Introduction
Global aquaculture production reached about 130.9 million tonnes in 2022, with Africa contributing approximately 1.9% (≈ 2.5 million tonnes) (FAO, 2024). Egypt, Nigeria, and Uganda collectively account for nearly 90% of Africa’s output, dominated by indigenous species such as Nile tilapia (Oreochromis niloticus) and African catfish (Clarias gariepinus) (Adeleke et al., 2020; FAO, 2018). Uganda’s fish export industry has grown substantially, with export earnings rising from USD 1.4 million in 1990 to over USD 120 million by 2021 (FAO, 2020). In parallel, fish consumption in East Africa has continued to rise, with per capita intake projected to increase from 4.8 kg in 2013 to 5.5 kg by 2022 (van Duijn et al., 2018), reinforcing the need for expanded production (Denis et al., 2020; Obiero et al., 2019).
Declining wild fish catches—from roughly 470,000 tonnes in 2011 to 389,000 tonnes in 2020, a reduction of nearly 17%—have accelerated the intensification of aquaculture to close the national fish supply gap (FAO, 2020; Ssempijja, 2019). This expansion, however, has increased the movement of live fish within Uganda and across its borders, raising biosecurity risks, particularly from broodstock and seed of unknown health status (Opiyo et al., 2018). Previous analyses identified major gaps in East Africa’s aquatic biosecurity framework, including limited research on priority pathogens, weak diagnostic capacity, insufficient regulation of therapeutics, and inadequate quarantine and risk assessment systems (Akoll & Mwanja, 2012). Yet, practical strategies for addressing these weaknesses in Uganda have received limited attention.
This paper therefore reviews Uganda’s aquaculture production systems, examines recent developments in fish health legislation and biosecurity governance, and proposes pragmatic, scalable measures to strengthen aquatic biosecurity and disease preparedness in the country.
Materials and Methods
This study was based on a structured review of published scientific literature, institutional reports, and international databases related to aquaculture production, fish health, and biosecurity governance in Uganda. Peer-reviewed articles were retrieved from major academic databases including Web of Science, Scopus, and Google Scholar. Additional information was obtained from official reports and policy documents issued by the Ministry of Agriculture, Animal Industry and Fisheries (MAAIF), the National Agricultural Research Organisation (NARO), and the National Fisheries Resources Research Institute (NaFIRRI).
Aquaculture production statistics and regional trends were compiled primarily from the Food and Agriculture Organization (FAO) databases and reports. Information on aquatic animal disease occurrence and international disease reporting was obtained from the World Organisation for Animal Health (WOAH) World Animal Health Information System (WAHIS). Government policy documents, legislation, and regulatory frameworks were also examined to assess institutional arrangements for fish health management and biosecurity.
The collected information was organized and analyzed under three thematic components relevant to aquaculture development in Uganda. First, the structure and production characteristics of the aquaculture sector were reviewed, including seed production systems, grow-out practices, and production trends. Second, available evidence on fish disease occurrence, pathogen reports, and existing health management practices was examined to assess the current disease risk landscape. Third, national biosecurity governance and legal frameworks were analyzed, including legislation, regulatory institutions, and implementation challenges.
Selected case examples were included to illustrate practical developments in Uganda’s aquaculture sector, particularly in relation to cage aquaculture expansion and regulatory reforms affecting fish health management. These examples were derived from government reports, field observations reported in previous studies, and sectoral assessments conducted by international development agencies. The case studies are intended to provide contextual insights into emerging trends in farm management, biosecurity adoption, and market development. Data on farmer perceptions were also obtained through field interviews conducted in 2023 with cage farmers operating along Lake Victoria in Mukono District.
Results
Uganda, a landlocked country in East Africa, is well suited to aquaculture thanks to abundant water resources, with wetlands and open water covering about 18% of its land area (Nsubuga, 2014). The industry primarily focuses on freshwater finfish—especially Nile tilapia (O. niloticus) and African catfish (C. gariepinus)—which perform well under Uganda’s climate and match local market preferences (FAO, 2022b, 2024; NARO, 2021). Reflecting this biological and market suitability, the Ministry of Agriculture, Animal Industry and Fisheries (MAAIF) has identified 31 of Uganda’s 135 districts as priority areas for aquaculture development, most of which are located near major water bodies (MAAIF, 2020). These 31 districts represent the primary aquaculture zones in Uganda (Fig. 1).
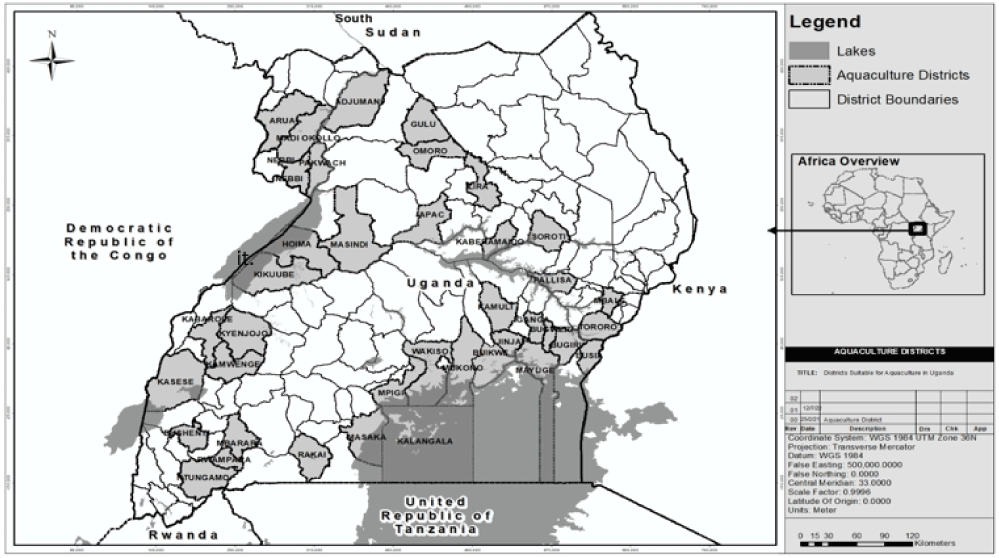
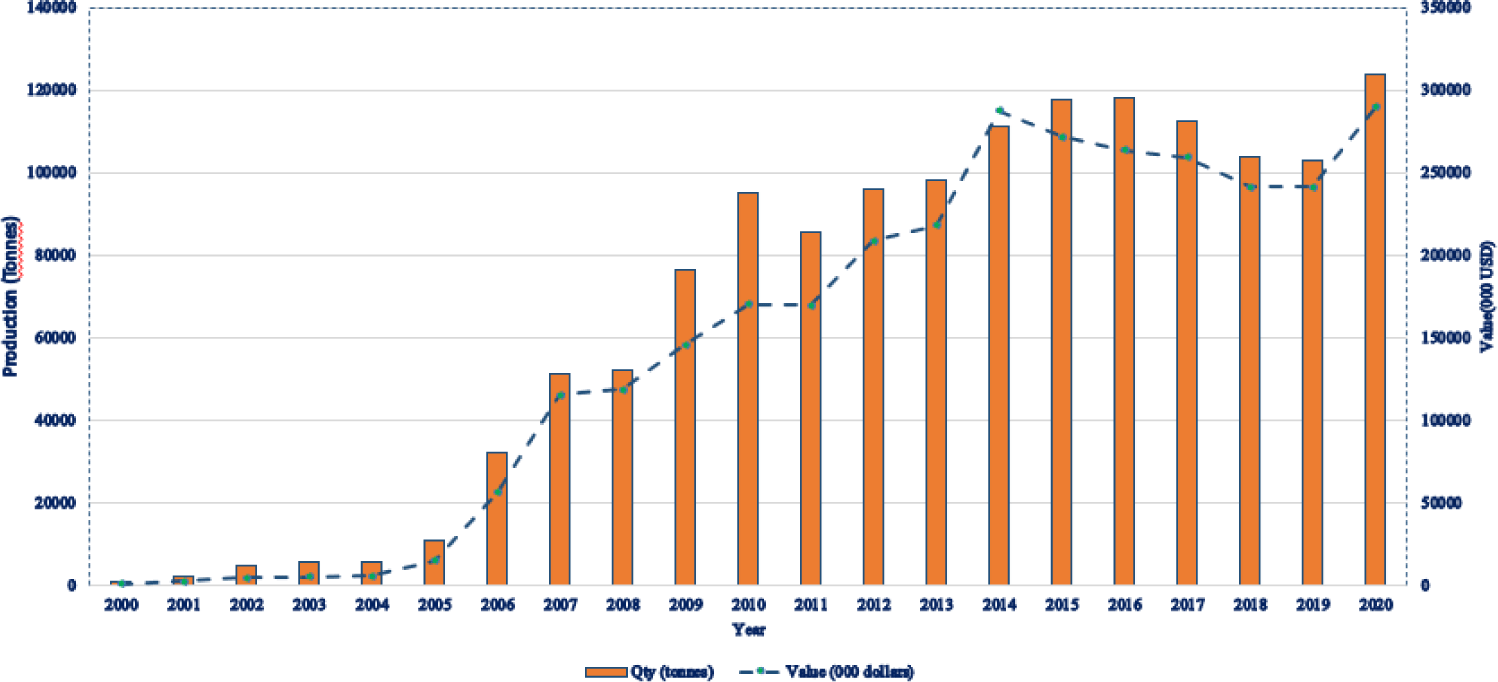
Aquaculture in Uganda dates back to the 1960s, initially centered on common carp (Cyprinus carpio) and Nile tilapia. By 1968, more than 11,000 ponds had been established, yet growth was constrained by reliance on informal seed exchanges among farmers. During the 1980s, shortages of quality seed and feed, limited technical support, and inconsistent policies prompted many farmers to abandon aquaculture. The sector revived in the 1990s as interest and investment returned. Production rose steadily, reaching 10,817 metric tonnes in 2005 and increasing to 123,897 metric tonnes—valued at approximately USD 289.5 million—by 2020 (Fig. 2). The sector is currently expanding at roughly 15% per year, with about 20,000 fish farmers operating 25,000 ponds and more than 3,000 cages. Aquaculture now contributes around 20% of national fish output, underscoring its importance within Uganda’s fisheries sector (MAAIF, 2020). Production has increased steadily since the early 2000s (Fig. 2), highlighting the sector’s growing role.
The recent expansion reflects converging economic, demographic, and environmental drivers (Netherlands Enterprise Agency [RVO], 2022; MAAIF, 2021). Increased private-sector investment—enabled by improved access to credit, technology adoption, and targeted government incentives—has fueled commercial development (RVO, 2022; MAAIF, 2021). Robust local and regional demand, sustained by population growth and evolving dietary preferences, has further accelerated growth (FAO, 2022b). At the same time, declines in capture fisheries due to overfishing and ecosystem pressures have heightened the strategic importance of aquaculture as a sustainable alternative (FAO, 2022b; RVO, 2022). Strong government support—spanning infrastructure investments, policy reforms, and capacity-building—has also played a role (MAAIF, 2021). Uganda’s favorable year-round climate, with average temperatures of 25–29°C and annual rainfall of 1,000–1,500 mm, enables continuous production cycles (Sserwambala, 2018).
Uganda’s aquaculture industry is a critical engine for employment, income generation, and food security, supporting an estimated 140,000 livelihoods directly and indirectly, including farmers, processors, and traders (FAO, 2022b). Fish accounts for over 50% of total animal protein intake in the average Ugandan diet, serving as a nutritional cornerstone for both urban and rural households (FAO, 2022b). Per capita fish consumption increased from 12.5 kg in 2013 to 13.5 kg in 2017—above the African average of 10.1 kg but still below the global average of 20.5 kg (FAO, 2017). Approximately 70%–75% of domestically produced fish is consumed within Uganda, bolstering local food and nutrition security, particularly in lower-income and rural communities (RVO, 2022). Overall, the broader fisheries sector contributes about 1.9% of national GDP and nearly 8% to agricultural GDP (MAAIF, 2021).
Under Section 29(1) of the Fish (Aquaculture) Rules, 2003, the production, distribution, or sale of fish seed requires a Fish Seed Production Certificate issued by the Chief Fisheries Officer (The Republic of Uganda, 2003). According to NARO (2021), Uganda had approximately 56 operational hatcheries, which collectively produced about 164 million fingerlings in 2021—consisting mainly of Nile tilapia (87.2%), African catfish (10.2%), and mirror carp (2.7%) (RVO, 2022). While this is the actual production figure, theoretical or installed hatchery capacity is estimated at around 300 to 400 million fingerlings annually, assuming full operational efficiency and optimal broodstock quality. This gap between actual and potential output reflects several constraints, including infrastructure limitations, seasonal production cycles, and technical capacity. To meet the national aquaculture production target of 1 million tonnes by 2025, the projected seed demand is approximately 2.5 billion fingerlings, highlighting a critical supply gap (MAAIF, 2021; RVO, 2022). Seed production techniques vary by species: catfish hatcheries typically use tank-based strip spawning, while tilapia seed is produced via natural spawning in dam-lined or earthen ponds (Matthew et al., 2015). Hatcheries are commonly categorized by output scale as small (~ 300,000 fingerlings/year), medium (300,000–1 million), and large-scale (over 1 million)—serving both local farmers and regional markets (Mwanja et al., 2007).
Despite advances, hatcheries face persistent challenges, notably high larval and fry mortality. While disease is commonly suspected—based on symptoms such as skin discoloration, opaque eyes, and hemorrhages—comprehensive etiological investigations are scarce, and the exact causes of mortality remain largely unverified due to diagnostic limitations (Akoll et al., 2012; Matthew et al., 2015).
Pond culture remains the dominant grow out method, particularly among small-scale farmers, with most of the country’s 25,000 ponds operating under extensive or semi-intensive systems (FAO, 2022b; Nazziwa, 2021). Extensive ponds use low stocking densities (approximately ≈ 3 fry/m2) and depend largely on natural productivity and organic fertilization to support feed and water quality (FAO, 2022b). Semi-intensive systems supplement natural productivity with commercial or farm-made feeds (e.g., maize bran) and incorporate apply some water management practices, although biosecurity and disease control are typically limited (van Duijn et al., 2018). Stocking densities of about 3 fish/m2 are most common, and typical yields range from 0.8–1.5 kg/m2 per 6–8-month cycle, with performance varying by farm management (Sserwambala, 2018). Pond sizes range from < 100 m2 to ≈ 6,000 m2, with 500–1,000 m2 most common (Sserwambala, 2018). Organic fertilizers, such as livestock manures, are widely used to enhance pond fertility and production (Opiyo et al., 2018).
Farming approaches include:
-
Monoculture: most commonly tilapia or catfish.
-
Polyculture: tilapia with catfish or common carp.
-
Integrated farming: fish ponds combined with vegetable or livestock production.
Fig. 3. illustrates the diversity of aquaculture systems in Uganda, ranging from small-scale earthen ponds to cage and tank-based production units.

Cage culture has expanded rapidly since its introduction to major lakes in 2006 and now accounts for a large share of commercial production, especially on Lake Victoria (Aura et al., 2024; Nazziwa, 2021). More than 3,000 cages are currently in operation, with cage-based tilapia farming accounting for as much as 70% of national aquaculture output (Nazziwa, 2021). The most common configuration is low-volume high-density (LVHD) cages of about 8–16 m³, typically stocked at 140–250 fish/m³ (Mbowa et al., 2017; NaFIRRI, 2018). Commercial-scale farms may employ larger cages at slightly lower densities (Aura et al., 2024; Nazziwa, 2021). LVHD cages can yield 60–80 kg/m³ per cycle, while high-volume low-density systems typically yield 20–40 kg/m³. Cages are designed for high survival rates (> 85%), and Nile tilapia generally reach market size (≈ 500 g) within 6–8 months (Aura et al., 2024; Nazziwa, 2021; Ogutu-Ohwayo et al., 2021). While LVHD cages offer high productivity, their high stocking densities inherently increase the risks of water quality deterioration and rapid pathogen transmission. Regular monitoring, waste management, and health inspections are therefore critical to mitigate potential environmental degradation and disease outbreaks (Aura et al., 2024; Ogutu-Ohwayo et al., 2021). Feeds used in Uganda’s aquaculture include both commercial pellets and farm-made formulations, typically based on maize bran, cottonseed cake, and fishmeal (FAO, 2022b). However, inconsistent nutrient profiles and lack of quality assurance in artisanal feeds may contribute to nutritional deficiencies, reduced growth performance, and increased disease susceptibility.
Since the introduction of cage technology on Lake Victoria, aquaculture productivity and rural household incomes in Uganda have increased markedly. Pilot farms employing low-volume high-density (LVHD) cages reported yields of up to 80 kg/m³ per production cycle (Mbowa et al., 2017). By 2023, more than 2,000 farming households were reported to have achieved more stable incomes and gained access to premium tilapia export markets. The combination of targeted extension services and national regulatory reforms appears to have improved farm profitability and contributed to reduced disease-related losses.
The implementation of cage registration and routine health monitoring has been associated with lower disease incidence and improved product quality assurance. Farmers operating in Mukono District on Lake Victoria consistently reported greater confidence in biosecurity practices and better market market opportunities (field interviews with cage farmers in Mukono District, 2023).
Fish Disease Occurrence and Health Management in Uganda
Fish diseases present a major economic challenge to Ugandan aquaculture, arising from complex interactions among host, pathogen, and environmental factors. They are broadly classified as infectious—caused by bacteria, viruses, fungi, or parasites—or non-infectious, resulting from environmental stressors, nutritional deficiencies, or genetic anomalies (Komarudin & Slembrouck, 2003; MAAIF, 2017).
In Uganda, official reports of fish diseases are relatively rare, and no major outbreaks have been formally documented (van Duijn et al., 2018). However, this likely reflects limited diagnostic capacity and weak surveillance systems rather than the actual absence of disease. Most small-scale farmers lack the training and tools needed to recognize disease signs, and reporting is often informal or anecdotal (Walakira et al., 2014). Published research have largely focused on the isolation and identification of specific pathogens, with limited attention to pathogenicity, epidemiology, or transmission dynamics (Akoll & Mwanja, 2012). This diagnostic gap highlights the need for ecological and epidemiological studies, as well as continuous farm-level monitoring and national surveillance (Akoll & Mwanja, 2012).
As a World Organisation for Animal Health (WOAH) member, Uganda participates in international cooperation on aquatic animal disease prevention. The WOAH World Animal Health Information Database (WAHIS) has reported no major aquatic disease outbreaks in Uganda since 2010 (WOAH, 2024). Nevertheless, risks of introduction and spread remain for several notifiable and highly infectious diseases:
-
Tilapia lake virus (TiLV): detected in Uganda but not yet responsible for large-scale clinical outbreaks (Mugimba et al., 2018).
-
Epizootic ulcerative syndrome (EUS): a fungal disease caused by Aphanomyces invadans, considered a regional concern.
-
Koi herpesvirus disease (KHV): a potential threat to carp if introduced into Ugandan waters (MAAIF, 2020).
Within Uganda’s aquaculture systems, several diseases have already been recognized, often associated with poor water management, high stocking densities, and movement of unscreened broodstock (Isyagi et al., 2009; Mwanja et al., 2007; van Duijn et al., 2018):
-
Columnaris infection in catfish, which is frequently linked to handling injuries or overcrowding, is commonly managed using salt baths (Akoll & Mwanja, 2012).
-
Vitamin A deficiency in tilapia, due to low-quality feed, causes corneal opacity and blindness (MAAIF, 2017).
-
White spot disease caused by Ichthyophthirius multifiliis occurs frequently in pond systems, requiring proactive biosecurity and water-quality management (van Duijn et al., 2018).
Parasitic infestations, particularly in Nile tilapia (O. niloticus) and African catfish (C. gariepinus), remain widespread and are aggravated by unregulated fish movements between hatcheries and grow-out sites (Akoll et al., 2012; Mwanja et al., 2007).
Overall, structured farm-level health strategies—spanning biosecurity, surveillance, and regulated broodstock movement, are largely absent. Addressing these vulnerabilities is essential for sustainability. International experiences provide useful guidance: Egypt’s mandatory hatchery disinfection and inspection protocols (da Costa et al., 2019), and Nigeria’s adoption of vaccination and probiotics at farm level (Oladimeji et al., 2020), illustrate best practices Uganda could adapt. Strengthening on-farm monitoring, farmer training, and a formal disease-reporting network would enhance early detection capacity and improve response coordination during disease outbreaks.
Effective biosecurity is fundamental to preventing and controlling infectious diseases, and underpins sustainable aquaculture development (Assefa & Abunna, 2018; Global Aquaculture Alliance, 2006; Yanong & Erlacher-Reid, 2012). Yet the implementation of measures such as health certification, quarantine, disinfection, and movement control remains suboptimal at both institutional and farm levels (Bera & Dhar, 2018). Although Uganda has developed national standards through the Fisheries and Aquaculture Act (2022) and the Fish (Aquaculture) Rules (2023), enforcement is inconsistent: more than 70% of production units reportedly remain unregistered or unlicensed, and many farms lack even basic screening or quarantine (Matthew et al., 2015; NARO-KARDC, 2020).
Diagnostic capacity remains limited to a small number of laboratories with inadequate geographic coverage (MAAIF, 2017). As a result, underreporting and late detection of outbreaks remain serious risks. At the national level, institutional coordination is weakened by regulatory grey areas most notably the exclusion of aquatic species under the Animal Disease Act (1964)—which constrains rapid response and compliance with international reporting obligations (Akoll & Mwanja, 2012).
Against this backdrop, a wide range of pathogens have been documented in Ugandan aquaculture species, including protozoans, monogeneans, cestodes, and bacterial agents (Table 1). These infections are particularly prevalent in Nile tilapia and African catfish, with implications for biosecurity planning, improved diagnostic capacity, and potential vaccine development.
| Type | Species | Host | References |
|---|---|---|---|
| Parasite | |||
| Protozoa | Apiosoma sp. | Clarias gariepinus | Florio et al. (2009); NARO (2017) |
| Epistylis sp. | Clarias gariepinus | Akoll et al. (2012); NARO (2017) | |
| Ichthyobodo sp. | Oreochromis niloticus | Akoll et al. (2012); Florio et al. (2009); Walakira et al. (2014) | |
| Ichthyophthirius multifiliis | Barbus paludinosus, Lebistes reticulatus, Oreochromis niloticus | NARO (2017); Walakira et al. (2014) | |
| Trichodina spp. | Clarias gariepinus, Oreochromis niloticus | Akoll et al. (2012); Florio et al. (2009); Walakira et al. (2014) | |
| Monogenea | Cleidodiscus sp. | Oreochromis niloticus | Walakira et al. (2014) |
| Cichlidogyrus sclerosus | Oreochromis niloticus, Clarias gariepinus | Akoll et al. (2012); Florio et al. (2009) | |
| Macrogyrodactylus congolensis | Clarias gariepinus | Akoll et al. (2012) | |
| Neodiplozoon polycotyleus | Barbus neumayeri | Chapman et al. (2000) | |
| Clinostomum cutaneum | Oreochromis niloticus | Akoll et al. (2012); Gustinelli et al. (2010); Walakira et al. (2014) | |
| Ornithodiplostomum sp. | Clarias gariepinus | Akoll et al. (2012) | |
| Zoogonoides sp. | Clarias gariepinus | Akoll et al. (2012) | |
| Cestoda | Amirthalingamia macracantha | Clarias gariepinus, Oreochromis niloticus | Akoll et al. (2012); Florio et al. (2009) |
| Amphilinidae | Clarias gariepinus | Akoll et al. (2012) | |
| Monobothrioides sp. | Clarias gariepinus | Akoll et al. (2012) | |
| Camallanidae sp. | Clarias gariepinus, Oreochromis niloticus | Akoll et al. (2012) | |
| Procamallanus sp. | Clarias gariepinus, Oreochromis niloticus | Akoll et al. (2012) | |
| Crustacea | Lamprolena sp. | Oreochromis niloticus | Akoll et al. (2012) |
| Acanthocephala | Acanthogyrus tilapiae | Oreochromis niloticus | Akoll et al. (2012); Florio et al. (2009) |
| Bacteria | |||
| Flavobacterium columnare | Clarias gariepinus, Oreochromis niloticus | Walakira et al. (2014) | |
| Aeromonas hydrophila | Clarias gariepinus, Oreochromis niloticus | Walakira et al. (2014) | |
| Aeromonas sobria | Clarias gariepinus, Oreochromis niloticus | Charles et al. (2016); Walakira et al. (2014) | |
| Edwardsiella tarda | Clarias gariepinus, Oreochromis niloticus | Wamala et al. (2018) | |
| Plesiomonas shigelloides | Clarias gariepinus, Oreochromis niloticus | Wamala et al. (2018) | |
| Proteus vulgaris | Clarias gariepinus, Oreochromis niloticus | Charles et al. (2016) |
In sum, Uganda’s aquaculture biosecurity is hampered by fragmented regulation, diagnostic and enforcement gaps, and incomplete adoption of best practices at farm level. At the national level, institutions such as the National Agricultural Research Organisation (NARO), the Directorate of Animal Resources under MAAIF, and National Fisheries Resources Research Institute (NaFIRRI) provide limited support for fish disease diagnostics and research. However, no dedicated aquatic animal health authority exists, highlighting the urgent need for a centralized national or regional body to coordinate surveillance, response, and farmer support.
Uganda’s legal framework for aquatic animal health includes constitutional, statutory, and subsidiary instruments. The Constitution of Uganda (1995) provides for the stewardship of natural resources (GOU, 1995), operationalized through the Fisheries and Aquaculture Act (2022). This Act prescribes minimum operational standards and regulates the movement of live animal movement, and imposes penalties for non-compliance. The Fish (Aquaculture) Rules (2023) set out requirements for seed certification, disease reporting, and farm inspections. Additional measures, such as the Fish Quality Assurance Rules (2017), further support food safety and conservation objectives.
Despite progress, enforcement remains uneven and key gaps persist. Notably, the Animal Disease Act (1964) excludes aquatic species, creating jurisdictional ambiguity that limits coordinated biosecurity oversight (Akoll & Mwanja, 2012). Institutions such as MAAIF’s Aquaculture Division, Makerere University, and NaFIRRI provide vital research and diagnostic support, but lack clear mandates, budgets, and extension capacity.
To overcome these constraints, harmonization of legislation is urgently required, specifically through amendment of the Animal Disease Act to include aquatic animals alongside stronger mandates and resources for regulatory and diagnostic agencies (FAO & WHO, 2021; MAAIF, 2017).
Field reports indicate that the introduction of mandatory licensing and biosecurity inspections under the Fisheries and Aquaculture Act (2022) (GOU, 2022) has already been associated with measurable benefits. Certified farms have reportedly experienced fewer disease outbreaks, improved buyer confidence, and greater market stability (MAAIF, 2023; RVO, 2022). Preliminary evidence also suggests that enhanced regulatory oversight may contributing to higher farm-gate prices and more reliable access to domestic and regional markets (MAAIF, 2023). These observations highlight the practical value of regulatory reform in strengthening fish health management and sector resilience.
Discussion
The findings of this review indicate that Uganda’s aquaculture sector faces several structural constraints in fish health management, particularly limited diagnostic capacity, weak disease surveillance, and inconsistent implementation of biosecurity regulations. These challenges increase the risk of undetected disease emergence and limit the effectiveness of current management practices. Addressing these constraints requires a pragmatic, multilayered biosecurity strategy that integrates farm-level management improvements with stronger institutional coordination.
Based on the gaps identified in Uganda’s aquaculture systems, several priority measures can be recommended to strengthen biosecurity implementation at both farm and institutional levels. Stock sourcing and quarantine are critical components of aquaculture biosecurity. Eggs, fry, and broodstock should be obtained, whenever possible, from certified suppliers, and quarantine procedures together with health checks should be implemented for all newly introduced stocks to reduce the risk of pathogen introduction (Matthew et al., 2015). Farm management improvements are also essential. Reducing environmental and handling stress, maintaining pond and cage infrastructure, optimizing stocking densities, and providing nutritionally balanced feeds can significantly enhance fish immunity and reduce disease susceptibility. Effective sanitation and disinfection practices are also essential for disease prevention. Routine cleaning and the application of approved disinfectants to equipment, personnel, and operational areas can significantly reduce the likelihood of pathogen transmission when routinely cleaned and disinfected (Global Aquaculture Alliance, 2006). Personnel and visitor management also play an important role in maintaining farm biosecurity. Training farm staff in basic biosecurity procedures, restricting access to sensitive production areas, and assigning defined operational roles can help minimize cross contamination among fish species, age groups, and production units. Proper record-keeping and compliance with management protocols further support effective biosecurity. Maintaining detailed records on stock sourcing, health monitoring, and sanitation practices, together with safe storage and management of feeds and chemicals, can improve traceability and disease response capacity. Given the large number of small-scale farmers in Uganda, affordable biosecurity approaches are particularly important. Low-cost measures such as simple footbaths, community-based fry health checks, and basic record-keeping systems can improve farm biosecurity when supported by extension services and peer-to-peer training initiatives (Isyagi & Fejzic, 2021).
However, farm-level biosecurity measures alone are insufficient without adequate institutional support. Strengthening regional diagnostic services, establishing real-time disease surveillance systems, and expanding extension services are essential to ensure early detection and rapid response to emerging disease threats. Ultimately, the development of a coherent and cost-sensitive national biosecurity framework—integrating regulatory oversight, farmer training, accessible diagnostic services, and coordinated disease reporting—will be critical to safeguarding sector productivity, ensuring effective disease prevention, and protecting rural livelihoods (Isyagi & Fejzic, 2021; Bera & Dhar, 2018; Global Aquaculture Alliance, 2006). To achieve sustainable growth and sector resilience, the government has established clear targets for 2025–2030 as outlined in Table 2. Experiences from other aquaculture-producing countries provide useful reference points. For instance, Egypt’s centralized broodstock certification program and Nigeria’s integration of aquatic animals into national animal health legislation have improved early disease detection and strengthened farm-level compliance (Akoll & Mwanja, 2012; El-Gamal et al., 2019; Oladimeji et al., 2020). These approaches may help inform biosecurity reforms in Uganda.
| Policy area | Target by 2030 | References |
|---|---|---|
| Annual aquaculture production | 1,000,000 metric tonnes | FAO (2022a); MAAIF (2021) |
| Registered hatcheries and farms | 100% compliance (all units licensed/registered) | MAAIF (2021) |
| Prevalence of major notifiable diseases | < 2% national prevalence of World Organisation for Animal Health (WOAH)-listed diseases | MAAIF (2021); WOAH (2024) |
| Fish certified disease-free | 95% of export and major domestic supply | FAO (2022b) |
| Export-eligible farms with health certificate | 90% or more of commercial farms | MAAIF (2021) |
| National fish consumption | 15 kg per capita | FAO (2022a) |
| Aquaculture share of total fish supply | ≥ 30% of national fish consumption | FAO (2022a); MAAIF (2021) |
Note: Targets reflect national and international strategies from FAO (2022a, 2022b), MAAIF (2021), and WOAH (2024). Target values may be periodically revised in official policy documents.
Conclusion
Uganda’s aquaculture sector has expanded considerably in recent years but remains vulnerable to infectious disease threats due to critical gaps in diagnostics, surveillance, and regulatory implementation. Most farms operate without standardized biosecurity protocols, and diagnostic services remain limited to a few institutions with inadequate geographic coverage. The absence of a national surveillance system further weakens early detection and coordinated response mechanisms.
At the farm level, limited farmer training and the low adoption of biosecurity measures—exacerbated by weak regulatory enforcement and insufficient extension services—continue to undermine disease prevention. In addition, the legal framework governing aquatic animal health remains fragmented, as aquatic species are still excluded from core animal health legislation, creating institutional ambiguity and regulatory gaps. A coordinated, multilevel approach that integrates stronger institutional capacity, expanded diagnostic services, farmer training, and regulatory reform will therefore be essential to safeguard the long-term sustainability of Uganda’s aquaculture industry.








