice were produced in a continuous EMR and evaluated based on their DPPH, FRAP, and 2,2’-azino-bis(3-ethylbenzthiazoline)-6-sulfonic acid (ABTS) antioxidant activities.
Materials and Methods
Protease enzyme neutrase (0.8 L–12.000 U/mL) were procured from Novozyme (A/S Bagsværd, Denmark). Membranes with MW cut-offs of 2, 4, 5, and 10 kDa were purchased from Microdyn-Nadir (Wiesbaden, Germany). Chemicals and reagents, including Folin–Ciocalteu reagent, methanol, ethanol, FeCl−, NaOH, acetate buffer (pH 3.6), acetic acid, 37% HCl, 40 mM HCl, KH2PO−, Na2HPO−, and Na2CO−, were sourced from Merck (Darmstadt, Germany). Additional materials, such as trichloroacetic acid (TCA), DPPH powder, ABTS powder, potassium persulfate, TPTZ, Lowry B reagent, tyrosine, n-hexane, Trolox, and bovine serum albumin (BSA), were obtained from Sigma-Aldrich (St. Louis, MO, USA). Filter papers (Whatman No.1 and 3) were acquired from Hawach Scientific (Xi’an, China) and TOYO (Chiyoda, Japan). Tissue and ultrapure water were supplied by OneMed (Sidoarjo, Indonesia).
The sample, comprising body juice byproducts from skipjack (K. pelamis) canning (produced from precooking stage), was obtained from the PT. Aneka Tuna Indonesia, located in Pasuruan, East Java. Samples were collected from batch SJ 32.33.34.35.36, produced between March 2 and March 7, 2023, with a filling date of FJ-230314. Acquisition was conducted in May 2023.
The hydrolysis process was conducted using EMR equipped with essential components, including a pressure regulator, proportional pressure regulator, substrate tank, aquarium, water bath, water pump, cutting mat, and a cutter for membrane preparation. The experimental setup was controlled and monitored using LabVIEW 2019 (Laboratory Virtual Instrumentation Engineering Workbench, NI, USA).
The substrate-to-enzyme and enzyme-to-substrate ratios were optimized using a batch reactor. To determine the substrate-to-enzyme ratio, two substrate concentrations, 1% and 2% (w/v), were tested against a fixed enzyme concentration of 15% (w/v). The substrates (1% and 2% w/v) were prepared by dissolving them in 0.01 M phosphate buffer at pH 7. Each substrate concentration was subjected to hydrolysis in a batch reactor under controlled conditions: an enzyme concentration of 15% (w/v), a reaction temperature of 50°C, and a stirring speed of 300 rpm. Hydrolysates were collected at predetermined time intervals of 0, 1, 2, 4, 5, and 7 h to evaluate the hydrolysis process.
The hydrolysate was diluted with distilled water at a ratio of 1:10 (v/v) and homogenized for 1 h using a magnetic stirrer. The resulting mixture was filtered through filter paper No. 3. Subsequently, 40 mL of HCl was gradually added to the filtrate to adjust the pH to 4.5. A 45 mL aliquot of the adjusted liquid was transferred to a centrifuge tube and centrifuged at 8,000×g for 30 min at 21°C. Each hydrolysate sample was assessed for antioxidant capacity using DPPH, FRAP, and ABTS assays.
The optimal substrate concentration identified in the previous analysis is utilized in this section. The substrate was dissolved in 0.01 M phosphate buffer at pH 7 and subjected to hydrolysis in a batch reactor using enzyme concentrations of 10% and 20% (w/v). The reaction was conducted for 7 h under controlled conditions: a temperature of 50°C and a stirring speed of 300 rpm. Samples were collected at specified time intervals of 0, 1, 2, 4, 5, and 7 h. The antioxidant capacity of each hydrolysate was evaluated using DPPH, FRAP, and ABTS assays.
To identify the threshold flux corresponding to the optimal antioxidant capacity, three different flux values were evaluated: 10.38 LMH (retention time: 7 h), 8.08 LMH (retention time: 9 h), and 6.07 LMH (retention time: 12 h). These tests were conducted using a 10 kDa membrane. The hydrolysis of body juice protein was performed using a substrate concentration of 2% (w/v) and neutrase enzyme concentration of 15% (w/v), with each condition applied to varying flux values.
The continuous reactor was utilized to evaluate the threshold flux at retention times of 7, 9, and 12 h and to produce bioactive peptides. The body juice sample was dissolved in 0.01 M phosphate buffer solution at pH 7 (w/v). The optimal enzyme concentration and substrate concentration, identified in prior experiments, were used in EMR. The membrane was prepared by cutting it to size and soaking it in pure water for 15 min with the glossy side facing downward. The membrane was then positioned at the bottom of the reactor, and the reactor was subsequently filtered.
The reactor was filled with body juice to approximately three-quarters of its volume, after which the enzyme (15% w/v) was added, followed by body juice. The reactor was operated for 7 h under controlled conditions: a temperature of 50°C, a stirring speed of 300 rpm, and a constant flux. Hydrolysates were collected at time intervals of 0, 2, 4, 5, and 7 h, as well as at the conclusion of the operation. The antioxidant capacity of each hydrolysate was analyzed using the DPPH, FRAP, and ABTS assays, and the protein content was determined using the Lowry method.
The production of bioactive peptides was conducted under optimized conditions, including a substrate concentration of 2% (w/v), an enzyme concentration of 15% (w/v), and a threshold flux of 8.08 LMH with a retention time of 9 hours. The process was run for 7 h at a temperature of 50°C. The resulting hydrolysate was separated by membrane filtration with molecular weight cut-offs (MWCO) of 2, 4, 5 kDa. Permeate samples were collected both before and after the operation and subsequently analyzed for antioxidant capacity using the DPPH assay.
The antioxidant capacity of the hydrolysates was evaluated using the DPPH, FRAP, and ABTS assays. The DPPH assay was conducted as described by (Sitanggang et al., 2021b), with Trolox used as the standard, an analog of vitamin E with known antioxidant activity (Iosageanu et al., 2021). A 0.3 mL of hydrolysate was added with 0.7 mL of distilled water, and 3.0 mL of 80 mM 2,2-diphenyl-1-picrylhydrazyl (DPPH). The solution was incubated for 30 minutes in a dark room at room temperature (25°C–30°C), and the absorbance was measured at 515 nm. Distilled water was used as a blank. The antioxidant capacity was expressed in μM TE/mL, with values calculated based on the standard curves of Trolox: y = –0.0024x + 0.949 (R2 = 0.9992) for the DPPH assay.
For the FRAP assay, 150 mL of the sample was reacted with 4.5 mL of FRAP reagent, and Trolox was used as the standard (Benzie & Strain, 1996). The vortexed solution was incubated in a dark room at a temperature of 30°C for 30 minutes, and the absorbance was measured at 593 nm. The antioxidant capacity was expressed as μM TE/mL, with values calculated based on the standard curves of Trolox: y = 0.0025x – 0.0053 (R2 = 0.9999) for the FRAP assay.
The ABTS assay, as outlined by (Puchalska et al., 2014), was used to measure antioxidant capacity, which was expressed as percentage inhibition. 1 mL of hydrolysate was added to 1 mL ABTS reagent. The absorbance was measured at 734 nm.
Protein content was quantified following the method described by Lowry et al., with BSA used as the standard (Lowry et al., 1951). A 0.3 mL of hydrolysate was added with 3.5 mL mL distilled water, and 5.5 mL reagent C. The vortexed solution was incubated for 15 minutes at a room temperature (21°C–25°C). Then, 0.5 mL reagent D was added to the incubated solution, and it was re-incubated for 30 minutes at a temperature 21°C–25°C. The absorbance was measured at 650 nm. The protein content was expressed as mg/mL and calculated based on the standard curve of BSA using the equation y = 0.005x + 0.0071 (R2 = 0.9914). Antioxidative peptide recovery was 55.28%.
Results
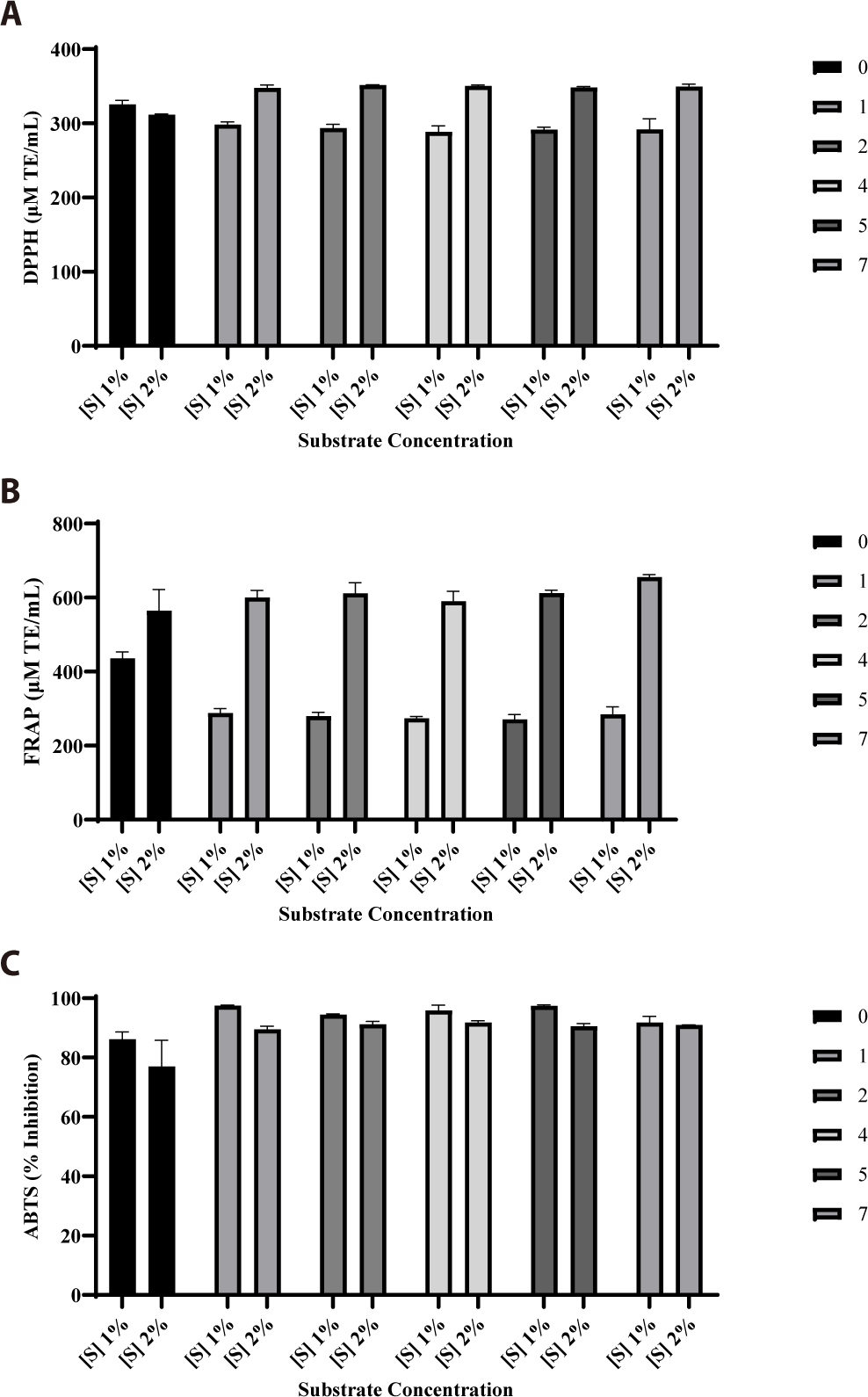
The substrate-to-enzyme ratio was determined using substrate concentrations of 1% and 2% skipjack tuna body juice, which was dissolved in phosphate buffer at pH 7. The substrate was reacted with 15% (w/v) neutrase enzyme for 7 h, and hydrolysates were collected at various time points, including 0, 1, 2, 4, 5, and 7 h. The antioxidant activities of the samples were subsequently measured using DPPH, FRAP, and ABTS assays (Fig. 1). Based on these measurements, the 2% substrate concentration with 15% enzyme concentration demonstrated superior antioxidant activities. The DPPH, FRAP, and ABTS assay results were 349.48 μM TE/mL, 655.40 μM TE/mL, and 90.93%, respectively.
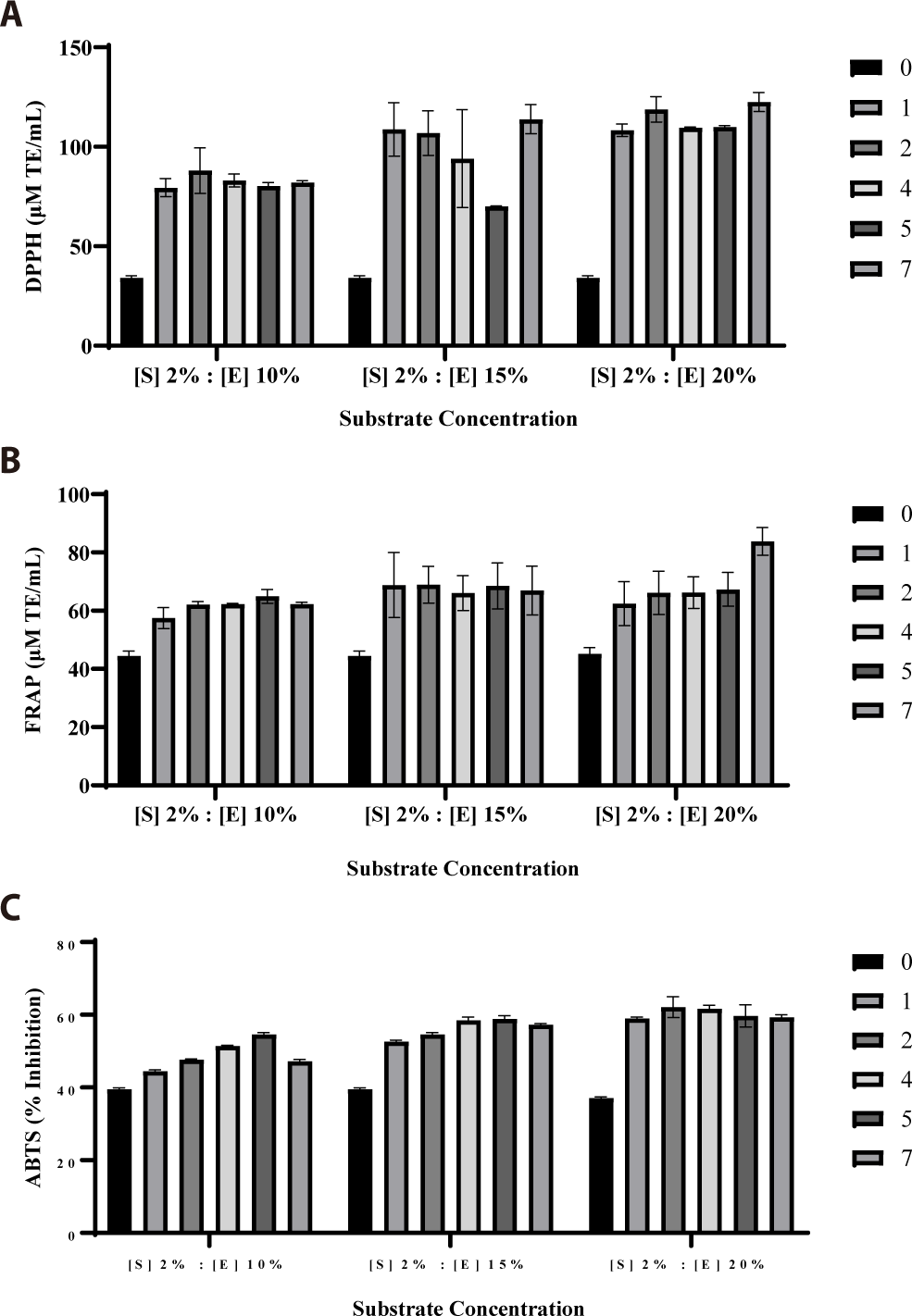
An optimal substrate concentration of 2%, identified in the previous step, was used to determine the enzyme-to-substrate ratio. Neutrase enzyme concentrations of 10%, 15%, and 20% (w/v) were reacted with the 2% (w/v) substrate concentration in a batch reactor for 7 h at 50°C and pH 7. Hydrolysates were sampled at time intervals of 0, 1, 2, 4, 5, and 7 h and assessed for antioxidant activity using the DPPH, FRAP, and ABTS assays. As shown in Fig. 2, the enzyme concentration of 15% and substrate concentration of 2% resulted in the highest production of antioxidative peptides, with DPPH, FRAP, and ABTS activities of 113.85 μM TE/mL, 66.68 μM TE/mL, and 57.29%, respectively.
The production of bioactive peptides requires the determination of the optimal flux size, which governs the contact time between the substrate and enzyme. Residence time, defined as the duration for which the substrate remains in the reactor, plays a critical role in modulating the interaction between the enzyme and substrate. Thus, optimizing the residence time is essential to prevent both under- and over-hydrolysis of the product. Flux sizes were determined based on the residence time, which was calculated using the volumetric flow rate, membrane area, and reactor volume (Sitanggang et al., 2022). In this study, three distinct residence times were assessed: 7, 9, and 12 h, corresponding to flux values of 10.38 LMH (7 h), 8.08 LMH (9 h), and 6.07 LMH (12 h).
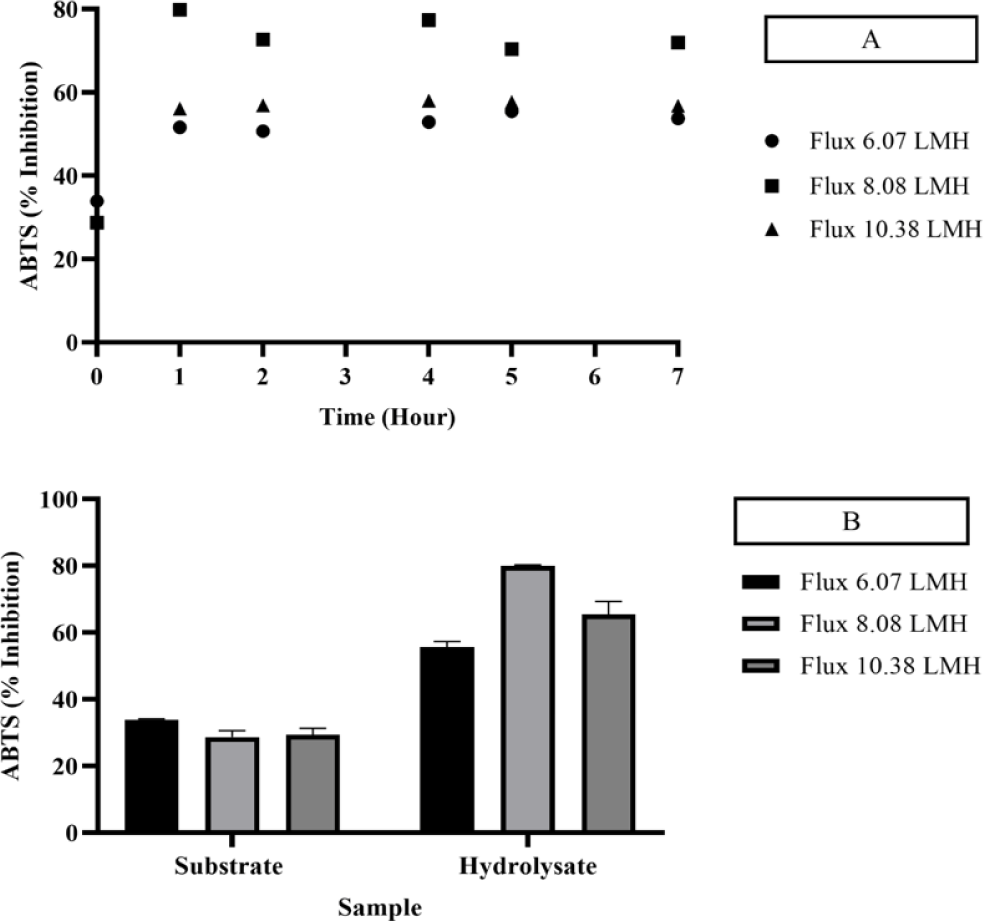
Hydrolysis of skipjack tuna body juice was conducted continuously within an EMR system, employing a 2% substrate concentration and a 15% neutrase enzyme concentration for a duration of 7 h at 50°C. The reactor was equipped with a 10 kDa membrane, maintained at a rotational speed of 300 rpm, and operated under a constant flux 8.08 LMH. The threshold flux was determined by evaluating the antioxidant activities, which were measured using DPPH (Figs. 3 and 4), FRAP (Fig. 5), and ABTS (Fig. 6) assays. Variations in flux size resulted in differing antioxidant activity levels, which were indicative of the influence of flux on enzymatic hydrolysis.
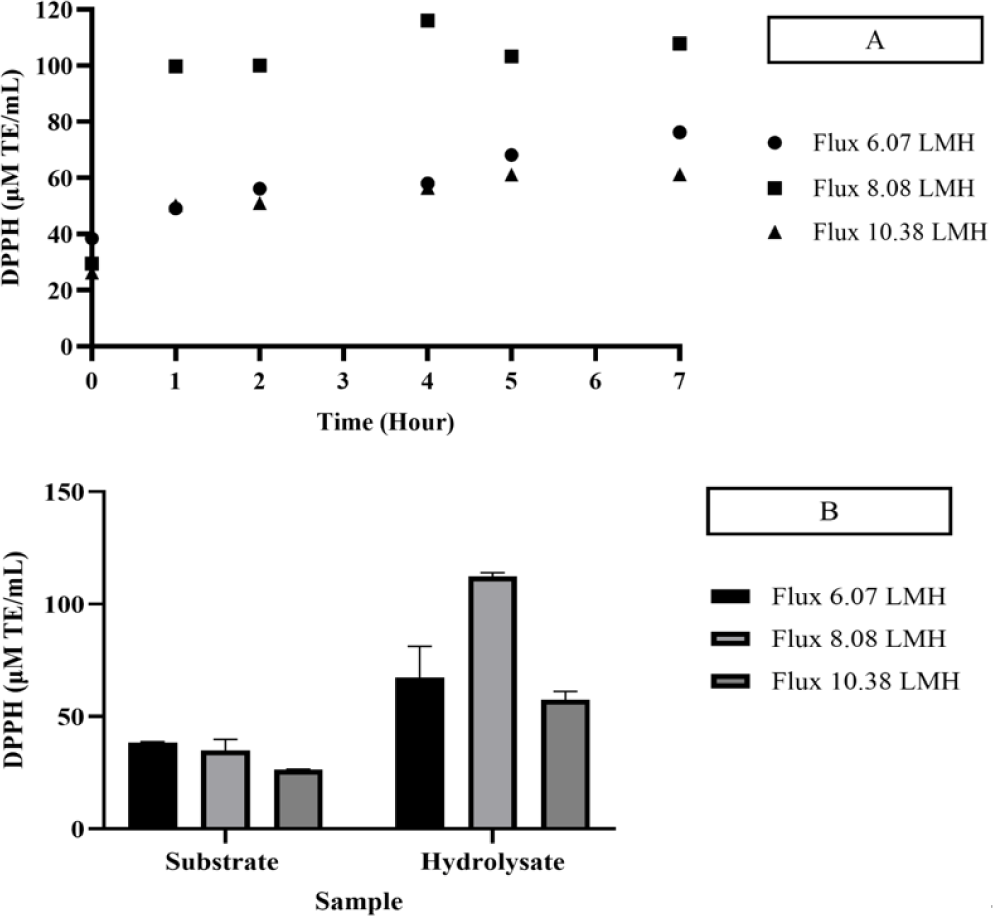
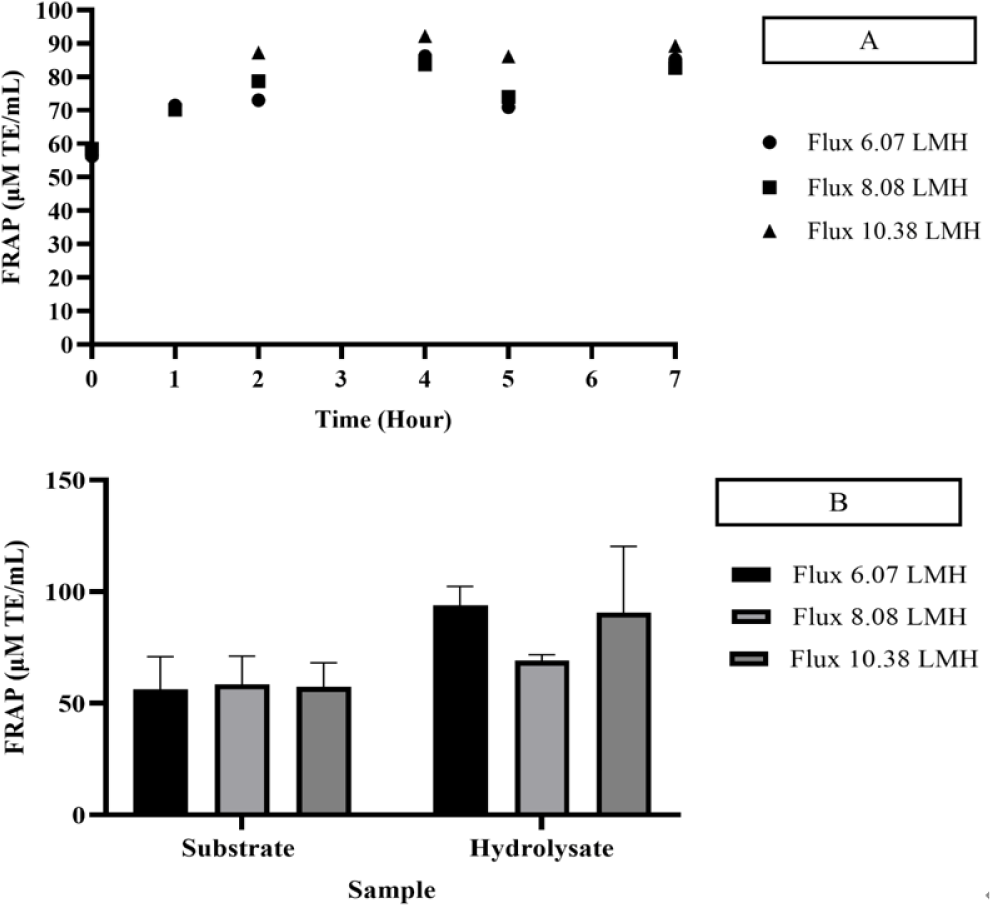
The protein content and antioxidant capacity of the permeate remained stable over a residence time of 9 h at a flux rate of 8.08 LMH. Specifically, the protein content during this extended residence period was determined to be 0.128 mg/mL. This finding suggests that prolonged residence times facilitate increased protein content, which is likely attributable to the extended duration available for enzymatic activity to effectively break down peptide bonds within the product.
The antioxidant activity of the permeate was evaluated using DPPH, FRAP, and ABTS assays. The DPPH assay recorded values of 29.48 μM TE/mL for the substrate and 112.29 μM TE/mL for the hydrolysate, while the FRAP assay yielded values of 58.280 μM TE/mL for the substrate and 68.78 μM TE/mL for the hydrolysate. Similarly, the ABTS assay reported antioxidant activities of 28.71% and 80.04% for the substrate and hydrolysate, respectively. Notably, the DPPH and ABTS assays demonstrated higher and stable antioxidant activities at a flux rate of 8.08 LMH, with significant enhancements compared to other flux rates. The increase in antioxidant activity was attributed to the presence of specific amino acids, including histidine, methionine, cysteine, phenylalanine, and tyrosine, which have been recognized for their role in enhancing DPPH and ABTS activity (Chalamaiah et al., 2015).
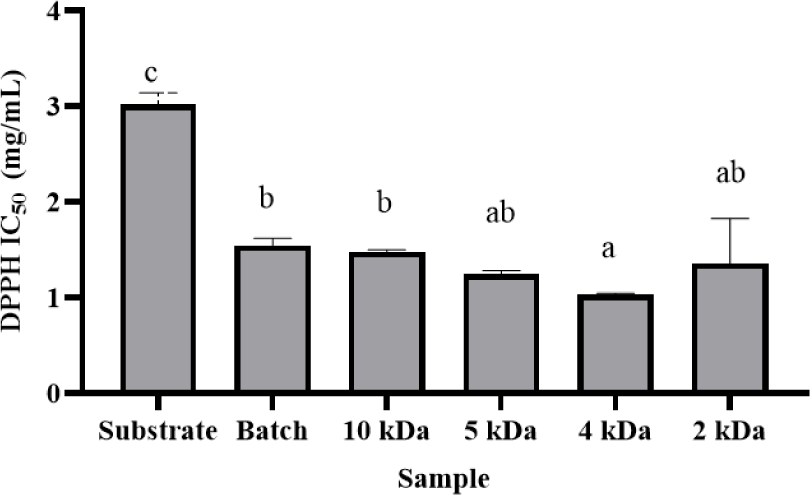
Antioxidative peptides from skipjack tuna body juice were successfully produced using a continuous EMR system. The process parameters were optimized with a substrate concentration of 2%, enzyme concentration of 15%, a membrane MWCO of 10 kDa, and a threshold flux of 8.08 LMH. The system was operated for 7 h at 50°C with a stirring speed of 300 rpm. To fractionate the hydrolysate, membranes with MWCOs of 2, 4, and 5 kDa were used. The determination of antioxidant activity was conducted using IC50 values derived from DPPH assays, as this method proved most effective for evaluating the antioxidative potential at the 7-hour time point (Fig. 7).
Discussion
In Aneka Tuna Indonesia factory, both yellowfin tuna (YF) and skipjack are utilized for raw materials of canned products. Canned tuna or skipjack produce different by-products, such as heads, viscera, during the sorting step, and body juice from pre-cooking steps. The last byproduct is generated from fish, either YF or skipjack, evaporated liquids, which are processed into a concentrated form of a fish-soluble concentrate called body juice. Usually, the body juice is for aquaculture purposes and animal feed. The notion is to increase the value-added of the body juice by hydrolysis of the body juice to obtain the antioxidative peptide. This study aimed to produce antioxidative peptides from skipjack tuna body juice using a continuous EMR system. Antioxidants are substances that inhibit oxidation processes (Utami, 2020). The generation of reactive oxygen species and free radicals during cellular respiration in humans is associated with the onset of various diseases, including diabetes, inflammation, cancer, neurodegenerative disorders such as Alzheimer’s and Parkinson’s diseases, aging-related complications, and hypertension. FPH have emerged as promising sources of antioxidative peptides. During enzymatic hydrolysis, peptide bond cleavage liberates bioactive peptides capable of chelating pro-oxidant metal ions, scavenging oxygen radicals, and inhibiting lipid peroxidation in food systems (Ng et al., 2024). The antioxidative activity of these peptides was assessed using DPPH, FRAP, and ABTS assays, as employing a single method is insufficient to comprehensively evaluate the antioxidant capacity of a compound (Chalamaiah et al., 2015).
Initially, the optimal substrate-to-enzyme ratio for producing antioxidative peptides was determined using a batch reactor system based on antioxidant activity measurements. The combination of a 2% substrate concentration and a 15% enzyme concentration demonstrated superior antioxidative activity, with DPPH, FRAP, and ABTS values of 349.48 μM TE/mL, 655.40 μM TE/mL, and 90.93%, respectively. When subjected to continuous hydrolysis, this ratio produced higher antioxidative peptides, yielding DPPH, FRAP, and ABTS activities of 113.854 μM TE/mL, 66.68 μM TE/mL, and 57.29%, respectively. These findings indicate that enzymatic reaction efficiency and substrate concentration significantly influence hydrolysate antioxidant capacity.
The highest antioxidative peptide activity was achieved with a 2% substrate concentration hydrolyzed by a 15% enzyme concentration over 7 h, outperforming other tested ratios. Conversely, a substrate concentration of 1% exhibited reduced activity, which was attributed to an excess of unutilized enzymes due to substrate limitation (Vitolo, 2020). Chi et al. similarly demonstrated that hydrolysates produced using alcalase and neutrase enzymes after a 4-hour hydrolysis period exhibited significantly higher DPPH scavenging activities, with EC50 values of 4.54 ± 0.43 mg/mL and 5.38 ± 0.15 mg/mL, respectively. In contrast, trypsin hydrolysates displayed the lowest activities (Chi et al., 2015). Roslan et al. further emphasized that excessive enzyme concentrations and prolonged reaction times were inefficient due to increased operational costs. Factors such as hydrolyzable bond concentration and hydrolysis duration play critical roles in controlling the hydrolysis rate, peptide MW distribution, and functional properties of FPH (Roslan et al., 2014).
In the initial phase of this study, the threshold flux was determined by evaluating both antioxidant activity and protein content. It was observed that at a residence time of 9 h (8.08 LMH), the protein content and antioxidant capacity of the permeate remained stable. Extending the residence time beyond 9 h, up to 12 h, resulted in a decline in antioxidant activity, likely due to the accumulation of under-hydrolyzed products. The 9-hour residence time represented an optimal balance, where the interaction between the enzyme and substrate was most effective, facilitating maximum antioxidant activity.
The duration of residence time critically influences enzyme performance, because prolonged exposure can lead to enzyme degradation during continuous EMR operations. Conversely, shorter residence times may limit the enzyme’s effectiveness in catalyzing the hydrolysis reaction, thereby reducing the product yield and activity (Sitanggang et al., 2022). These findings highlight the importance of optimizing reaction parameters to maintain enzyme stability and efficiency while maximizing antioxidative peptide production.
The antioxidative peptides generated from skipjack tuna body juice via a continuous EMR system were assessed through IC50 values determined by the DPPH assay. The IC50 values for the substrate, batch hydrolysate, and hydrolysates filtered using membranes with MWCOs of 10, 5, 4, and 2 kDa were 3.02 ± 0.12, 1.54 ± 0.08, 1.48 ± 0.02, 1.25 ± 0.04, 1.03 ± 0.02, and 1.36 ± 0.47, respectively. As depicted in Fig. 7, hydrolysates processed through the 4 kDa membrane exhibited superior antioxidant activity compared to other filtrates.
The enhanced efficacy of membranes with smaller MWCOs can be attributed to their capacity to continuously produce lower MW peptide fractions while effectively retaining enzymatic components within the reactor (Sitanggang et al., 2015). These observations are consistent with previous findings, indicating that peptide size substantially influences the antioxidant capacity of protein hydrolysates and their respective fractions. Hydrolysates from dark muscle skipjack tuna, when filtered through a 3 kDa MWCO membrane, demonstrated significantly higher antioxidant activity than those processed with larger membranes due to the superior interaction of smaller peptides with reactive radicals, thereby mitigating oxidative processes (Chi et al., 2015).
Furthermore, Hsu (2010) suggested that peptides function as electron donors, stabilizing reactive radicals, and effectively terminating chain reactions that propagate oxidative damage. MW is a critical determinant of antioxidant activity, with smaller peptides enriched in aromatic and hydrophobic amino acid residues exhibiting greater efficiency in interrupting lipid peroxidation pathways by readily neutralizing free radicals (Sierra-Lopera & Zapata-Montoya, 2021). Recent studies have demonstrated that FPH and peptides exhibiting antioxidant properties can be released through the enzymatic hydrolysis of fish proteins. The findings of this study align with those reported by (Halim et al., 2018) where eel protein hydrolysates filtered through a 3 kDa membrane exhibited the highest DPPH radical scavenging activity compared to crude hydrolysates and those filtered using 10 kDa and 5 kDa membranes.
The enhanced antioxidant activity observed with the smaller MW fractions is attributed to the increased hydrogen availability at the N-terminal regions, which facilitates the scavenging of free radicals through hydrogen atom donation. This mechanism underscores the potential of hydrolysates to function as secondary antioxidants by effectively mitigating oxidative damage and inhibiting radical propagation.
In conclusion, the findings of this study emphasize the efficacy of utilizing a continuous EMR system coupled with Neutrase to valorize by-products from skipjack tuna (K. pelamis) canning into antioxidative peptides, with substantial potential for nutraceutical applications. Optimization of the substrate-to-enzyme ratio (2% substrate and 15% enzyme) and operational parameters within the EMR system resulted in the production of peptides with heightened bioactivity, as substantiated by the enhanced results of the DPPH, FRAP, and ABTS antioxidant assays. Membrane fractionation employing various MWCOs (10, 5, 4, and 2 kDa) revealed that the smaller peptide fractions exhibited superior antioxidant properties, thereby highlighting the critical role of molecular size in modulating bioactivity.
Furthermore, this study provides novel insights into the efficient hydrolysis of tuna cooking juice for the generation of bioactive peptides. The continuous EMR system not only facilitated the attainment of high antioxidative activity, but also addressed the key limitations of conventional batch hydrolysis methodologies, including suboptimal enzyme reutilization, scalability constraints, and inconsistencies in product quality. By preserving the protein content and antioxidative capacity under optimized flux conditions, this work underscores the feasibility of large-scale applications.
The enhanced performance of peptide fractions processed through membranes with smaller MWCOs presents opportunities for refining peptide isolation strategies to further augment bioactivity. The demonstrated capacity of these antioxidative peptides to scavenge free radicals and chelate metal ions makes them promising candidates for functional foods and pharmaceutical interventions aimed at mitigating oxidative stress-related pathologies.
Hence, the integration of neutrase-assisted continuous EMR technology offers a transformative approach to the sustainable management of fish canning by-products, providing both economic and environmental advantages. Future investigations could focus on further bioactivity profiling and the development of applications to fully exploit the therapeutic potential of these peptides.








