Introduction
The demand for nutritious and safe functional foods continues to expand as more individuals recognize the health-promoting benefits of probiotics. Probiotics were first appreciated for their benefit to maintaining the gut microbiome balance, but now associated with many potential health benefits and are becoming a preferred choice for many who are seeking to elevate their well-being (Dhopatkar et al., 2023). Functional foods enriched with probiotics production is rising quickly in the global market and particularly in the Asia-Pacific region, where a “fourth meal” concept around regular consumption of vitamins and supplements has developed and increased the amount of these functional foods consumed (IPA, 2023).
Fermented food plays a vital role in the human diet since ancient times. Fermentation process not only preserve food by preventing spoilage but also improve microbiological safety, nutritional quality, and sensory attributes. These benefits have led to the development of various fermented products using both animal and plant-based raw materials. In recent years, growing scientific evidence supports the health-promoting effects of fermented foods contributing to their increasing global consumption (Şanlier et al., 2019). During fermentation, various microbes contribute to this process by producing enzymes, which can generate bioactive peptides recognized for their potential health-supporting properties. This interplay between microbes and compounds not only enriches the sensory qualities of food but also promotes overall wellness. Fermentation may occur spontaneously driven by naturally occurring microbes or be initiated with starter cultures (Peralta et al., 2008).
Fermented fish has been common part of Southeast Asian diets since 15th century, often eaten as a side plate or used as a flavoring in daily meals (Hajeb & Jinap, 2012). In particular, Indonesia has a rich tradition of such foods including fermented fish or shrimp (Terasi), fermented mackerel fish (Ikan Peda), and fermented fish (Rusip). Fermented foods can also serve as effective carriers of probiotics, thereby creating functionality in the food matrix as well as divesting probiotic microorganisms their associated health benefit. Among probiotic candidates, spore-forming Bacillus have gained attention due to their resilience under harsh conditions, including heat, desiccation, acidic pH, and bile salts (Colom et al., 2021). These properties enhance their viability in food matrices and gastrointestinal environments.
Bacillus species are not only resilient in harsh environments but also provide numerous health benefits. Bacillus spp. help combat harmful microbes by outcompeting them, which supports a healthy immune system. Additionally, these bacteria can enhance nutrient absorption and may assist with digestive issue, including diarrhea, irritable bowel syndrome, and ulcerative colitis. This positive effects extend to skin health, as Bacillus species can help prevent bacterial infection including vaginosis. Due to these valuable properties, Bacillus species are becoming increasingly popular in the production of food and supplements, catering to both technical requirements and consumer health interests (Adibpour et al., 2019).
Bacillus coagulans stands out as a remarkable species within the Bacillus genus due to its unique physiological and biochemical traits. This bacterium is highly efficient at producing lactic acid by fermenting various sugars, including glucose, maltose, and mannitol. Notably, B. coagulans is one of the most researched heat-tolerant strains of Bacillus. It is a Gram-positive, spore-forming bacterium distinguished by its ability to thrive in rigorous conditions, such as high temperatures, freezing environment, and dryness. Importantly, B. coagulans is non-pathogenic organism and is Generally Recognized as Safe (GRAS) issuance by the U.S. Food and Drug Administration (FDA), making it safe choice for use in food products and dietary supplementary (Su & Xu, 2014; USFDA, 2018).
Probiotics are becoming increasingly for their role as natural preservatives. These beneficial microorganisms produce various substances including diacetyl, hydrogen peroxide, bacteriocins, and organic acids. Recent studies emphasizes the significant role probiotics play in managing both pathogenic organisms and those that cause food spoilage. This emerging trends suggests that incorporating probiotics into food preservation could lead to safer, more sustainable options for longevity food (Konuray & Erginkaya, 2018). In particular, several strains of B. coagulans have demonstrated inhibitory effects against harmful bacteria, and in vitro studies have also shown its antioxidant properties (Abdhul et al., 2015). Given their potential health benefits, the characterization and isolation of novel probiotic strains remain a critical area of research. It is important to understand the functionality and potential use in food industry, as even strains from the same species exhibit great variability in their physiological, functional, and sensory aspects (Meleh et al., 2020).
Thus, the objective of this study is to isolate a probiotic strain among Bacillus spp. and characterize its probiotic properties from traditional Indonesia fermented foods, including fermented fish or shrimp (Terasi), fermented mackerel fish (Ikan Peda), and fermented fish (Rusip). The isolated strain will be evaluated for its probiotic potential based on acid and bile salt tolerance, antibiotic susceptibility, antimicrobial activity, and in vitro antioxidant properties.
Materials and Methods
Traditional Indonesian fermented fish products, such as fermented anchovy fish (Rusip), fermented shrimp paste (Terasi Udang), fermented fish paste (Terasi Ikan), and fermented mackerel fish (Ikan Peda) was acquired from multiple coastal regions of Indonesia in July 2024. The samples were obtained from local markets and small-scale traditional producers in major production areas across Java, Sumatera, and Bangka Belitung. All products were acquired in their commercially available and traditionally fermented forms.
Approximately, 25 g of each traditional Indonesian fish fermented products were incorporated with 225 mL of phosphate-buffered saline (pH 7.0) to obtain a 1:10 dilution and subjected to homogenization by stomaching (Interscience, St. Nom, France). One milliliter of the homogenized sample was inoculated into DeMan, Rogosa and Sharpe (Becton Dickinson, Franklin Lakes, NJ, USA) broth and incubated at 37°C for 24 h. Afterward, the broth culture was serially diluted, 100 μL of each dilution was spread onto the surface of triplicate agar plates containing Bromocresol purple as an indicator and incubated for 24 h at 37°C. Selected well-isolated colonies were subcultured into fresh De Man, Rogosa and Sharpe broth to create stock cultures. To ensure their viability for the long term, these cultures were stored at –20°C in MRS broth that enriched 500 μL liquid culture with 50% glycerol (1:1, v/v; Baick & Kim, 2015 with slight modification).
Genomic DNA was extracted with an AccuPrep Genomic DNA Extraction Kit (Bioneer, Daejeon, Korea). The 16S rRNA gene was amplified using universal bacterial primers, with the forward targeting the conserved 5’ region and the reverse primer annealing to the conserved 3’ region of gene. Polymerase chain reaction (PCR) amplification was performed using primer 27F (5’-AGAGTTTGATCCTGGCTCAG-3’) and 1492R (5’-GGTTACCTGTTACGACTT-3’). The total (25 µL) consisted of 1 µl each of 27F and 1492R, 12.5 µL Taq PCR mix (Promega, Madison, WI, USA), and 9.5 µL ddH2O. The thermal-cycling condition: An initial denaturation at 94°C for 1 minute, followed by 30 cycles of denaturation at 94°C for 1 minute, annealing at 52°C for 1 minute, and extension at 72°C for 1 minute. Following the last cycle, a final extension was conducted 72°C for a final extension of 10 minutes. We separated the PCR products by electrophoresis on a 1% (w/v) agarose gel, and assessed the nucleotide sequences with the NCBI BLAST to address the sequence similarity (Ramesh et al., 2015). Selected sequences of representative strains were aligned using ClustalW of the molecular evolutionary genetic analysis (MEGA) 11 software package, and Neighbor-joining method with bootstrap values based on 500 replications was used to constructed a phylogenetic tree of representative strains.
Culture of each isolate grown overnight culture were inoculated at the concentration of 106 CFU/mL into MRS broth culture medium (Becton Dickinson) at pH 2, pH 3, pH 4, pH 5, and pH 6 then incubated at 37°C for 4 h. The viable cell counts were determined using the pour-plate method, and plates with agar were incubated for 48 h at 37°C (Zoumpopoulou et al., 2008 with modifications). The number of colony-forming unit (CFU/mL) was recorded, and percentage survival was calculated using the following Equation:
Bile salts (0.1%, 0.2%, and 0.3%) were incorporated into the MRS broth culture medium (Becton Dickinson). The isolates were inoculated into these broths with varying bile salt concentrations and incubated for 24 h at 37°C. Following incubation, serial dilutions were performed and the viable cell numbers were determined by plating the samples on MRS agar (Shehata et al., 2016 with modifications). The cell survival rate were calculated using formula:
The antibiotic susceptibility of two strains (P5 and F2) against antibiotics, including ampicillin, chloramphenicol, streptomycin, erythromycin, and tetracycline were assessed, each antibiotic was prepared in the solvent through a series of two dilutions. The cell-free supernatant of B. coagulans isolate strain were tested at concentration 512 to 0.25 μg/mL using serial dilutions. Two-fold serial dilutions were performed in Mueller-Hinton Broth. Aliquots of the cultures were dispensed into 96-well microplates in triplicate. Followed by 24 h of incubation at 37°C, antibiotic resistance was calculated by the minimum inhibitory concentration (MIC) as outlined to European Food Safety Authority (EFSA) guidelines (FEEDAP et al., 2018).
Bacterial suspensions of Escherichia coli KCTC 1682 (EC), Staphylococcus aureus KCTC 1916 (SA), Listeria monocytogenes KCTC 3569 (LM), Streptococcus mutans KCCM 40105 (SM), methicillin-resistant Staphylococcus aureus (MRSA) KCCM 40510 were prepared. The EC (1682), SA (1916) and LM (3569) from KCTC, and SM (40105) and MRSA (40510) from Korean Culture Centre of Microorganisms (KCCM). Each microorganism was cultured for 12 h and subsequently diluted 1:100 in the sterile tryptic soy broth (Becton Dickinson; Khan et al., 2022; Tabassum et al., 2025). The F2 and P5 cell-free supernatant (CFS) were tested at concentrations ranging from 4,096 to 2 µg/mL using serial dilutions. Aliquots of the treated cultures were dispensed into 96-well polystyrene microplates (300 µL per well) in triplicate and incubated at 37°C for 24 h. MIC values were determined by visual inspection, defined as the lowest concentration resulting in ≥ 90% growth inhibition compared with the control. Bacterial growth was further quantified by measuring optical density at 600 nm using a microplate reader (BioTek, Winooski, VT, USA).
Free radical scavenging activity of the strains was evaluated using the 2.2-diphenyl-1-picrylhydrazyl (DPPH) assay. It is widely used methods known for its simplicity and rapid preparation (Gulcin, 2020). In brief, an aqueous solution of 0.2 mM DPPH was made freshly, and 0.8 mL of cell-free supernatant (107 CFU/mL) was mixed with 1 mL of DPPH solution. After incubating the mixture in the dark for 30 minutes, the absorbance at 517 nm was measured with a microplate reader (BioTek). L-ascorbic acid on DPPH was used as a positive control. Free radical scavenging activity were calculated as follow:
Sample: absorbance of the mixture of sample and DPPH; Blank: absorbance of blank.
The ABTS radical cation was obtained by combining a 7.4 mM ABTS solution with potassium persulfate (2.6 mM) and incubating overnight in the dark at room temperature. Before the assay, the ABTS solution was dissolved to reach an absorbance of 0.70 ± 0.03 at 734 nm. The assay consisted of adding a 20 µL cell-free supernatant to 180 µL of the diluted ABTS radical solution in a 96-well microplate. The mixture was kept under dark conditions for 10 min (Re et al., 1999). L-ascorbic acid was used as a positive control. The ABTS radical scavenging activity (%) were calculated, as follows:
A is the control absorbance, and B is the sample absorbance.
The FRAP reagent was prepared by mixing 0.3 M sodium acetate buffer (pH 3.6), 10 mM 2,4,6-tri(2-pyridyl)-1,3,5-triazine (TPTZ) in 40 mM HCl, and 0.02 M ferric chloride (FeCl3.H2O) in a ratio of 10:1:1 (v:v:v). The mixture was incubated for 15 min at 37°C. For the assay, the FRAP reagent (150 µL) was added to 50 µL cell-free supernatant in a 96-well microplate and kept in dark for 20 min. Absorbance was measured at 593 nm (Benzie & Strain, 1996).
Results and Discussions
Probiotics, often found in fermented foods, are recognized for their health benefits. It can help alleviate symptoms of lactose intolerance, manage high cholesterol levels, and play a role in preventing and treating cardiovascular issues like atherosclerosis (Rashed et al., 2022). To identify novel, robust probiotic strains, we investigated traditional Indonesian fermented fish products, including fermented fish paste (Terasi Ikan), fermented shrimp paste (Terasi Udang), fermented anchovy fish (Rusip), and fermented mackerel fish (Ikan Peda).
In this study, twelve bacterial strains were identified from traditional Indonesian fermented fish products, including 3 strains from the Bacillus genera (B. coagulans and Bacillus sp.), 1 strain from the Heyndrickxia genera (Heyndrickxia coagulans), 2 strains of Enterococcus (Enterococcus durans and Enterococcus thailandicus), and 4 strains of Weissella (Table 1). The molecular relationship between F2 and P5 isolates strain and other closely similar species was illustrated by a phylogenetic tree based on 16S rRNA genes (Fig. 1). Isolate F2 was confirmed to be H. coagulans with highest similarity of 99.93% and P5 was closest to B. coagulans at 100% similarity. Isolate F2 and P5 may therefore be considered as H. coagulans and B. coagulans. Isolate F2 and P5 was clustered in the same group as B. coagulans, H. coagulans, and Heyndrickxia acidiproducens. B. coagulans was reassigned into the Weizmannia genus owing to its clear phylogenetic separation. B. coagulans form a distinct monophyletic lineage, separate from the Bacillus subtilis and Bacillus amyloliquefaciens groups, which are traditionally classified under the genus Bacillus. Furthermore, the study identified specific conserved signature deletions that are characteristic of the clade encompassing B. coagulans. These molecular markers set this group apart from other Bacillus species, reinforcing the justification for a distinct genus. Nonetheless, recent progress in phylogenomic investigations has led to a subsequent reclassification involving Weizmannia coagulans and the Heyndrickxia genus. This taxonomic revisions stems from more comprehensive genomic evidence, which demonstrated that B. coagulans exhibits a closer evolutionary affinity with members of the Heyndrickxia genus. The refined analysis indicated that the unique lineage containing B. coagulans corresponds more closely with Heyndrickxia, as supported by updated genetic signatures and reconstructed phylogenetic trees (Gupta et al., 2020; Rao et al., 2023). Among them, two strains of B. coagulans P5 (Acc. Number PV774563) and H. coagulans F2 (Acc. Number PV762941) were selected for further study because of their spore-forming ability. B. coagulans exhibits exceptional resistance and survivability under adverse conditions, including low-oxygen environments, gastrointestinal transit (e.g., exposure to bile salts and gastric acid), and various food processing stresses. Additionally, this strain secretes extracellular enzymes, such as amylase and protease, which enhance fermentation and improve the nutritional quality of food products (Konuray & Erginkaya, 2018). B. coagulans is also recognized as an efficient microbial cell factory for eco-friendly biosynthesis process, such as lactic acid, that offer the sustainable alternative to chemical synthesis. Since it is a moderately thermophilic organism, B. coagulans has a minimal risk of microbial contamination made it suitable for open fermentation processes.
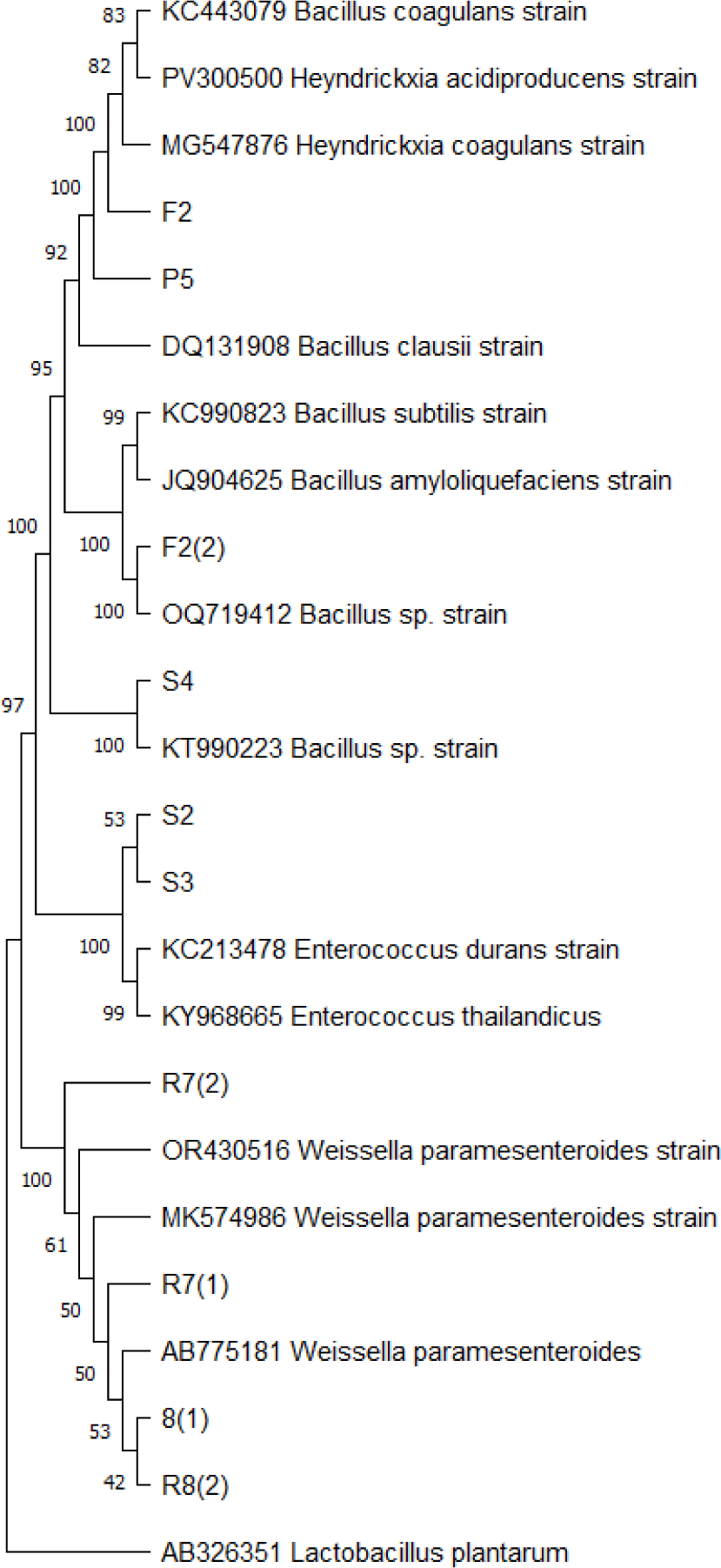
Consistent with previous findings, Bacillus species were predominant in traditional Indonesian fermented shrimp paste and are known for their roles in both fermentation and food safety. Specifically, B. coagulans, produces the bacteriocin “coagulin”, which exhibits antimicrobial properties. This bacterium is Gram-positive, aerobic or facultatively aerobic, and rod-shaped, also be able to form spores at 37°C (Altun & Erginkaya, 2021). In this study, strains P5 and F2 were isolated from fermented shrimp paste (Terasi Udang) and fermented fish paste (Terasi Ikan), respectively. These isolates exhibited a round shape and purple coloration on Gram staining. Their cell walls are rich in peptidoglycan composed of N-acetylglucosamine and N-acetylmuramate, along with amino acids including D-glutamic acid, L-alanine, D-alanine, and diaminopimelic acid. The presence of teichoic acids (TA) linked by phosphodiester bonds, contributes to cell wall integrity and resistance in acidic environment (Reuben et al., 2020).
B. coagulans presence in Terasi may originate from raw shrimp ingredients or environmental exposure, during manual handling and fermentation. These microbes are integral to the biochemical reactions that lead to the odor and flavor characteristic of fermented food. A previous study reported that Terasi starter cultures were composed of B. coagulans, Bacillus megaterium, Micrococcus kristinae, Bacillus pumilus, and Bacillus brevis (Surono & Hosono, 1994). According to Indonesian Pharmaceutical and Food Control Authority Regulation Number 38/2023, only 16 microorganisms are permitted for use in processed foods. These include: B. coagulans, Bifidobacterium breve, Bifidobacterium animalis, Bifidobacterium lactis, Bifidobacterium longum, Lactobacillus casei, Lactobacillus acidophilus, Lactobacillus fermentum, Lactobacillus paracasei, Lactobacillus lactis, Lactobacillus plantarum, Lactobacillus rhamnosus, Lactobacillus citreum, Lactobacillus delbrueckii subsp. bulgaricus and Streptococcus salivarius subsp. thermophilus (BPOM, 2023). B. coagulans is also listed in the Australian Government (2024) starter cultures database for use in human food or beverages, human cosmetic applications, in vitro laboratory work, in vivo work in laboratory organisms, and human therapeutic use. It is also included in the raw materials list permitted for food in accordance with the Food and Food Additives Codex (MFDS, 2019).
In the FAO/WHO, it was concluded that both in vitro tests are useful to evaluate probiotic properties, especially their ability to withstand acidic environments. Acid tolerance is important for defining probiotics ability to withstand gastric stress, viability and activity over time in acidic foods. The ability to survive through the gastrointestinal tract is one of important factors in establishing probiotic efficacy. The stomach’s harsh environment, with a pH typically ranging from 1.5 to 4.5, significantly challenges microbial survival, especially with prolonged exposure. In this study, isolate P5 had a survival rates ranging from 67.93%–97.81% and F2 survival rates ranging from 69.55%–98.32% across pH 2.0 to pH 6.0 (Fig. 2). Both isolates retained viability at a pH 3.0 after 4 hours of incubation, demonstrating significant resistance to acid. The rates of survival reported in this study were statistically meaningful (p < 0.05), and indicate the role of pH in affecting probiotic viability. For probiotic strains, rates of survival of 50% or greater after exposure to acid is commonly regarded as indicator of acid tolerance. When microbial cells are exposed to highly acidic conditions (e.g., pH 3), proton influx exceeds their capacity to maintain homeostasis. This excessive proton entry lowers intracellular pH, disrupts regulatory mechanisms, and leads to impaired physiological functions, protein denaturation, as well as DNA and membrane damage (Foster, 2004). The mechanism via which inorganic and organic acids affect microbial cells is different, but both kinds of stress result in intracellular acidification and damage to biochemical processes (Chung et al., 2006). These findings suggest that both isolates P5 and F2 can survive in the stomach conditions, supporting their use as probiotics (Casarotti & Penna, 2015).
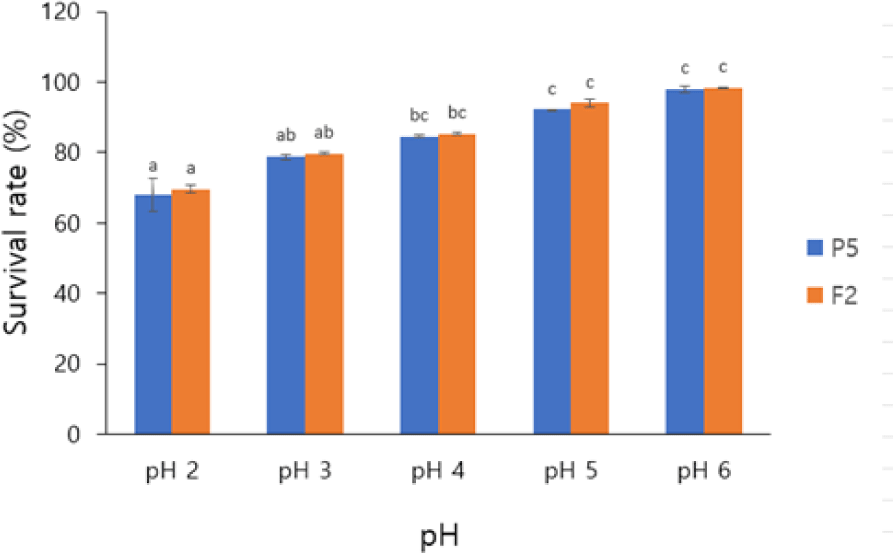
The functional relevance of acid tolerance is further underscored by studies linking probiotic-derived acid production to pathogen inhibition. For instance, Chaudhari et al. (2022) indicates that B. coagulans SKB LAB-19 inhibits pathogenic bacteria like E. coli and Clostridium perfringens by producing lactic acid. This dual role, surviving gastric transit and exerting antimicrobial effects, highlights the therapeutic potential of acid-resistant probiotics in mitigating enteric infections and modulating gut microbiota.
Tolerance of bile salt is a key trait of probiotic strains to survive and perform functionality in the small intestine. Bile salts are critical for the establishment of gut microbiome and for supporting their metabolic function in a host’s gut. Due to their surfactant properties bile salts can disrupt bacterial cell membranes, leading to cell lysis. Therefore probiotic bacteria must be able to survive under these harsh conditions to maintain their functionality (Monte et al., 2009).
Both isolates showed high survival, ranging from 74.60%–97.49%. However, a significant loss of viability was noted at the highest bile salt concentration (0.3%). The survival rates at 0.03% bile salts were 74.60% for strain P5, and 83.14% for strain P2 (Fig. 3). The statistical analysis indicates a significant loss of survival at 0.3% bile salt (p < 0.05) and had no significant differences at 0.1% and 0.2% These results indicate that 0.3% bile salt concentration induces considerable physiological stress which could affect the viability of probiotic isolates. The bile salt tolerance observed in our B. coagulans isolates compares favorably with other probiotic strains reported in the literature. For instance, Lactobacillus reuteri L-3 and Pediococcus pentosaceus 2–5 showed good tolerance at 0.1% bile salt (81.08% and 79.76% survival rates, respectively) but demonstrated dramatically reduced survival rates of less that 1% at 0.3% bile salt concentration after 6 h of incubation (Wang et al., 2022a). These comparison highlight that our B. coagulans isolates with the survival rates above 74% at 0.3% bile salt demonstrate relatively robust bile tolerance compared to many traditional lactic acid bacteria probiotics.
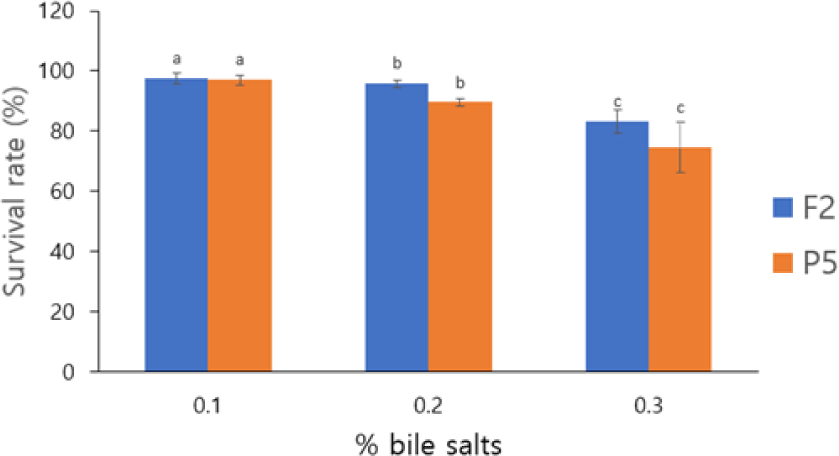
Probiotic bacteria employ various adaptive strategies to survive bile salt stress, such as the expression of specific proteins, modification of cell membrane composition, and production of bile salt hydrolase (BSH) to detoxify bile salts (Bustos et al., 2018). Collectively, these mechanisms contribute to the ability of B. coagulans P5 and F2 to maintain high viability in the bile salts presence, supporting their potential as effective probiotic candidates.
The important aspect of antibiotic resistance within the food industry is that it results in multidrug-resistant pathogens, and additional increase in foodborne infections. Bacterial species such as probiotic bacteria naturally acquire antibiotic resistances that help them survive. The EFSA gives recommendations for antibiotic susceptibility testing. This form defines the MIC as the lowest effective dose of an antibiotic that prevent detectable bacterial growth. This study determined antibiotic susceptibility for CFS of B. coagulans P5 and F2 isolates by using antibiotics specified by EFSA recommendations for the genus Bacillus.
The MIC of each antibiotic (ampicillin, chloramphenicol, streptomycin, erythromycin, and tetracycline), for each isolates, was found to range from 0.5 to 8 μg/mL (Table 2). As FEEDAP (2012) has not established specific antibiotic breakpoints for B. coagulans, the susceptibility of the tested strains was interpreted based on EFSA-defined breakpoints for Bacillus spp. Based on criteria, both F2 and P5 were classified as susceptible to all seven tested. Previous studies have reported varying antibiotic susceptibility profiles among probiotic Bacillus strains. Altun & Erginkaya (2021) reported that several antibiotics were effective against B. coagulans strains, including ciprofloxacin, chloramphenicol, erythromycin, ampicillin, kanamycin, clindamycin, streptomycin, rifampin, gentamycin, vancomycin, and tetracycline. These studies highlight the importance of conducting regular antibiotic susceptibility testing for probiotic strains.
| Antibiotics | MIC values (µg/mL) | Cut-off value | Susceptibility1) | |
|---|---|---|---|---|
| F2 | P5 | |||
| AMP | 4 | 8 | Nr | Nr |
| CHL | 4 | 4 | 8 | S |
| STM | 4 | 4 | 8 | S |
| ERY | 0.5 | 1 | 4 | S |
| TET | 4 | 1 | 8 | S |
Food contamination and foodborne illnesses are serious public health issues in developing countries, creating a significant responsibility for food safety systems. The antimicrobial activity of probiotic strains is one an important aspect of the bacterial candidate selection process for functional foods, as it reduces the propensity of pathogenic and spoilage bacteria to cause spoilage. The present study explored the antimicrobial activity of the two CFS of B. coagulans isolates (P5 and F2), providing MIC values (Table 3). The probiotic strains were shown an inhibitory effect on pathogens tested. Two isolates were exhibit to E. coli, L. monocytogenes, S. mutans, S. aureus, and methicillin-resistant S. aureus. The observed antimicrobial activity is consistent with previous reports demonstrating the broad-spectrum efficacy of B. coagulans strains. The discovery of antimicrobial activity seen among the two isolates (F2 and P5) may be due to the bacteriocins found in the cell-free extracts, as prior research showed bacteriocin-producing strains more potent against Gram-positive bacteria than Gram-negative pathogens (Gao et al., 2019). Moreover, these results and recent literature underscore the promise B. coagulans isolates as effective probiotic with significant antimicrobial properties suitable for use in health and food application.
| Isolates code | Antimicrobial activity (MIC as µg/mL) | ||||
|---|---|---|---|---|---|
| EC | SA | LM | SM | MRSA | |
| F2 | 512 | 512 | 512 | 2048 | 512 |
| P5 | 1024 | 1024 | 512 | 2048 | 512 |
Probiotic-mediated pathogen suppression typically occurs through the secretion of antimicrobial metabolites, including organic acids (lactic acid and acetic), hydrogen peroxide, and bacteriocins, often modulated by pH. Kralik et al. (2018) reported the antimicrobial potency of cell-free supernatants (CFS) from Lactobacillus spp. and Enterococcus spp. was reduced when pH exceeded 4.5. Furthermore, the composition of CFS strongly affects inhibitory efficacy, with bacteriocins or antimicrobial peptides (AMPs) serving as key agents. Pathogen inhibition by probiotic is also shaped by chemical, physical, and nutritional environmental factors (Hung et al., 2021; Zare Mirzaei & Lashani, 2018).
Antioxidants help preserve food and promote human health by reducing reactive oxygen species (ROS)-induced cellular damage and regulating oxidative stress. Antioxidants scavenge free radicals and inhibit cellular oxidative damage. The present study evaluated the antioxidant potential activity of two B. coagulans isolates using DPPH, ABTS, and FRAP assays (Fig. 4). The DPPH and ABTS assays, which measure radical scavenging activity, showed significant difference (p < 0.05) between isolates F2 and P5, suggesting strain-specific differences in bioactive metabolites including peptides, exopolysaccharides, and organic acids, which can donate electrons or hydrogen atoms to neutralize free radicals (Sui et al., 2020). Both isolates, however, showed lower scavenging activity than ascorbic acid as positive control, which is consistent with previous study that indicating that probiotic-derived antioxidants generally exert moderate effects compared with pure antioxidant compounds. Nevertheless, accumulating evidence indicates that certain probiotic bacteria strains possess antioxidant potential, supporting their application as natural antioxidant agents (Mishra et al., 2015; Mirzaei et al., 2021). Although lower than ascorbic acid, the antioxidant activity of B. coagulans remains biologically relevant, supporting its potential application in functional foods. The FRAP assay indicated that the two B. coagulans isolates exhibited strong reducing activity, with no statistical significant difference (p > 0.05), indicating that both isolates possess similar reducing power. This reflects comparable levels of reductive compounds, including short-chain fatty acids and organic acids, capable of donating electrons to reduce ferric ions (Mirzaei et al., 2021).
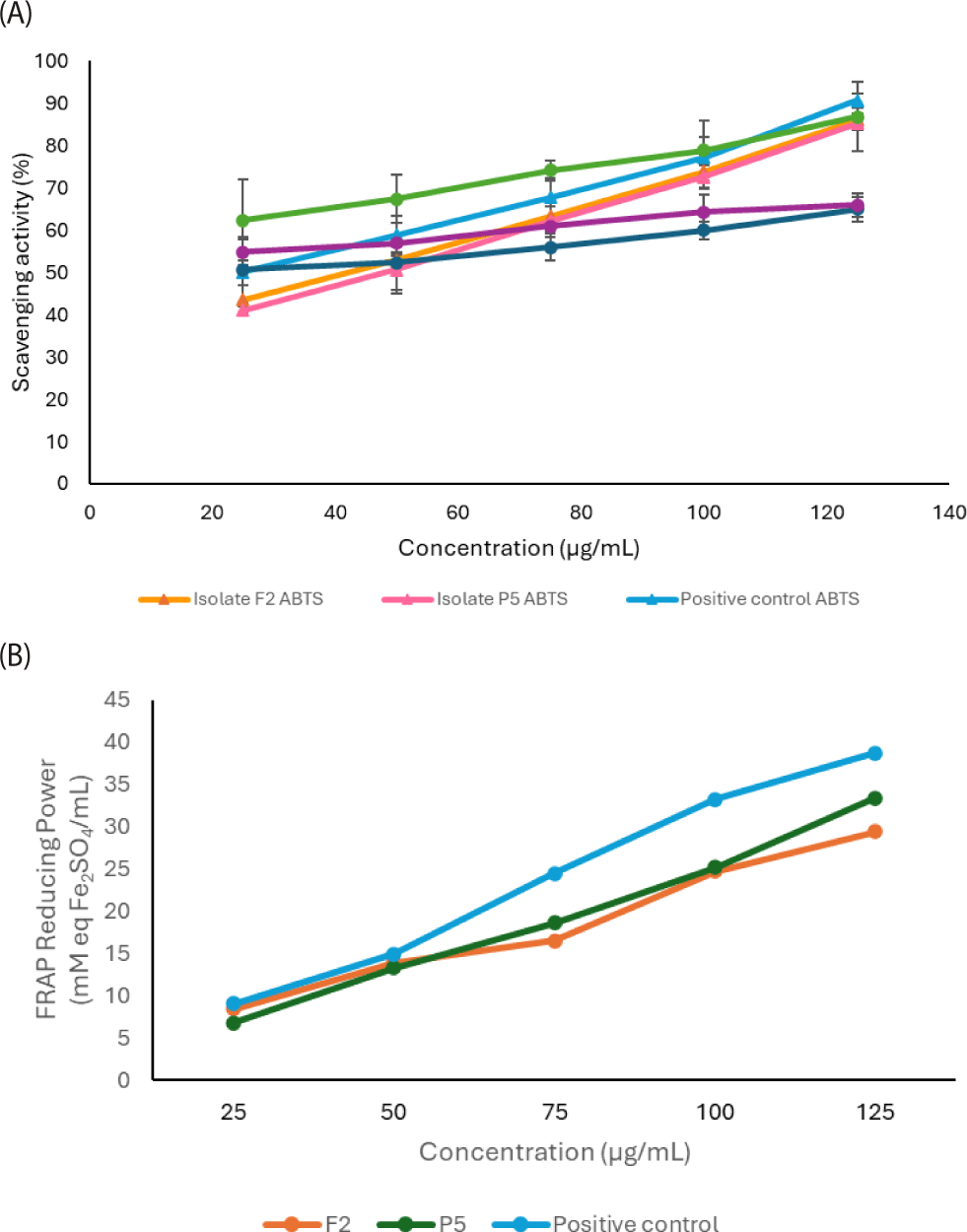
These results demonstrate that both B. coagulans isolates possess considerable in vitro antioxidant capacity, with P5 isolate showing a slight advantage. This level of activity is consistent with recent reports on B. coagulans strains. Sui et al. (2020), for instance, observed B. coagulans strains exhibiting high lactase-producing activity demonstrated notable antioxidant potential, with scavenging capacities of 35% for DPPH free radicals, 39% for hydroxyl radicals, and 14.8% for superoxide anion radicals. Panritdam et al. (2019) reported Lactobacillus fermentum FF (Thai fermented fish) and FB (Thai fermented sliced beef) showed the maximum DPPH scavenging activity were 65% and 61%, respectively. Fermentation can boost the antioxidant capacity of foods in several ways: by increasing phenolic compounds, enhancing the production of beneficial metabolites, stimulating the formation of antioxidant substances, improving antioxidant enzyme activity, and converting existing compounds into more active antioxidant forms (Abarshi et al., 2021). There is considerable recent interest in cell-free culture supernatants (CFCS) from probiotic bacteria, producing metabolites with biological activity excreted during fermentation. CFCS have been widely utilized as in vitro models to evaluate the antioxidant potential of probiotics. Key antioxidant components in these supernatants include exopolysaccharides (EPS), bioactive peptide, antioxidant enzymes, and manganese ions. Additionally, intestinal microbiota can produce antioxidant compounds by converting dietary substances through enzyme-driven reactions (Kim et al., 2022).
The differences observed between radical scavenging assays and reducing power highlight the multifaceted nature of antioxidant activity in probiotics. DPPH and ABTS reflect free radical neutralization, whereas FRAP measures electron-donating ability and overall reducing capacity. Therefore, the observed strain-specific differences in radical scavenging but similar reducing power indicate that distinct probiotic strains may contribute differently to oxidative stress mitigation and functional food properties. Together, these results support the prospective use of B. coagulans isolates in functional food ingredients with robust antioxidant properties, either through radical scavenging mechanism or via metabolites that enhance antioxidant capacity of the food.
Conclusion
B. coagulans F2 and P5 isolate show promising potential as probiotics with good viability in gastrointestinal conditions (acidic environments and high bile salt concentrations) and showed antimicrobial activity against E. coli, Streptococcus mutans, Listeria monocytogenes, S. aureus, and MRSA, as well as higher antioxidant activity. Both strains also showed sensitivity to the most of antibiotics assessed, which is advantageous, particularly since these antibiotics involve public health. This study suggests a low chance of driving the spread of antibiotic resistance. Collectively, the successful isolation B. coagulans from traditional Indonesian fermented fish products indicates they represents promising probiotic candidates for developing functional foods containing live beneficial cultures.








