Introduction
Shell-boring Polydora species are a major threat to the sustainability and profitability of shellfish aquaculture in Korea (Sato-Okoshi et al., 2012). These polychaetes damage the shells of economically important mollusks including oysters, mussels, scallops, clams, and abalone. Such infestations increase the hosts’ vulnerability, reduce growth, elevate the risk of secondary infections, and cause substantial economic losses due to product devaluation (Boonzaaier et al., 2014; Lee et al., 2020; Martinelli et al., 2020; Sato-Okoshi et al., 2015; Simon & Sato-Okoshi, 2015). South Korea, a global leader in shellfish production, has experienced a growing prevalence of Polydora-related problems in recent years (Lee et al., 2020; Park et al., 2021; Sato-Okoshi et al., 2012; Won et al., 2013).
The family Spionidae, particularly the Polydora complex, represents one of the most ecologically dominant groups of benthic invertebrates in aquaculture regions (Abe et al., 2019; Sato-Okoshi et al., 2015). These worms are notable for their adaptive reproductive strategies, wide substrate utilization, and strong invasive potential (Abe et al., 2019; Radashevsky & Pankova, 2006; Sato-Okoshi et al., 2023). Polydora species inhabit diverse environments, including mollusk shells, limestone, and sediments. They bore into and occupy hard substrates through both mechanical abrasion and chemical dissolution (Abe et al., 2019; Blake, 1983; Glasby & Timm, 2008; Simon & Sato-Okoshi, 2015). Their boring activity not only decreases aquaculture productivity but also facilitates the unintentional spread of invasive populations via mollusk stock translocation (Bailey-Brock, 2000; Elton, 2020; Mikac et al., 2025; Sato-Okoshi et al., 2023). Among affected countries, Korea’s dependence on intensive shellfish aquaculture increases the potential impact of Polydora infestations. Despite the persistence of these infestations, awareness and understanding of their significance remain limited within the Korean aquaculture industry.
In Korea, the presence of shell-boring Polydora was first formally documented through morphological identification by Sato-Okoshi & Abe (2012). They reported the presence of Polydora haswelli, Polydora aura, Dipolydora spp., and Polydora uncinata, the latter synonymized with Polydora hoplura (Sato-Okoshi et al., 2017), in several host species including economically important mollusks (Table 1). Specifically, P. haswelli was found in Atrina pectinata, Chlamys farreri, Crassostrea gigas, Haliotis discus, Omphalius rusticus, Pinctada fucata, and Rapana venosa. P. aura, on the other hand, was detected in C. gigas, H. discus, and A. pectinata. P. uncinata was identified exclusively in C. gigas. Won et al. (2013) confirmed P. hoplura as a major invasive species in abalone aquaculture facilities in Wando, southwest Korea. More recently, molecular analyses by Lee et al. (2020) confirmed P. haswelli and P. hoplura as the dominant shell-boring species in abalone from Wando, thereby providing genetic evidence supporting earlier morphological identifications. Further work by Lee et al. (2021) identified P. haswelli in four shellfish species from South Korea: cockle (Scapharca subcrenata), mussel (Mytilus galloprovincialis), oyster (C. gigas), and scallop (Argopecten irradians).
Data from Sato-Okoshi et al. (2012).
Accurate identification of these species is a pressing need, both for biosecurity and for mitigating economic loss. However, taxonomy remains challenging due to cryptic species complexes, intraspecific variation, and overlapping morphological characters (Radashevsky et al., 2006; Sato-Okoshi & Abe, 2012; Sato-Okoshi et al., 2012). Although morphology-based identification remains foundational, molecular techniques have proven indispensable because of high morphological similarity and pigmentation variability (Lee et al., 2020; Sato-Okoshi & Abe, 2012). Therefore, an integrative approach combining morphological and molecular methods is widely recommended for the accurate detection and management of these shell-boring species (Sato-Okoshi et al., 2017).
The review by Sato-Okoshi et al. (2015) placed principal emphasis on morphological descriptions, distribution records and the molecular phylogeny available up to that time for shell-boring polydorid worms in East Asia and Australia. In Korean waters, their earlier study (Sato-Okoshi et al., 2012) relied solely on morphological features to characterize local infestations. However, during the past decade, new molecular and integrative approaches have produced substantial evidence that challenges the accuracy of earlier identifications and highlights unresolved taxonomic uncertainties. Subsequent studies, such as those by Lee et al. (2020, 2021), reported P. haswelli as the predominant shell-boring species in Korean aquaculture systems, while only P. hoplura was detected in abalone aquaculture sites. Moreover, Sato-Okoshi et al. (2017) demonstrated through combined morphological and molecular analyses that P. hoplura and P. uncinata represent a single species. Recently, Malan et al. (2020) suggested that P. haswelli, previously considered a primary shell borer, may in fact be a tube-dwelling form, and that many records attributed to P. haswelli might correspond to Polydora neocaeca or other cryptic, shell-boring taxa. Meanwhile, many sequence records in public databases remain labeled under historical names, complicating automated identifications. This ongoing debate highlights the limitations of morphology-based taxonomy and underscores the need for integrative approaches that combine molecular, histological, and ecological data.
This review responds to these developments by integrating recent Korean and international studies to provide a comprehensive synthesis of current knowledge on shell-boring polydorids. In contrast to previous reviews that focused primarily on morphology or molecular data alone, this work adopts an interdisciplinary approach combining morphological, molecular, histological, and ecological perspectives. It further emphasizes the implications of these findings for aquaculture management and national biosecurity, addressing the prevalence, impact, and control of shell infestations. Importantly, this review introduces a standardized histological approach for assessing gonadal development and reproductive cycles in Polydora, thereby filling a critical knowledge gap in annelid reproductive biology. By linking reproductive dynamics with infestation ecology, this synthesis offers practical insights for monitoring, prevention, and sustainable management of polydorid outbreaks in Korean shellfish aquaculture.
Polydora Species and Their Impact on Shellfish
Spionid polychaetes, particularly those belonging to the polydorid complex, constitute one of the most ecologically dominant and economically detrimental benthic invertebrate groups, especially in coastal aquaculture zones. They often form the most abundant polychaete taxa in both biomass and species richness within coastal benthic ecosystems (Abe et al., 2019; Sato-Okoshi et al., 2015). The family Spionidae, described by Grube (1850), is among the most species-rich and biomass-dense annelid groups, distributed across a spectrum of marine environments from deep sea to estuarine and intertidal habitats (Abe et al., 2019; Glasby & Timm, 2008). Within this family, the polydorids comprise nine major genera; Polydora (Bosc, 1802), Dipolydora (Verrill, 1881), Pseudopolydora (Czerniavsky, 1882), Boccardia (Carazzi, 1895), Polydorella (Augener, 1914), Tripolydora (Woodwick, 1964), Boccardiella (Blake & Kudenov, 1978), Carazziella (Blake & Kudenov, 1978), and Amphipolydora (Blake, 1983); all of which share the morphological hallmark of modified major spines in the fifth chaetigerous segment (Sato-Okoshi et al., 2023; Wetzer et al., 1998).
Polydora species exhibit extensive substrate plasticity, occupying habitats ranging from mud tubes in sediments to calcareous burrows in mollusk shells, limestone, and corals (Abe et al., 2019; Blake, 1983; Simon & Sato-Okoshi, 2015). Several species are also known to form symbiotic or commensal relationships with sponges, brachiopods, and other invertebrates, sometimes even competing with hosts for food by suspension or deposit feeding (Abe et al., 2022; Sato-Okoshi et al., 2023). Their reproductive diversity and environmental adaptability contribute significantly to their invasive potential (Radashevsky & Pankova, 2006).
Polydora species cause abnormal shell formation in mollusks by creating U-shaped burrows that accumulate mud, fecal matter, and detritus, leading to visible mud blisters. These blisters significantly reduce the commercial value of shellfish, particularly in the half-shell oyster trade (Haigler, 1969; Handley & Bergquist, 1997; Martinelli et al., 2020; Shinn et al., 2015; Zottoli & Carriker, 1974). During processing, especially shucking, these blisters can rupture and contaminate the meat, rendering the product unsuitable for consumption (Martinelli et al., 2020; Morse et al., 2015; Shinn et al., 2015). This not only reduces the commercial value but also weakens shell integrity, making mollusks more vulnerable to predation and disease (Martinelli et al., 2020). In addition to the aesthetic damage, the structural integrity of the shell is weakened by heavy Polydora infestations (Sato-Okoshi et al., 2017). Infested mollusks are more prone to shell breakage and environmental stress, as well as higher predation pressure (Buschbaum et al., 2007). For instance, Littorina littorea gastropods infested with Polydora ciliata show significantly weaker shells than uninfested individuals (Buschbaum et al., 2007). Similarly, Pacific oysters infested with P. hoplura, Polydora cornuta, or Boccardia semibranchiata exhibit reduced growth rates and poorer condition indices (Chambon et al., 2007).
Physiologically, infestation imposes a metabolic burden on the host, as energy that would normally support growth and reproduction is diverted to nacre production in an attempt to isolate and seal the worm burrows (Boonzaaier et al., 2014; Kojima & Imajima, 1982; Lleonart et al., 2003; Wargo & Ford, 1993). This energy trade-off correlates with reduced glycogen and lipid stores, slower growth rates, and lower condition indices in infested C. gigas populations compared with uninfested ones (Royer et al., 2006; Wargo & Ford, 1993). In severe cases, infestation can result in significantly higher mortality rates in C. gigas within aquaculture environments (Dinamani, 1986; Lleonart et al., 2003; Owen, 1957). Reports also indicate that abalone cultivation has been halted in severely infested systems due to mass mortality and irreversible shell damage (Sato-Okoshi & Abe, 2013; Shinn et al., 2015).
Recent histological and molecular studies reveal deeper tissue-level effects beyond shell damage. A Polydora blister coverage of approximately 45% of the shell has been associated with a 36% reduction in oyster tissue condition (Wargo & Ford, 1993). In Yesso scallops, infection induced a marked increase in mucous cells within the mantle epithelium, suggesting tissue-level stress and impaired physiological function (Sun et al., 2022). These findings demonstrate that Polydora infestation affects both shell structure and soft tissue integrity.
Globally and particularly in East Asia, the rapid expansion of aquaculture has facilitated the anthropogenic translocation of mollusks and, with them, sessile and boring polychaetes such as polydorids (Bailey-Brock, 2000; Elton, 2020; Sato-Okoshi et al., 2023). This human-mediated dispersal has accelerated the spread of non-native populations, including in Korean coastal aquaculture systems where oysters, abalone, and scallops are intensively farmed (Radashevsky et al., 2006; Sato-Okoshi & Abe, 2012; Simon & Sato-Okoshi, 2015). Introduced worms can quickly establish persistent populations, reducing biodiversity and threatening production efficiency in farmed shellfish (Martinelli et al., 2020; Royer et al., 2006). They may infest native mollusks and outcompete indigenous polychaetes, altering nutrient cycling and sediment bioturbation (Bailey-Brock, 2000; Elton, 2020). Historical outbreaks, such as the collapse of oyster farms in Australia and Hawaii caused by Polydora websteri, illustrate the combined threat of economic loss and ecological disruption. These events highlight the urgent need for improved molecular, diagnostic, and management strategies. (Martinelli et al., 2020).
Despite its growth and economic significance, the sustainability of Korea’s shellfish aquaculture sector is increasingly challenged by biological and environmental threats (Kim et al., 2024). Climate change, disease outbreaks, harmful algal blooms, and biofouling organisms collectively threaten long-term productivity. Among these, shell-boring spionid polychaetes of the genus Polydora have become particularly problematic (Lee et al., 2020; Sato-Okoshi et al., 2012, 2015).
Documented cases in the Republic of Korea already point to substantial farm-level impacts from shell-boring Polydora species. Early morphological surveys confirmed infestations within oysters, pearl oysters, abalones and scallops along the southern and western coasts (Sato-Okoshi et al., 2012). Field reports from abalone farms in Wando described severe shell degradation, growth suppression and episodic mortality events consistent with heavy Polydora infestations (Won et al., 2013). Recent studies from Wando and other southern aquaculture sites confirmed the presence of P. haswelli and P. hoplura in cultured abalone, while P. haswelli was also detected across multiple bivalve hosts, including cockles, mussels, oysters, and scallops (Lee et al., 2020, 2021). This evidence indicates that the problem persists and remains underrecognized in Korean aquaculture management and monitoring programs.
Although quantitative economic data are still limited, local shellfish processors have reported downgrading of half-shell products due to blistering, increased trimming during processing, and higher batch rejection rates during peak infestation periods. These reports align with global trends showing reduced market value and poor condition indices in infested bivalves (Chambon et al., 2007; Handley & Bergquist, 1997; Martinelli et al., 2020).
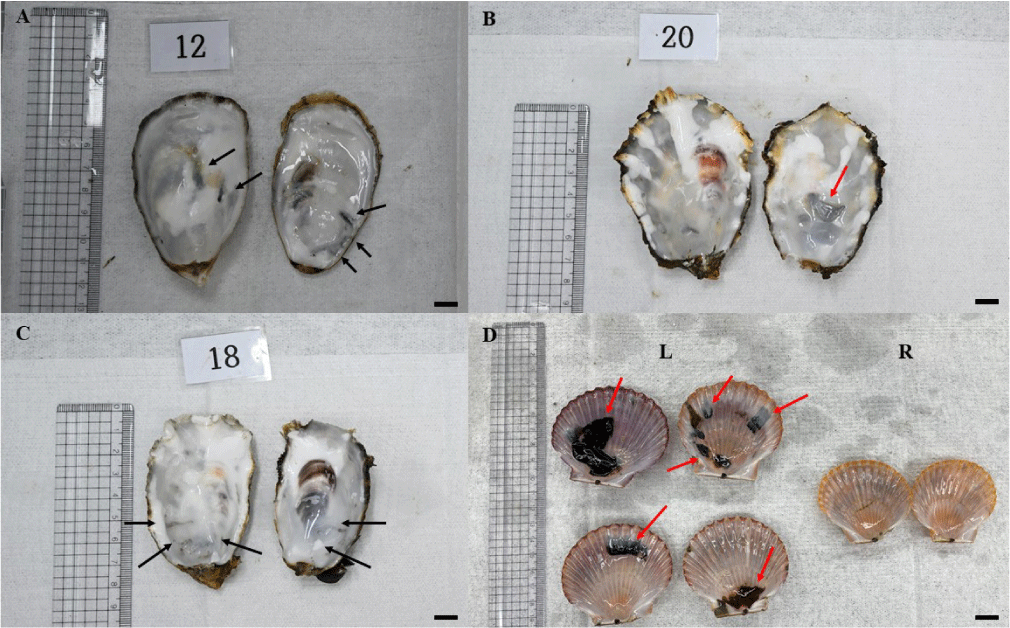
In our ongoing survey, farmed Pacific oysters (C. gigas) exhibited mean infestation prevalence of 62.67% ± 20.87 in Geoje, 56.11% ± 12.50 in Goheung, and 56.67% ± 34.12 in Namhae (n = 180 per site). By contrast, scallops (A. irradians) from Tongyeong showed higher rates at 86.11% ± 8.80, and natural oysters from the same region reached 84.17% ± 16.72. Interestingly, seed oysters sampled in Tongyeong initially showed no Polydora infestation. However, infestation rates rose sharply from 3.33% in July and abruptly reaching 100% in both August and September, highlighting how rapidly prevalence can escalate under favorable conditions. These findings provide insight into the regional prevalence and infection dynamics within Korea. Although infestations appear locally confined, the weakening of shell integrity, especially in scallops with extensive blistering, poses a major production risk (Fig. 1). With infection levels exceeding 50% across several aquaculture sites, these results underscore the urgent need for routine surveillance and control strategies to ensure the long-term sustainability of Korean shellfish farming.
Significance of Shellfish Aquaculture Management in Korea and Proposed Countermeasures
East Asia remains a global center of shellfish aquaculture, with the Republic of Korea ranking among the world’s leading producers and serving as the second-largest exporter of oysters after China. This success is supported by the nation’s extensive coastline, favorable marine environments, and well-developed aquaculture infrastructure (FAO, 2020; Park, 2025; Park et al., 2021). Over the past three decades, Korea’s aquaculture output has nearly tripled, showing an average annual growth rate of more than 6% (FAO, 2020). Shellfish farming, particularly along the southern coast of South Gyeongsang Province, is a cornerstone of both regional and national economies (FAO, 2020; Hyun et al., 2013; NASA Earth Observatory, 2019).
Pacific oysters (C. gigas) dominate Korean shellfish aquaculture, accounting for more than 75% of national production and serving as a key export commodity (MOF, 2018). In 2020, total oyster production reached approximately 300,000 tons, with Tongyeong, Korea’s primary oyster-farming region, contributing nearly 80% of this volume (NASA Earth Observatory, 2019). The ria-type coastline of Tongyeong, characterized by sheltered bays and extensive longline systems, provides ideal conditions for large-scale suspended oyster farming (Hyun et al., 2013; NASA Earth Observatory, 2019). To further enhance its global market share, the Ministry of Oceans and Fisheries has announced plans to double oyster exports to USD 160 million and raise annual production to 400,000 tons by 2030 (Park, 2025).
Beyond oysters, Korean shellfish aquaculture includes mussels (Mytilus edulis), scallops (A. irradians), Manila clams (Venerupis philippinarum), and abalone (H. discus hannai), all of which constitute major components of the nation’s mariculture output. In 2018, shellfish and seaweed aquaculture collectively removed more than 140,000 tons of carbon and nitrogen from coastal waters, highlighting the ecosystem benefits of these low-input, high-value systems (Park et al., 2021). In addition to their economic importance, cultured bivalves play vital ecological roles such as nutrient cycling, water-quality enhancement, and organic-matter sequestration, thereby supporting coastal resilience and environmental sustainability (FAO, 2020; Park et al., 2021).
Effective management of shell-boring worms in aquaculture relies on robust monitoring systems, movement controls, and farm-level hygiene protocols. In Korea, shellfish aquaculture is heavily reliant on longline suspension systems, bag culture, and rack culture of species such as C. gigas and H. discus hannai (abalone). With intensifying production and increasing seed translocation, the risk of spreading shell-boring worms remains high. However, regulated monitoring programs and translocation controls specific to polydorids are still underdeveloped.
Korea’s shellfish aquaculture is dominated by suspended long-line cultivation of Pacific oysters in South Gyeongsang and South Jeolla provinces, complemented by lantern-net culture of scallops and land-based tank systems for abalone. Although these methods maximize productivity, they also heighten the risk of biofouling, facilitating the unnoticed spread of shell-boring worms via contaminated gear, seed, and shells transferred among farms (Park et al., 2021). The Ministry of Oceans and Fisheries (MOF, 2024) recently outlined an expansion plan for shellfish aquaculture, yet formal protocols for mudworm control remain absent from biosecurity certification frameworks.
Globally, few jurisdictions have formal regulations that specifically target “mudworm” (shell-boring polychaete) translocations or outbreak monitoring. Several international case studies offer valuable management models. In the United States, the state of Washington has recently acknowledged the occurrence of shell-boring Polydora spp. and identified gaps in aquaculture regulation. Seed-transfer permits did not list polydorids as pests, and no targeted screening was required. Therefore, a coordinated review recommended tiered monitoring programs, strict seed and gear movement protocols, and rapid outbreak reporting to state agencies (Spencer et al., 2021). On the U.S. West Coast, field trials on commercial farms demonstrated that short-duration freshwater exposures and oxidative dips effectively suppressed Polydora in oysters without compromising meat quality, demonstrating how simple interventions can be adapted to local farming conditions (Martinelli et al., 2022).
Australia’s management framework provides additional insights for Korea. At the state level, these worms are classified as “significant risks,” and the movement of infected stock is tightly regulated. In New South Wales, farmer training and mitigation programs emphasize strategic air-drying and iodine baths, while Tasmania and Victoria enforce translocation controls and maintain abalone-specific management programs (Handlinger et al., 2004; Nell, 2007; Victorian Fisheries Authority, 2015). Even Canada, which once considered mudworms “negligible regulatory significance,” has begun funding studies to assess P. websteri outbreaks, showing that management priorities must evolve alongside environmental change (DFO, 2017).
Given the Korean environment and culture systems, hatchery biosecurity is especially crucial. Establishing “clean-seed” zones, performing periodic disinfection of infrastructure, and maintaining traceability systems for seed origin, nursery history, and farm transfer records are urgently needed. Farm level measures such as high-pressure cleaning (≥ 2,000 psi), periodic sun drying of gear, and air-drying of oysters followed by iodine rinsing could be readily integrated into routine operations (Martinelli et al., 2022; Nell, 2007). Routine, risk-based surveillance should be implemented, including quarterly blister scoring, standardized burrow counts, and simple polymerase chain reaction (PCR) screening of high-risk sites during larval settlement peaks. Implementing such monitoring would align Korea’s aquaculture operations with international sustainability standards.
Parallel research into mitigation strategies should also be prioritized. This includes the evaluation of water-filtration systems in nurseries, the testing of chemical or physical disinfection treatments during seed holding, and the development of shell-resistant strains through selective breeding. Predictive modeling of larval dispersal, in combination with environmental drivers such as temperature and salinity fluctuations, could enhance early-warning systems for infestation peaks. Strengthening collaboration among local fisheries offices, research institutions, and aquaculture farmers would facilitate a coordinated regional response to manage outbreaks effectively.
At the policy level, Korea can strengthen prevention by codifying a national translocation code of practice for shellfish that mirrors Victoria’s risk-based restrictions. This framework should link lease or subsidy eligibility to compliance with biosecurity plans and create a centralized reporting system for biofouling incidents. Integrating such measures into the forthcoming Aquaculture Industry Advancement Strategy (MOF, 2024) would ensure that disease prevention, stock certification, and sustainability assessments explicitly address Polydora risks. Given recent findings showing site-specific Polydora prevalence exceeding 50% in major production bays, a coordinated national management framework is urgently required. A combined approach involving movement hygiene, molecular surveillance, and seasonally targeted mitigation offers a proactive strategy for minimizing biofouling losses and maintaining the competitiveness and sustainability of Korea’s shellfish aquaculture.
Burrowing Mechanism of Polydora Species
After a planktonic larval stage, Polydora worms settle onto suitable substrates, typically calcareous shells, favoring edges, cracks, or concentric grooves on the valve surface (Martinelli et al., 2020). Settlement occurs once larvae reach approximately 17–18 setigers, guided by shell texture and curvature (Hill, 2007; Qian, 1999). The larva initially attaches near the shell margin, forms a temporary mud tube, and then penetrates the shell matrix (Hill, 2007). Penetration is achieved through a combination of mechanical abrasion and chemical dissolution (Blake & Evans, 1972; Haigler, 1969; Sato-Okoshi & Okoshi, 2000). The prostomium and modified chaetae on the fifth segment grind the shell surface (Haigler, 1969), while a viscous secretion weakens the calcium carbonate, enabling the soft-bodied worms to excavate hard substrates (Blake & Evans, 1972; Sato-Okoshi & Okoshi, 2000).
The completed burrow typically has a U-shape with two visible openings that reach the outer shell surface (Hopkins, 1958; Loosanoff & Engle, 1943). Inside, the worm lines the cavity with a tube composed of mud, shell fragments, mucus, and debris (Haigler, 1969; Handley & Bergquist, 1997). The external openings serve as both entrances and exits, facilitating feeding, respiration, waste expulsion, and sometimes gamete release (Liu & Hsieh, 2000; Qian & Chia, 1997; Radashevsky & Cárdenas, 2004).
As the worm grows, it gradually expands the burrow, penetrating deeper into the shell. This process stimulates the host to deposit additional nacreous layers that seal and isolate the tunnel (Blake & Evans, 1972; Rice et al., 2018; Zottoli & Carriker, 1974). Over time, debris and fecal matter accumulate within the cavity, forming a distinctive “mud blister” visible from the shell interior. These blisters are irregular in shape, dark in color, and significantly reduce the aesthetic and structural quality of the shell (Lezzi & Mazziotti, 2024; Medcof, 1946). A single shell may host multiple worms, each occupying its own burrow (Sato-Okoshi & Okoshi, 2000). Cases of six or more Polydora individuals per valve are not uncommon, and densities of up to 115 worms in a single scallop shell have been reported, indicating high infestation levels (Teramoto et al., 2013). In South Korea, the infestation intensity of Polydora species has been observed to range from 3–5 worms and up to 20–35 individuals per shell (Sato-Okoshi et al., 2012). Table 2 shows the number of polydorid worms infesting and isolated from cultured oyster shells in southeastern Korean waters, specifically in Goseong and Geoje.
Data from Sato-Okoshi et al. (2012).
Infestation peaks typically occur during cooler months, coinciding with periods of increased larval settlement and recruitment (Maciel et al., 2010; Mori et al., 1985; Teramoto et al., 2013). While infestations rarely cause immediate host mortality, they weaken shell structure, slow growth, and increase susceptibility to predation and secondary infections (Bergman et al., 1982; Davinack & Hill, 2022; Galosi et al., 2024; Zhang et al., 2020). In response, the host shell secretes additional layers of organic and calcareous material to block further penetration and repair the damaged area. This defensive deposition forms the characteristic “blisters” associated with Polydora infestation (Sato-Okoshi et al., 2012).
Haigler (1969) provided detailed experimental evidence of P. websteri boring behavior. The study confirmed that worms penetrate all shell layers, through a combination of repeated forward-backward scraping and localized acid secretion. Boring continued even after removal of the giant setae, indicating that mechanical rasping alone was insufficient. The acidic secretion, likely a metabolic by-product, dissolves calcium carbonate, enabling continued burrow formation.
P. ciliata exhibits a similar mechanism, boring into limestone, chalk, and shells (Casoli et al., 2019; Dorsett, 1961; Sato-Okoshi et al., 2023; Simon, 2011). Dorsett (1961) concluded that mechanical rasping explains only soft-substrate boring, while calcareous penetration must involve chemical dissolution. The mucus secreted by P. ciliata contains acidic mucopolysaccharides that chelate calcium ions and weaken the shell structure (Chughtai & Knight-Jones, 1988; Cobb, 1969). Histochemical staining with Alcian blue confirmed the presence of acidic mucosubstances in the ventral epithelium and segmental glands, indicating their role in chemical erosion. The U-shaped burrows are reinforced by sediment bound with mucus, which gradually extended through coordinated body movements that maintain tube integrity (Chughtai & Knight-Jones, 1988; Dorsett, 1961).
Further structural evidence from Polydora pygidialis, Dipolydora giardi, and Dipolydora. bidentata revealed similar dual processes. Scanning electron microscopy (SEM) showed concentric holes and worm-eaten textures, particularly in foliated shells, indicating localized acid dissolution. Parallel and lateral scratches suggested mechanical abrasion caused by setal movements (Sato-Okoshi & Okoshi, 2000). Together, these findings confirm that chemical softening and mechanical scraping act synergistically to enable burrow formation across diverse shell types.
The entire burrowing process is gradual, taking several weeks or even months to complete. The coordinated use of mechanical abrasion, chemical dissolution, and mucus secretion, along with specialized feeding adaptations, demonstrates a highly evolved strategy for colonizing hard substrates. Within these excavated tunnels, worms gain protection and feed on suspended particles and detritus using their extended palps, improving nutrition and survival (Casoli et al., 2019; Dorsett, 1961; Martinelli et al., 2020). In our ongoing study, characteristic burrow morphologies observed in oysters and scallops from the Geoje, Namhae, Goheung, and Tongyeong regions are shown in Fig. 1.
Morphology and Ecology of Polydora Species
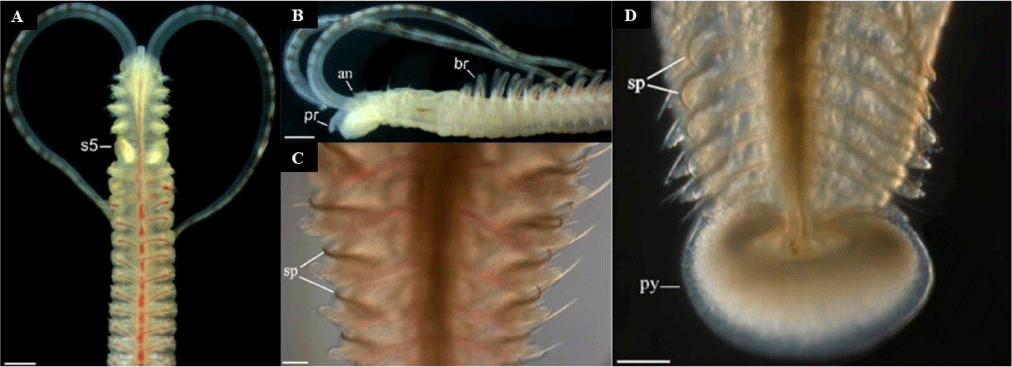
P. uncinata is a relatively large polydorid, reaching up to 40 mm in length under culture conditions (Sato-Okoshi et al., 2017). Key morphological features include prominent black bands on the palps, variable pigmentation on both the dorsal and ventral surfaces of the anterior chaetigers, a short occipital tentacle, and distinctive recurved notochaetae on posterior segments (Fig. 2). However, some individuals may exhibit lighter or absent pigmentation (Radashevsky et al., 2017; Sato-Okoshi et al., 2015). The prostomium is weakly incised, and the caruncle extends to at least the third chaetiger. The species typically has up to four eyes arranged in an irregular pattern, with palps displaying black and brown pigmented spots. Branchiae begin at the seventh chaetiger and extend posteriorly. Notably, the fifth segment bears modified falcate spines with a lateral tooth alongside pennoned chaetae, and the posterior notopodia have a recurved hook and filamentous capillary. The pygidium is disk-like with a broad dorsal opening (Radashevsky et al., 2017; Sato-Okoshi et al., 2015).
P. hoplura displays complex reproductive strategies, including poecilogony; production of larvae via both planktotrophic and adelphophagic modes (David et al., 2014, 2021; Radashevsky & Olivares, 2005). These reproductive strategies, coupled with a shortened or absent planktonic stage, enable the species to rapidly colonize new environments and maintain stable populations, especially in aquaculture systems (David et al., 2014).
The species is widely distributed across Asian and Australian waters, infesting both wild and cultured mollusks. In South Korea, heavy infestations have been reported in suspended oyster farms along the southern coast, often associated with international seed transfers (Sato-Okoshi et al., 2012; Sekino et al., 2003). Similar outbreaks in Chile followed the introduction of Japanese abalone stock, highlighting the risk of global spread and the importance of quarantine and monitoring protocols (Radashevsky & Olivares, 2005).
Overall, the poecilogonous development, environmental tolerance, and reproductive capacity of P. hoplura underpin its success as an invasive pest. This substrate-bound reproductive strategy, together with shortened pelagic phases, allows rapid colonization of local environments while maintaining the potential for long-distance dispersal through planktotrophic larvae (Radashevsky et al., 2017). Therefore, persistent infestations and rapid population growth highlight the need for continued surveillance and management in aquaculture operations worldwide.
Originally described from southeastern Australia (Blake & Kudenov, 1978), P. haswelli is now widespread, including confirmed records in New Zealand (Read, 2010; Read & Handley, 2004) and Korea (Sato-Okoshi et al., 2012). Current evidence indicates that P. haswelli is broadly distributed across the Indo-Pacific and potentially extends into the North Atlantic region. In Korea, this species is widespread, occurring in both wild and cultured shells across central and southern regions (Lee et al., 2020, 2021; Sato-Okoshi et al., 2012).
This medium to large polydorid can reach up to 45 mm in length in Korean specimens, significantly larger than those found in New Zealand, where they reach up to 28 mm (Read, 2010; Sato-Okoshi et al., 2012). Morphologically, individuals are recognizable by conspicuous black bands across the palps, spotty dark pigmentation on the anterior chaetigers, a shallowly incised prostomium, and a caruncle extending to the end of the third or mid-fourth chaetiger. Eye number varies from absent to up to four. Branchiae begin around chaetiger seven and continue posteriorly (Fig. 3). The fifth segment bears characteristic falcate spines with lateral flanges, accompanied by slender pennoned chaetae. The pygidium ranges from disk-shaped to flaring. Palps and prostomium exhibit black or brown spots in life, though pigmentation patterns may fade in preserved specimens. Live individuals consistently display distinct banding on palps and dark markings on anterior segments (Blake & Kudenov, 1978; Sato-Okoshi et al., 2012).

In Korea, P. haswelli is commonly found in both wild and cultured shellfish, with notable infestations documented in suspended oyster farms, where individuals often display black-dotted palps protruding from mud tubes. It is one of two Korean boring polydorids with black pigmented palps, which requires careful identification to avoid confusion with P. uncinata (Sato-Okoshi et al., 2012). The close morphological resemblance between P. haswelli (Australia, New Zealand, and Korea), P. cf. haswelli (Brazil), P. neocaeca (USA), Polydora limicola (Canada), and Polydora sp. (Japan) underscores the need for integrative studies combining morphological, ecological, and molecular approaches to clarify species boundaries (Radashevsky et al., 2006; Read, 2010; Sato-Okoshi & Abe, 2013; Sato-Okoshi & Okoshi, 1997; Sato-Okoshi et al., 2012).
A recent taxonomic reassessment by Malan et al. (2020) resolved a long-standing confusion surrounding P. haswelli (Blake & Kudenov, 1978), historically reported as a shell-boring species across the Indo-Pacific, including Korea. Their integrative morphological and molecular analyses demonstrated that shell-boring worms previously identified as P. haswelli are conspecific with P. neocaeca (Williams & Radashevsky, 1999), originally described from the northeastern United States. The true P. haswellisensu stricto is a sand tube-dwelling form inhabiting soft substrates rather than calcareous shells. Morphologically, P. neocaeca is distinguished by the shape of the modified spines on chaetiger 5, showing a slender main fang with a distinct flange, and by characteristic methyl-green staining patterns absent in P. haswelli (sensu stricto). Molecular data from 18S rRNA and cytochrome oxidase I (COI) sequences revealed that shell-boring specimens from Japan, China, and South Africa share identical haplotypes with P. neocaeca from Rhode Island, confirming their conspecificity. Consequently, Malan et al. (2020) recommended that all shell-boring forms previously referred to as P. haswelli be reassigned to P. neocaeca, while P. haswelli should be regarded as a tube-dwelling species. This clarification has important implications for Korean records, as most local “P. haswelli” infestations likely represent P. neocaeca, emphasizing the need for re-evaluation of archived specimens using integrative morphological and molecular diagnostics.
P. aura was first described in Japan (Sato-Okoshi, 1998) and is now prevalent in both wild and cultured shells across Korea (Sato-Okoshi et al., 2012). This species is known for creating large, elongated U-shaped burrows in abalone, fan-muscle, and oyster shells, frequently accompanied by dark deposits on the interior shell surface. Its distribution extends from western to southeastern Korea, and it is especially common in suspended culture oyster systems, easily identified by prominent mud tubes (Sato-Okoshi et al., 2012).
Morphologically, P. aura reaches up to 40 mm in length and typically displays a light orange body and palps; however, pigmentation can vary. The prostomium is weakly incised and features a short, inconspicuous occipital tentacle; the caruncle extends from the end of chaetiger 2 to 4. The species possesses one to four eyes (sometimes absent), with branchiae starting at chaetiger 7. On chaetiger 5, falcate spines alternate with pennoned chaetae, with ventral chaetae only on this segment. The posterior chaetigers are characterized by tightly bundled, cylindrical, short needle-like notochaetae, which do not protrude through the cuticle, together with long and short capillaries. The pygidium forms a wide, flaring disk (Sato-Okoshi et al., 2015; Won et al., 2013).
Ecologically, the shell-boring behavior and morphology of Korean P. aura closely resemble conspecifics from Japan and Australia (Sato-Okoshi, 1998; Sato-Okoshi et al., 2008). However, Korean and Australian populations primarily invade mollusk shells, whereas Japanese populations also bore into coralline algae (Sato-Okoshi, 2000). Additionally, Korean and Australian specimens produce discrete egg capsules, unlike the stringed capsules of Japanese populations, indicating possible ecological divergence and reproductive differentiation (Sato-Okoshi et al., 2012). A comparison of key morphological characters among three prominent species, P. haswelli, P. aura, and P. uncinata, occurring in Korean waters, is summarized in Table 3.
Data from Sato-Okoshi et al. (2012).
Among the lesser-known spionid polychaetes in Korean waters, Sato-Okoshi et al. (2012) reported a small, slender species of Dipolydora that closely resembling Dipolydora alborectalis described by Radashevsky (1993). These worms reach up to 10 mm in length and exhibit a tan, unpigmented body, bilobed prostomium, a caruncle extending to chaetiger 3, and a pygidium divided into four equal lobes. No special notochaetae are present on posterior segments. Ecologically, this species is uncommon and highly localized, collected only in low numbers from wild C. farreri scallop shells in Taean, western Korea. These scallops attach via byssal threads, and multiple Dipolydora individuals sometimes cohabit a single narrow, complexly branched burrow with multiple openings. Reproductive structures were absent during the May collections, potentially indicating seasonal reproduction or reproductive inactivity. This species shows close morphological and ecological similarity to Dipolydora concharum but differs by its smaller size and more intricate burrow branching (Sato-Okoshi, 2000; Sato-Okoshi et al., 2012).
Another Dipolydora species, D. giardi, is more frequently encountered and reaches up to 8 mm in length. Individuals are light tan, lack pigmentation and eyes, and develop branchiae starting from chaetiger 9 or 10. The pygidium has a disk-like shape, typically with a larger ventral lobe and two smaller dorsal lobes, which sometimes fuse. D. giardi is commonly found in the hinge areas of subtidal mussels (Mytilus coruscus) in Taean and, less frequently, in R. venosa shells from Wando. They exhibit patchy distributions and secrete strong mucus, granting a sticky texture and adherence to shell surfaces. While reproductive characteristics of Korean D. giardi populations have not been described, their widespread occurrence indicates a stable presence in local benthic communities. Although never as abundant as dominant shell borers like P. haswelli or P. aura, D. giardi contributes to the diversity of boring polychaete assemblages along Korean coasts (Sato-Okoshi et al., 2012).
Global Molecular Approaches Based on 18S rRNA gene and Cytochrome Oxidase I and Evidence for Shell-Boring Polydora in Korea
Accurate identification of shell-boring Polydora species is essential for monitoring and managing mollusk aquaculture infestations. Morphological identification alone is often unreliable because of high intraspecific variation and overlapping diagnostic features among closely related species (Lee et al., 2020). For this reason, molecular markers, especially nuclear 18S rRNA gene and mitochondrial COI (or cox1), have become indispensable in resolving taxonomic ambiguity and elucidating phylogenetic relationships within the genus (Hall et al., 2004; Sato-Okoshi & Abe, 2012).
Early studies demonstrated the value of 18S rRNA for species-level discrimination in Polydora. Specific primer sets such as 18S-1F1/1R632, 18S-2F576/2R1209, and 18S-3F1129/R1772 successfully differentiated morphologically similar taxa, confirming the robustness of this region for interspecific resolution (Sato-Okoshi & Abe, 2012; Teramoto et al., 2013). These primers had previously been applied by Nishitani et al. (2012) in their study on kleptoplastidy in the dinoflagellate Dinophysis mitra, demonstrating their versatility. They enabled successful amplification and species-level discrimination of Polydora brevipalpa, P. uncinata, and P. aura (Sato-Okoshi & Abe, 2012), as well as P. onagawaensis (Teramoto et al., 2013). Subsequent phylogenetic reconstructions using these primers confirmed their specificity and reliability, clarifying relationships such as the sister-taxon status of P. websteri, Polygala calcareia, and P. haswelli (Sato-Okoshi & Abe, 2013).
To resolve the longstanding confusion between P. uncinata and P. hoplura, further molecular work expanded analyses to include nuclear 28S rRNA, mitochondrial 16S, and cytochrome b (cyt b) genes. Nuclear gene analyses (18S, 28S) revealed complete identity between P. uncinata and P. hoplura, while mitochondrial genes showed some variation but no diagnostic differences. Shared haplotypes across samples from Japan, Australia, and South Africa confirmed that these taxa are conspecific, leading to their formal synonymization (Sato-Okoshi et al., 2017).
The combined use of 18S rRNA and mitochondrial COI has proven valuable in other contexts as well. For example, Martinelli et al. (2020) used these markers, including the Dorid_COI.3F and Dorid_COI.1R primer pair (Williams et al., 2017) targeting the COI region, and the 18S-1F1/18S-1R632 pair targeting 18S rRNA, to confirm the identity of P. websteri in Washington State. The COI marker showed zero intraspecific divergence, while the 18S marker provided clear species-level discrimination despite greater sequence variability.
Despite its widespread application as a standard marker for DNA barcoding across the animal kingdom (Aksöyek et al., 2016), the mitochondrial COI marker has notable limitations in polydorid and other annelids. Universal COI primers often fail to amplify target regions; they can generate non-specific products or amplify host DNA in shell-dwelling polychaetes (Mioduchowska et al., 2018; Sharma & Kobayashi, 2014). Host DNA contamination remains a recurring issue because polychaetes live embedded in mollusk tissue, making pure DNA extraction difficult (Mioduchowska et al., 2018). These limitations were also observed in the study by Lee et al. (2020, 2021), where conventional universal COI primers consistently showed weak amplification or failed to produce PCR products. In contrast, the Polydora-specific cox1 (cox1-Polyd-F1/R1 and cox1-Polyd-F1/R1R2) reliably produced 441 bp and 466 bp amplicons, respectively, with the latter showing the highest efficiency. Because molecular studies of shell-boring Polydora species in Korean waters remain limited, their work represents the first comprehensive molecular confirmation of P. haswelli and P. hoplura in Korea using mitochondrial and nuclear markers, including cox1, 18S rDNA, and internal transcribed spacer (ITS) regions. Although ITS primers yielded non-specific bands, 18S rDNA amplified successfully with the A/SSU-inR1 primer pair, producing clear 584 bp sequences suitable for diagnostic analysis. This framework provides a reliable approach for accurate species-level identification of shell-boring Polydora in Korean aquaculture.
Overall, the combined nuclear 18S rRNA gene and Polydora-specific mitochondrial cox1 markers represent the most robust and complementary molecular strategy for precise identification and surveillance of Polydora species. These markers should be prioritized in diagnostic programs and molecular surveillance frameworks to monitor and trace the biogeographic spread of invasive shell-boring taxa in aquaculture environments.
Reproductive Biology of Polychaetes
Although various aspects of the life cycle of Polydora species, such as larval development, egg capsule morphology, and reproductive timing have been documented, most studies emphasize external morphology, developmental observations, or molecular identification. To date, no histological studies have thoroughly detailed the internal reproductive structures of Polydora.
P. hoplura remains one of the best-studied examples of reproductive diversity within the genus. It exhibits poecilogony, a rare condition in which a single species produces both planktotrophic and adelphophagic larvae (David et al., 2014). According to David et al. (2014), females deposit long strings of egg capsules attached to burrow walls by fine filaments. In the planktotrophic pathway, eggs are small and pale, cleavage begins within 48 hours, and by the second day, larvae with defined prototrochs and eyespots appear. By day 8, larvae reach the three-chaetiger stage with functional ciliary bands and swimming chaetae. They are released to feed on suspended algae, developing through 16 to 18 chaetigers over about 40 days before settlement. In contrast, the adelphophagic mode involves larger, yolk-rich eggs, often consuming nurse eggs enclosed within the capsules. This mode enables a shorter developmental period, with settlement occurring in approximately 16 to 17 days. Molecular analyses show that both developmental types share identical haplotypes, confirming they belong to one polymorphic species rather than distinct taxa. This reproductive flexibility enables P. hoplura to adapt to diverse environmental conditions, contributing to its persistence and dominance in aquaculture systems. The predominance of either developmental pathway appears to depend on environmental and maternal factors, reflecting the species’ remarkable adaptive capacity.
Similar to P. hoplura, reproduction in P. haswelli (actually reflecting traits of P. neocaeca) involves deposition of encapsulated eggs within self-excavated burrows, typically occurring during late spring and early winter months (May, November, and December) (Sato-Okoshi et al., 2012). However, unlike P. hoplura, the reproductive mode of P. haswelli is strictly planktotrophic, with no evidence of poecilogony. These capsules form elongated strings attached to burrow walls, with synchronous development across all eggs. By December, embryos typically reach the morula stage, and by May, larvae bearing two eyespots are released. Females attach egg capsules to the burrow walls in linked strings (Radashevsky et al., 2006). The number of eggs per capsule varies, but related species often contain one to four eggs per capsule (Radashevsky, 1994; Radashevsky et al., 2006). Larvae develop intracapsularly to the three-chaetiger stage before entering a planktonic phase, feeding actively until they reach 16 to 18 chaetigers and settle. After settlement, juveniles initially build silty tubes before boring into calcareous shells.
Larval development of P. aura is also planktotrophic, but detailed information on other reproductive features remains scarce (Sato-Okoshi, 2015). Polydora robi females also produce egg capsules attached to burrow walls, each containing fertilized eggs that develop into larvae without nurse eggs. Larval release occurs at the three-segment stage after 4.6 to 7.5 days of development. Microscopic and SEM observations reveal gradual differentiation, including yolk mass reduction, paired eyespot formation, and elongation of swimming chaetae. This developmental pattern illustrates the conservative nature of early ontogeny among shell-boring polydorids (Williams, 2001).
Additionally, ultrastructural investigations of P. ciliata have provided important insight into spermiogenesis using transmission electron microscopy. Gametogenic segments exhibit a continuum from spermatocytes to mature spermatozoa, marked by nuclear elongation, chromatin condensation into dense fibrils, centriolar fossa formation, acrosomal vesicle development, and mitochondrial reorganization (Gao et al., 2014). The mature sperm conform to the “direct sperm-transfer” type common among spionids, indicating functional and evolutionary conservation of fertilization mechanisms across Polydora species. These cytological features underscore the genus’s capacity to maintain efficient reproductive output under environmental stress, reinforcing its success as a dominant shell-boring group in marine ecosystems.
Despite the major economic impact of Polydora species on shellfish aquaculture, no comprehensive histological descriptions of their reproductive systems currently exist. Only one published account of histological work on a Polydora species: Bin et al. (2015) described the histology of P. brevipalpa. Their study found that the body structure is primarily composed of epidermis, musculature, and the digestive system, with a high concentration of gland cells in the cuticular layers. They also reported a well-developed musculature and detailed organization of the alimentary tract, including distinct glandular pouches. To date, no published studies have described gonadal development in any Polydora species through detailed histological analysis. This represents a major gap, given the crucial role of reproductive biology in understanding population dynamics, life cycles, and the timing of infestation events essential for effective aquaculture management.
This deficiency extends beyond Polydora, reflecting a broader lack of histological research across annelid taxa. Much of the current knowledge is inferred from related spionids and serpulids, where detailed histological protocols and analyses have been developed. For example, MacCord & Amaral (2007) established a robust histological framework for Scolelepis goodbodyi, identifying stages of gonadal development and emphasizing the importance of histology for recognizing reproductive phases not evident from external morphology. This approach revealed six distinct gonadal development stages: Stage 1 (Initial Development) marked by pre-vitellogenic oocytes and abundant primary spermatocytes; Stage 2 (Proliferation) showing increased gametogenic setigers and presence of some mature gametes; Stage 3 (Premature) where coelomic spaces were nearly filled with mature gametes; Stage 4 (Mature), with complete filling of mature oocytes or spermatozoa; Stage 5 (Partially Spawning), characterized by partial depletion of gametes due to spawning; and Stage 6 (Recovery), a post-spawning phase with empty or regenerating gonads. Key observations revealed continuous gametogenesis without strict synchrony, as multiple reproductive stages were present within the population each month. Development was also asynchronous between sexes, with males more frequently in early stages and females more often in proliferative or spawning phases.
Similarly, Cotter et al. (2003) documented the reproductive cycles of serpulid polychaetes Pomatoceros lamarckii and Pomatoceros triqueter worms from Bantry Bay, southwest Ireland, using monthly histological sampling. They classified the reproductive development microscopically into seven stages: Stage 0 (Inactive), Stage 1 (Early Developing), Stage 2 (Late Developing), Stage 3 (Ripe), Stage 4 (Spawning), Stage 5 (Postspawning), and Stage 6 (Resorbing) (Table 4). Gametogenesis was extraovarian, with spermatogonia released from testes and oocytes maturing freely in the coelomic cavity. Ripe individuals were observed throughout the year, suggesting an extended breeding season with multiple reproductive peaks rather than a single annual cycle.
Data from Cotter et al. (2003).
Building on these models and the standard histological approaches outlined by Howard et al. (2004), our current work aims to develop standardized histological protocols to investigate reproductive development in shell-boring Polydora species from Korean coastal waters. This approach adapts methods previously used for Spionidae and Serpulidae, involving careful specimen relaxation and fixation, embedding, sectioning, and staining for microscopic examination of gonadal tissue. By establishing clear histological criteria for gametogenic staging, this research will provide new insights into Polydora reproductive ecology, inform aquaculture management, and facilitate targeted control during periods of peak reproductive activity.
Proposed Histological Method for Observing Gonadal Development in Polydora spp.
The overall workflow for histological preparation and staining of Polydora specimens is illustrated in Fig. 4. Specimens were collected directly from infected mollusk hosts (mainly oysters and scallops) by carefully cracking the shells and preserving the soft tissues for histological analysis. To ensure optimal preservation of gonadal structures, live worms were gently extracted from their burrows, as Polydora tissues are extremely fragile and easily damaged. We recommend trimming the burrow margins and carefully opening them to access the worm, followed by immediate anesthetization in a 7% magnesium chloride (MgCl₂) solution to minimize physiological stress and gamete disruption.
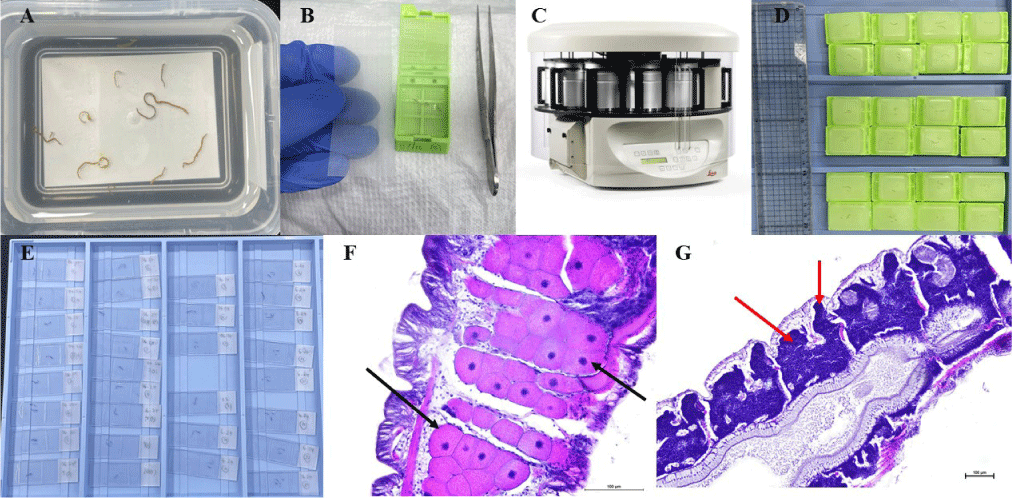
Relaxed specimens were fixed in Davidson’s fixative for at least 24 hours at room temperature to optimize preservation of gonadal architecture. After fixation, tissues were transferred to 70% ethanol for storage (Cotter et al., 2003, Howard et al., 2004).
Tissue processing, which prepares the sample for paraffin embedding through dehydration and clearing (Del Valle, 2022), was carried out using an automated tissue processor (Yang et al., 2021). Dehydration followed the method of Howard et al. (2004) with slight modifications, using a graded ethanol series (70%; 80%; 95%; and 100%), followed by clearing in xylene. Care was taken to avoid excessive heat and prolonged ethanol exposure, as these conditions can cause over-dehydration, leading to structural distortion and poor staining quality. Complete xylene clearing and adequate paraffin infiltration were essential to ensure proper tissue preservation and facilitate fine sectioning. Insufficient paraffin penetration may result in excessive tissue shrinkage and desiccation, compromising histological integrity (Suvarna et al., 2019).
Embedding was performed using a modular embedding center equipped with a paraffin dispenser, cooling plate, and cassette storage (Suvarna et al., 2019). After embedding, paraffin blocks were stored in a refrigerator to maintain tissue stability (Howard et al., 2004). During the sectioning process, blocks were kept in an ice bath to facilitate consistent cutting. Embedded specimens were sectioned at a thickness of 4–6 µm using a rotary microtome (Yang et al., 2021). Due to the delicate nature of Polydora tissues, we recommend a manual approach to sectioning rather than automated trimming. Automatic paraffin removal can easily dislodge or damage fragile specimens. Therefore, the paraffin surrounding the tissue block should first be manually trimmed, and sections should be cut slowly and precisely to preserve structural integrity. Sections were then floated on a 70% ethanol solution and carefully transferred onto a heated water bath to allow even tissue spreading on glass slides (Kumar et al., 2012). The mounted slides were dried overnight at 42°C in preparation for subsequent staining procedures (Howard et al., 2004).
On the following day, hematoxylin and eosin (H&E) staining was performed. Slides were first incubated on a slide dryer at 60°C for at least 10 minutes until the paraffin melted completely. Deparaffinization was achieved by immersing the slides in xylene for approximately 1 minute and 30 seconds, followed by several dips (3–4 times) in a 1:1 mixture of xylene and 100% ethanol until the tissue sections turned white. Rehydration was carried out through sequential immersion in decreasing concentrations of ethanol (100%, 95%, 80%, and 70%), each lasting 5–10 seconds, followed by rinsing in tap water for 3–5 minutes. Hematoxylin staining was carried out for 5 minutes, followed by a 1-minute water rinse, a 30-seconds immersion in bluing solution, and another 1-minute rinse in water. Eosin staining was performed for 3 minutes, after which the undersides of the slides were gently wiped clean. Dehydration was completed by sequential dips in 70%, 80%, and 95% ethanol (5–10 seconds each), followed by a 20-second dip in 100% ethanol. To remove residual ethanol, slides were dipped 3–4 times in a 1:1 mixture of xylene and ethanol. Finally, slides were stored in xylene until cover slipping was completed using Canada balsam or Malinol as the mounting medium. Since both H&E are water-soluble dyes, effective deparaffinization with xylene and proper rehydration are essential before staining (Yang et al., 2021). Hematoxylin, a basic dye, binds selectively to acidic structures such as nucleic acids, producing blue or purple-stained nuclei. Eosin, an acidic dye, stains basic components such as cytoplasm and muscle fibers pink to red, enhancing overall tissue contrast under light microscopy (Dey, 2022; Fischer et al., 2008). This optimized workflow ensures high-quality histological sections suitable for identifying gametogenic stages and evaluating reproductive development in shell-boring Polydora.
Conclusion and Future Perspectives
Shell-boring Polydora species remain a persistent and economically significant challenge to the sustainability of shellfish aquaculture in Korean coastal waters. The economic losses resulting from shell damage, reduced growth, and product devaluation highlight the urgent need for continued research and targeted management measures. The cryptic morphology of these worms, coupled with their wide host range and adaptive reproductive strategies, makes detection, identification, and control particularly difficult for aquaculture producers and resource managers.
Over the past decade, notable progress has been achieved in the molecular identification and monitoring of Polydora species through mitochondrial cox1 and nuclear 18S rRNA gene markers. These molecular tools have significantly improved the reliability of species discrimination and deepened our understanding of infestation pathways and population connectivity. Nevertheless, major knowledge gaps remain, particularly in the histological characterization of reproductive development. Establishing standardized histological protocols will be vital for defining gonadal stages, understanding reproductive cycles, and identifying environmental triggers linked to outbreak dynamics.
Looking ahead, a multidisciplinary and integrative research framework will be essential. Future studies should expand both molecular and histological surveys across multiple farming regions and seasons, linking host distribution, environmental parameters, and reproductive timing. In addition, developing rapid, field-deployable molecular diagnostics and evaluating environmentally sustainable control methods could substantially improve detection and management of Polydora infestations. By addressing these research priorities, Korean aquaculture can move toward more proactive management strategies, minimizing economic losses while enhancing global understanding of shell-boring polychaete ecology and evolution.








